UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
FORM
(Mark One)
For the fiscal year ended
OR
For the transition period from: __________ to __________
Commission File Number:

(Exact name of registrant as specified in its charter)
| (State or other jurisdiction of incorporation or organization) | (I.R.S. Employer Identification No.) |
| (Address of Principal Executive Offices) | (Zip Code) |
Registrant’s telephone number, including
area code:
Securities registered pursuant to Section 12(b) of the Act:
| Title of each class | Trading Symbol(s) | Name of each exchange on which registered | ||
| The | ||||
| The |
Securities registered pursuant to Section 12(g) of the Act:
None.
Indicate by check mark if the registrant is a
well-known seasoned issuer, as defined in Rule 405 of the Securities Act. Yes ☐
Indicate by check mark if the registrant is not
required to file reports pursuant to Section 13 or Section 15(d) of the Act. Yes ☐
Indicate by check mark whether the registrant
(1) has filed all reports required to be filed by Section 13 or 15(d) of the Securities Exchange Act of 1934 during the
preceding 12 months (or for such shorter period that the registrant was required to file such reports), and (2) has been subject
to such filing requirements for the past 90 days.
Indicate by check mark whether the registrant
has submitted electronically every Interactive Data File required to be submitted pursuant to Rule 405 of Regulation S-T (§ 232.405
of this chapter) during the preceding 12 months (or for such shorter period that the registrant was required to submit and post such
files).
Indicate by check mark whether the registrant is a large accelerated filer, an accelerated filer, a non-accelerated filer, a smaller reporting company, or an emerging growth company. See the definitions of “large accelerated filer,” “accelerated filer” and “smaller reporting company” and “emerging growth company” in Rule 12b-2 of the Exchange Act.
| Large accelerated filer | ☐ | Accelerated filer | ☐ |
| ☒ | Smaller reporting company | ||
| Emerging growth | |||
If an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards provided pursuant to Section 13(a) of the Exchange Act. ☐
Indicate by check mark whether the registrant
has filed a report on and attestation to its management’s assessment of the effectiveness of its internal control over financial
reporting under Section 404(b) of the Sarbanes-Oxley Act (15 U.S.C. 7262(b)) by the registered public accounting firm that prepared or
issued its audit report.
If securities are registered pursuant to Section
12(b) of the Act, indicate by check mark whether the financial statements of the registrant included in the filing reflect the correction
of an error to previously issued financial statements.
Indicate by check mark whether any of those error corrections are restatements that required a recovery analysis of incentive-based compensation received by any of the registrant’s executive officers during the relevant recovery period pursuant to §240.10D-1(b). ☐
Indicate by check mark whether the registrant
is a shell company (as defined in Rule 12b-2 of the Exchange Act). Yes ☐ No
The aggregate market value of the voting and non-voting
common stock held by non-affiliates of the registrant as of the last business day of the registrant’s most recently completed second
fiscal quarter was $
As of March 31, 2025, there were
DOCUMENTS INCORPORATED BY REFERENCE
Portions of the registrant’s definitive proxy statement relating to its 2025 annual meeting of shareholders (the “2025 Proxy Statement”) are incorporated by reference into Part III of this Annual Report on Form 10-K where indicated. The 2025 Proxy Statement will be filed with the U.S. Securities and Exchange Commission within 120 days after the end of the fiscal year to which this report relates.
| Auditor Firm Id: | Auditor Name: | Auditor Location: |
| Auditor Firm Id: 688 | Auditor Name: Marcum, LLP | Auditor Location: San Francisco, CA. |
TABLE OF CONTENTS
i
Cautionary Statement Regarding Forward-Looking Information
This Annual Report on Form 10-K (this “Report”) contains forward-looking statements under federal securities laws, including within the meaning of the Private Securities Litigation Reform Act of 1995. In some cases, you can identify forward-looking statements by the following words: “anticipate,” “believe,” “continue,” “could,” “estimate,” “expect,” “intend,” “may,” “ongoing,” “plan,” “potential,” “predict,” “project,” “should,” or the negative of these terms or other comparable terminology, although not all forward-looking statements contain these words. Forward-looking statements are not a guarantee of future performance or results and will not necessarily be accurate indications of the times at, or by, which such performance or results will be achieved. Forward-looking statements are based on information available at the time the statements are made and involve known and unknown risks, uncertainties and other factors that may cause our results, levels of activity, performance or achievements to be materially different from the information expressed or implied by the forward-looking statements in this Report, including those described under the heading “Risk Factors” contained in Item 1A of this Report.
In particular, forward-looking statements include, but are not limited to, any statements that are not statements of current or historical facts, such as statements relating to our expectations for the clinical and preclinical development, manufacturing, regulatory approval, and commercialization of our product candidates, the accuracy of our estimates regarding expenses, future revenues and capital requirements, our ability to execute our plans to develop and market new drug products and the timing and costs of these development programs, and estimates of the sufficiency of our existing capital resources combined with future anticipated cash flows to finance our operating requirements.
Forward-looking statements include, but are not limited to, statements about:
| ● | The need for additional funding, including that our current cash on hand is expected to last us until only December 2025, if we do not raise funding in the future; |
| ● | our ability to create or purchase a front end for our back end blockchain casino intellectual property assets, commercialize our planned iGaming casino, obtain required licenses and customers, and come to mutually agreeable contractual terms with third parties and suppliers, and ultimately generate revenues through such operations; |
| ● | expectations for the clinical and preclinical development, manufacturing, regulatory approval, and commercialization of our biotechnology focused product candidates; |
| ● | the uncertainties associated with the clinical development and regulatory approval of the Company’s drug candidates, including potential delays in the enrollment and completion of clinical trials, issues raised by the U.S. Food and Drug Administration (FDA), the European Medicines Agency (EMA) and the U.K. Medicines and Healthcare products Regulatory Agency (MHRA); |
| ● | regulatory developments in the United States and foreign countries; |
| ● | our success in retaining or recruiting, or changes required in, our officers, key employees or directors; |
| ● | current negative operating cash flows and our potential ability to obtain additional financing to advance our business and the terms of any further financing, which may be highly dilutive and may include onerous terms; |
| ● | the accuracy of our estimates regarding expenses, future revenues and capital requirements; |
| ● | the Company’s reliance on third parties to conduct its clinical trials, enroll patients, and manufacture its preclinical and clinical drug supplies, the ability to come to mutually agreeable terms with such third parties and partners, and the terms of such agreements; |
ii
| ● | the terms of the Company’s current licensing agreements, and the termination rights associated therewith; |
| ● | estimates of patient populations for the Company’s planned products; |
| ● | unexpected adverse side effects or inadequate therapeutic efficacy of drug candidates that could limit approval and/or commercialization, or that could result in recalls or product liability claims; |
| ● | the Company’s ability to fully comply with numerous federal, state and local laws and regulatory requirements, as well as rules and regulations outside the United States, that apply to its product development activities; |
| ● | challenges and uncertainties inherent in product research and development, including the uncertainty of clinical success and of obtaining regulatory approvals; and uncertainty of commercial success; |
| ● | the ability of the Company to execute its plans to develop and market new drug products and the timing and costs of these development programs; |
| ● | changing rates of inflation and interest rates, and economic downturns, including potential recessions, as well as macroeconomic, geopolitical, health and industry trends, pandemics, acts of war (including the ongoing Ukraine/Russian conflict, and Israel/Hamas conflict) and other large-scale crises; |
| ● | estimates of the sufficiency of our existing capital resources combined with future anticipated cash flows to finance our operating requirements; |
| ● | the review and evaluation of strategic transactions and their impact on shareholder value; the process by which the Company engages in evaluation of strategic transactions; the outcome of potential future strategic transactions and the terms thereof; |
| ● | our ability to maintain our listing of our common stock and public warrants on the Nasdaq Capital Market; |
| ● | the reliance on suppliers of third-party gaming content and the cost of such content, and reliance on third-party service providers for our planned iGaming platform, including but not limited to marketing, customer service and data management services; |
| ● | the ability of the Company to obtain gaming licenses; |
| ● | the Company’s reliance on its management; |
| ● | the potential effect of economic downturns, recessions, tariffs, changes in interest rates and inflation, and market conditions, including recessions, decreases in discretionary spending and therefore demand for our products, and increases in the cost of capital, related thereto, among other affects thereof, on the Company’s operations and prospects as a result of increased inflation, tariffs, increasing interest rates, global conflicts and other events; |
| ● | the Company’s ability to protect its proprietary information and intellectual property (IP); |
| ● | the ability of the Company to compete in the iGaming market; |
| ● | the effect of current and future regulation, the Company’s ability to comply with regulations (both current and future) and potential penalties in the event it fails to comply with such regulations and changes in the enforcement and interpretation of existing laws and regulations and the adoption of new laws and regulations that may unfavorably impact our business; |
| ● | the ability to compete against existing and new competitors; |
| ● | the ability to manage expenses associated with sales and marketing and necessary general and administrative and technology investments; |
| ● | general consumer sentiment and economic conditions that may affect levels of discretionary customer purchases of the Company’s products, including potential recessions and global economic slowdowns; and |
| ● | other risks and uncertainties, including those described under “Risk Factors”, herein. |
Any forward-looking statements in this Report reflect our current views with respect to future events or to our future financial performance and involve known and unknown risks, uncertainties and other factors that may cause our actual results, performance or achievements to be materially different from any future results, performance or achievements expressed or implied by these forward-looking statements. Given these uncertainties, you should not place undue reliance on these forward-looking statements. All forward-looking statements included herein speak only as of the date of the filing of this Report. All subsequent written and oral forward-looking statements attributable to the Company, or persons acting on its behalf, are expressly qualified in their entirety by the cautionary statements above. Except as required by law, we assume no obligation to update or revise these forward-looking statements for any reason, even if new information becomes available in the future.
iii
PART I
Item 1. Business.
General
The information included in this Annual Report on Form 10-K should be read in conjunction with the consolidated financial statements and related notes included at the end of this report.
Our logo and some of our trademarks and tradenames are used in this Report. This Report also includes trademarks, tradenames and service marks that are the property of others. Solely for convenience, trademarks, tradenames and service marks referred to in this Report may appear without the ®, ™ and SM symbols. References to our trademarks, tradenames and service marks are not intended to indicate in any way that we will not assert to the fullest extent under applicable law our rights or the rights of the applicable licensors if any, nor that respective owners to other intellectual property rights will not assert, to the fullest extent under applicable law, their rights thereto. We do not intend the use or display of other companies’ trademarks and trade names to imply a relationship with, or endorsement or sponsorship of us by, any other companies.
The market data and certain other statistical information used throughout this Report are based on independent industry publications, reports by market research firms or other independent sources that we believe to be reliable sources; however, we have not commissioned any of the market or survey data that is presented in this Report. Industry publications and third-party research, surveys and studies generally indicate that their information has been obtained from sources believed to be reliable, although they do not guarantee the accuracy or completeness of such information. We are responsible for all of the disclosures contained in this Report, and we believe these industry publications and third-party research, surveys and studies are reliable. While we are not aware of any misstatements regarding any third-party information presented in this Report, their estimates, in particular, as they relate to projections, involve numerous assumptions, are subject to risks and uncertainties, and are subject to change based on various factors, including those discussed under the section entitled “Risk Factors” of this Report. These and other factors could cause our future performance to differ materially from our assumptions and estimates. Some market and other data included herein, as well as the data of competitors as they relate to 180 Life Sciences Corp., is also based on our good faith estimates.
Our fiscal year ends on December 31. Interim results are presented on a quarterly basis for the quarters ended March 31, June 30, and September 30, the first quarter, second quarter and third quarter, respectively, with the quarter ended December 31st being referenced herein as our fourth quarter. Fiscal 2024 means the year ended December 31, 2024, whereas fiscal 2023 means the year ended December 31, 2023.
Reverse Stock Splits
Effective on December 19, 2022 at 12:01 a.m. Eastern Time, we affected a 1-for-20 reverse stock split of our then outstanding common stock, with any fractional shares rounded up to the nearest whole share. Effective on February 28, 2024 at 12:01 a.m. Eastern Time, we affected a 1-for 19 reverse stock split of our then outstanding common stock with any fractional shares rounded up to the nearest whole share.
In connection with the reverse splits discussed above (the “Reverse Stock Splits”), all outstanding options, warrants, and other securities entitling their holders to purchase or otherwise receive shares of common stock were adjusted, as required by the terms of each security. The number of shares available to be awarded under the Company’s equity incentive plans were also appropriately adjusted. Following the Reverse Stock Splits, the par value of the common stock remained unchanged at $0.0001 par value per share. The Reverse Stock Splits did not change the authorized number of shares of common stock or preferred stock.
The effects of the Reverse Stock Splits have been retroactively reflected throughout this Report.
1
Definitions
Unless the context requires otherwise, references to the “Company,” “we,” “us,” “our,” “180 Life”, “180LS” and “180 Life Sciences Corp.” refer specifically to 180 Life Sciences Corp. and its consolidated subsidiaries. References to “KBL” refer to the Company prior to the November 6, 2020 Business Combination (discussed and defined below).
In addition, unless the context otherwise requires and for the purposes of this Report only:
| ● | “CAD” refers to Canadian dollars; | |
| ● | “Exchange Act” refers to the Securities Exchange Act of 1934, as amended; | |
| ● | “£” or “GBP” refers to British pounds sterling; | |
| ● | “SEC” or the “Commission” refers to the United States Securities and Exchange Commission; and | |
| ● | “Securities Act” refers to the Securities Act of 1933, as amended. |
Where You Can Find Other Information
We file annual, quarterly, and current reports, proxy statements and other information with the SEC. Our SEC filings are available to the public over the Internet at the SEC’s website at www.sec.gov and are available for download, free of charge, soon after such reports are filed with or furnished to the SEC, on the “Investors”—“SEC Filings”—“All SEC Filings” page of our website at www.180lifesciences.com. Copies of documents filed by us with the SEC are also available from us without charge, upon oral or written request to our Secretary, who can be contacted at the address and telephone number set forth on the cover page of this Report. Our website address is www.180lifesciences.com. The information on, or that may be accessed through, our website is not incorporated by reference into this Report and should not be considered a part of this Report.
Our Company
We were formed as a clinical stage biotechnology company, focused on the development of therapeutics for unmet medical needs in chronic pain, inflammation and fibrosis by employing innovative research, and, where appropriate, combination therapy. In September 2024, we completed the acquisition of certain source code and intellectual property relating to the back-end technology platform to operate an online blockchain casino, and moving forward, we plan to focus the majority of our operations on the creation of the online blockchain casino, while looking to monetize certain prior development stage therapeutic product candidates for unmet medical needs.
Planned iGaming Casino Operations
On September 29, 2024, we entered into an Asset Purchase Agreement (the “Purchase Agreement”) with Elray Resources, Inc. (“Elray”). Pursuant to the Purchase Agreement, Elray agreed to sell us certain source code and intellectual property relating to an online blockchain casino (the “Purchased Assets”) in consideration for 1,000,000 shares of then newly designated Series B Convertible Preferred Stock (the “Preferred Stock”, and the shares of common stock issuable upon conversion thereof, the “Conversion Shares”) and warrants to purchase 3,000,000 shares of common stock of the Company (the “Purchase Warrants” and the shares of common stock issuable upon exercise thereof, the “Purchase Warrant Shares”). The acquisition of the Purchased Assets was completed on September 30, 2024.
The Purchase Agreement includes (i) customary covenants of each of the parties and confidentiality requirements; and (ii) customary indemnification requirements of the parties, subject to a $25,000 deductible.
2
The closing of the transactions contemplated by the Purchase Agreement was subject to certain customary conditions to closing, including the filing of the designation of the Preferred Stock with the Secretary of State of Delaware, and the receipt by the Company of an opinion of Hempstead & Co., LLC to the effect that, as of the date of such opinion and subject to the assumptions, qualifications, limitations and such other factors deemed relevant by Hempstead & Co., LLC, as set forth in such opinion, the purchase price to be paid by the Company was fair, from a financial point of view, to the Company, which opinion was received verbally on September 29, 2024, which conditions to closing were either satisfied or waived by the parties on September 30, 2024.
The acquisition contemplated by the Purchase Agreement closed on September 30, 2024 (the “Closing” and such date, the “Closing Date”).
Following the Closing, Elray agreed to provide support and assistance to the Company in connection with the building and launching of a fully operational casino operation utilizing the Purchased Assets, at no cost to the Company for a period of six months following the Closing, provided that such assistance shall not exceed 40 hours per week without the prior written approval of Elray (the “Post-Closing Assistance”). To date, Elray has provided code and certain supporting material to the Company. The Post-Closing Assistance will also require Elray to assist the Company with obtaining payment gateways and licensing where required, acknowledging that the Company will require a front end (the “Front-End Development”). Following the Closing, at the request and sole discretion of the Company, the Company may request Elray for an additional cost to help the Company complete the Front-End Development organically. The Company also has the option to request that Elray identify for the Company a vendor or vendors that would sell such a front end for one or more casinos that will operate on the Purchased Assets at a cost to be agreed between such vendor and the Company, in the Company’s sole discretion. As of the date of this filing, no such agreements have been entered into. The Company has sole discretion to determine which, if any, vendor it retains for the Front-End Development or in respect of a front-end acquisition.
The Purchase Agreement also restricts Elray, in perpetuity, from copying, selling, assigning, hypothecating, or otherwise transferring the Purchased Assets to any other party, without the prior written consent of the Company, and provides for the Company to be the sole owner of the Purchased Assets, except that Elray shall be authorized to retain and use the Purchased Assets for its own benefit and utilize such assets to provide SaaS solutions and hosted casino solutions to third party companies.
Pursuant to the Purchase Agreement, we agreed to file a proxy statement with the SEC (the “Proxy Statement”) to seek stockholder approval for the issuance of the Conversion Shares and Warrant Shares, under applicable rules of the Nasdaq Capital Market, as soon as reasonably practicable. We also agreed to use our reasonable best efforts to: (i) cause the Proxy Statement to be mailed to our stockholders as promptly as practicable following sign off from the SEC on such Proxy Statement, or no later than the 20th day after such preliminary Proxy Statement was filed with the SEC, in the event the SEC did not notify the Company of its intent to review such Proxy Statement, and (ii) ensure that the Proxy Statement complied in all material respects with the applicable provisions of the Securities Act and Exchange Act. We were also required to hold a shareholders meeting to seek shareholder approval for the issuance of the Conversion Shares and Warrant Shares promptly after the SEC confirmed that it has no comments on such Proxy Statement (the “Stockholder Approval”, and the date of such Stockholder Approval, the “Stockholder Approval Date”). The Stockholder Approval was received at our 2024 Annual Meeting of Stockholders held on December 27, 2024. As a result, the Series B Convertible Preferred Stock had a fixed conversion rate of 1.318 to 1 and was convertible into an aggregate of 1,318,000 total shares of common stock. On March 27, 2025, all 1,000,000 shares of the Series B Convertible Preferred Stock were converted into 1,318,000 shares of common stock at the option of the holder thereof.
We are planning to strategically enter into the online gaming industry, utilizing the Purchased Assets, i.e., our newly acquired “back-end” gaming platform, which incorporates blockchain technology and cryptocurrency operability (the “Gaming Technology Platform”). The Company plans to use this technology platform to establish a blockchain-based business aimed at the global iGaming market, focusing initially only on the cryptocurrency market and in the future, potentially the traditional fiat currency wagering market. Initially focusing on B2C (business-to-consumer) online casinos, the Company also plans to expand into a B2B (business-to-business) model, offering a seamless blockchain-enabled technology platform for gaming operators worldwide. In addition, management has identified certain global iGaming industry characteristics and trends that they believe make potential acquisition opportunities attractive. Management believes that the combination of the Gaming Technology Platform and the strength of a Nasdaq listing make the Company an attractive consolidation vehicle for the iGaming industry, and plans to work to identify potential acquisitions (although no targets exist at present).
3
Other than the Purchase Agreement and the consulting agreement with the technology consultant, discussed above, the Company has not entered into any material agreements in connection with the iGaming business to date.
A. The Gaming Technology Platform
The Gaming Technology Platform includes components designed to support online casinos using blockchain technology. These components are as follows:
| 1. | Blockchain Casino Operations Back-End: A robust infrastructure designed to support casino game programming through secure and scalable server/cloud solutions, database management, and blockchain payment processing. |
| 2. | Blockchain to Fiat Seamless Exchange Technology: A proprietary system allowing players to deposit and withdraw using cryptocurrency, while maintaining fiat-based wallets and gaming sessions, provided that each cryptocurrency and fiat currency will be kept separate and users will not be able to deposit in one crypto currency and withdraw in another, or in a fiat based currency, and vice versa. We believe that this cutting-edge technology offers a significant competitive advantage in the growing blockchain casino market. |
| 3. | Blockchain API Technology: A sophisticated application program interface (API) that facilitates reliable fiat-to-blockchain and blockchain-to-fiat payment gateways (provided that as discussed above, we do not plan to offer exchanges between cryptocurrencies and/or between fiat and cryptocurrency, and plan to initially only offer cryptocurrency wagering), supporting credit cards, e-wallets, and multiple cryptocurrencies while enabling blockchain-exclusive online casino operations. |
| 4. | Player Account Management: A secure system for managing player accounts, including registration, login, and player profiles, alongside know-your-client (KYC) and anti-money laundering (AML) compliance features. |
| 5. | Loyalty Systems for Blockchain Users: A customer relationship management system designed to foster player loyalty and enhance the lifetime value of each customer by catering to blockchain users. |
| 6. | Affiliate Tracking System: A tool to monitor traffic, registrations, and referral sources, allowing for scalable growth through affiliate partnerships and the distribution of referral fees and royalties. |
At the heart of the acquisition of the Gaming Technology Platform is proprietary source code, as well as specialized implementation know-how, much of which will be transferred from Elray to the Company, as detailed below. Management intends to further develop both the proprietary source code, enhancing and expanding the commercial potential of the Gaming Technology Platform, as well as continue to build expertise in the operation of the Gaming Technology Platform. Management believes that the existing Gaming Technology Platform, plus future potential enhancements, provide a strong competitive barrier, and position the Company well as the iGaming industry moves towards both greater cryptocurrency acceptance, and the safety of blockchain enabled platforms.
B. Industry Background
The global iGaming casino market has rapidly ascended as a major player in the digital entertainment industry, experiencing remarkable growth fueled by technological advancements, increased internet penetration, and evolving consumer preferences. According to a report by Statista, the global online gaming sector is estimated to reach $97 billion in 2024, and is projected to grow to nearly $133 billion by 2029. The cryptocurrency-based iGaming sector is growing even faster, albeit from a smaller base value, with SOFTSWISS estimating growth of over 20% between 2022 and 2023 for crypto-based bets, propelled by the increasing adoption of digital currencies like Bitcoin and Ethereum, which offer more secure, faster, and lower-cost transactions. According to XDA.io, the cryptocurrency component of the iGaming industry is estimated to have a size of approximately $40 billion, and is projected to increase to $158 billion by 2028.
4
Regionally, Europe leads the market, accounting for over 45% of the global iGaming market share in 2022, according to Global Market Insights, thanks to well-established regulatory frameworks in key countries such as the UK, Malta, and Sweden. In North America, research by Statista shows that the U.S. market has seen exponential growth following a 2018 U.S. Supreme Court case which struck down the federal ban on sports betting, which has led to an expansion in both online casino and sports betting offerings across multiple states. The U.S. iGaming market is projected to reach $39.8 billion by 2029, growing at a compounded annual growth rate (CAGR) of 9.8%, compared to projected revenue of $25 billion in 2024, according to Statista, driven primarily by sports betting and casino games. Canada is also emerging as a lucrative market due to favorable regulatory changes, contributing to North America’s strong growth.
Latin America has also become a rapidly expanding market, with countries such as Brazil, Mexico, and Colombia at the forefront. The region’s iGaming market is expected to grow at a CAGR of over 13% from 2022 to 2028, driven by increased mobile internet usage and the gradual liberalization of gambling laws. Meanwhile, Asia and Africa, although still relatively nascent in the iGaming sector, are expected to hold significant potential. The Asian market, led by countries such as India and Japan, could see a surge in online gaming revenues due to rising smartphone penetration and regulatory shifts. Statista notes that India alone is expected to reach $2.9 billion in online gambling revenue by 2024, growing at a 6% CAGR to just under $4 billion by 2029. Africa, with its expanding mobile infrastructure, is another emerging market, particularly in countries like South Africa and Nigeria.
The types of games contributing to this growth are diverse. Online casinos—comprising slots, table games, and live dealer games—dominate the iGaming market, contributing to over 37% of total market revenues in 2024 according to Statista. Sports betting is another key growth sector, with mobile sports betting estimated to account for around 70% of the sports betting market in many regions as early as 2020 according to Grand View Research. Management believes that this trend will continue, particularly in South America, Africa and parts of Asia as mobile penetration increases. Grand View Research also estimates that the global sports betting market will grow at a CAGR of 10-12% from 2023 to 2030, reaching $180 billion in revenue by 2030. Additionally, eSports betting is seeing rapid adoption, appealing particularly to younger demographics. According to Market Research Future, the eSports betting market is expected to grow from $9.9 billion in 2022 to over $30 billion by 2032. Management believes that much of this growth will come from the growing popularity of eSports, plus the continued proliferation of mobile devices, especially in Latin America, Asia and Africa.
Management believes that the future of the iGaming market is filled with opportunities. Cryptocurrency adoption in online casinos offers not only reduced transaction costs but also faster processing times and increased privacy, which we believe are highly appealing to modern gamers. The use of blockchain technology is also expected to bring about greater transparency and trust, with provably fair gaming systems becoming increasingly popular. Virtual reality (VR) and augmented reality (AR) are also expected to revolutionize the user experience, making online gambling more immersive. We also expect that the mobile gaming sector will continue to grow as mobile devices become more powerful and widely accessible, especially in emerging markets.
Management has conducted preliminary research, and identified a number of elements which require ongoing monitoring as the Company works to commercialize its first online casino. First, regulatory uncertainty remains one of the biggest challenges, as governments in different regions continue to establish or modify laws governing online gambling. Second, cybersecurity threats are also a growing concern, given the increasing sophistication of cyberattacks targeting online casinos and payment systems, which by the very nature of the underlying business, offer prime opportunity for security and personal information breaches. Third, there is heightened scrutiny around social responsibility, with a focus on gambling addiction and the need for responsible gaming measures, which could lead to tighter regulations in various jurisdictions. Management believes the Company’s Gaming Technology Platform, which management believes will allow for significant customer interaction, will help the Company address this risk. Competition in the market is fierce, with operators constantly investing in new technologies and marketing to attract and retain customers, further increasing operational costs, all of which will require the Company to continue to invest heavily in both the Gaming Technology Platform and other growth opportunities.
5
C. B2C Focus: Blockchain-Enabled Online Casinos
Our immediate focus is on launching B2C online casino operations in high-growth international markets. The Company’s blockchain technology enhances transparency and operational efficiency, providing players with a secure and trustworthy gaming experience, which management believes will be a key differentiator. Key features of this platform include:
| ● | Enhanced Transparency: Blockchain technology ensures secure, immutable transaction records, boosting player trust and confidence in the platform. |
| ● | Seamless Cryptocurrency Integration: Players can deposit and withdraw funds using cryptocurrency while maintaining fiat (traditional currency) wallets and gaming sessions, provided that each cryptocurrency and fiat currency will be kept separate and users will not be able to deposit in one crypto currency and withdraw in another, or in a fiat based currency, and vice versa, catering to the growing demand for crypto-friendly platforms. |
| ● | User Experience: The Gaming Technology Platform’s customer relationship management (CRM) system is designed to enhance player loyalty and increase the lifetime value of customers through personalized engagement and targeted promotions. |
Management is focused on three steps to operationalize the launch of the Company’s first online casino:
First, the Company is working to source a “front-end” customer interface, which is required for operation and commercialization of the Gaming Technology Platform, with plans to evaluate multiple, competitive potential suppliers of customer facing products and to acquire or create a “front-end”, funding permitting. The “front-end” is effectively the user interface (“UI”), where clients interact with the Company, accessing services provided by the Gaming Technology Platform. The specific UI type design, branding and functionality is dependent on whether the Company focuses on offering a B2B or B2C solution (or both).
The Board of Directors of the Company will make an informed decision as to whether the Company will utilize the Gaming Technology Platform acquired in a B2B or B2C format (or both). This decision will determine whether the Company will build its own “frontend” UI solution or acquire further assets that could potentially shorten time to market. No decision as to whether to pursue a specific B2B or B2C approach has yet been made, nor have any agreements been reached with any asset vendors, potential acquisition targets or partners. At present, the status of the Company’s product(s) remains under analysis and review.
That being said, the Company is currently in discussions with potential operators to provide them B2B turnkey solutions, and is also in discussion regarding the potential acquisition of additional assets that would assist the Company in the launch of a fully functional B2C operation. These conversations are early stage and exploratory, with no agreement in principle reached on any agreement. As noted above, the Company is currently focused on a B2C offering as a primary strategy, but continues to evaluate potential B2B opportunities as well.
Second, management is evaluating certain well known and high governance gaming jurisdictions to pursue initial licensing – no final determination has yet been made as to which jurisdiction(s) to pursue. An acquisition of a license could be through ab initio application, or through acquisition of an established company that has already been granted a license.
6
Third, management is beginning to evaluate a variety of games, which in the iGaming industry are generally sourced from third-party suppliers, to populate the planned front-end customer interface.
Currency-Agnostic Play and Gaming Variety
Management believes that a standout feature of the Gaming Technology Platform is its ability to offer currency-agnostic play, enabling customers to easily switch between traditional fiat currencies and cryptocurrencies during gaming sessions, provided that each cryptocurrency and fiat currency will be kept separate and users will not be able to deposit in one crypto currency and withdraw in another, or in a fiat based currency, and vice versa. Management believes this flexibility makes the customer interaction experience better, and therefore makes the Company’s ultimate gaming content accessible to a wider audience and supports a more seamless gaming experience for both traditional and crypto-savvy players. In addition, the Gaming Technology Platform is designed to support a wide variety of games, ranging from traditional offerings such as live casino games, including poker and blackjack, to newer rapid-play games such as crash games (where players bet on a multiplying curve. The longer a player stays active, the more they can win, and they are free to cash out at any time. If the game crashes before they have cashed out, they lose), as well as potentially in the future, live sports betting, all of which we believe appeal to the evolving preferences of today’s global players. We are confident that this blend of game styles will provide a comprehensive gaming experience, catering to the fast-changing tastes of both casual and high-frequency players across different markets.
High-Growth iGaming Jurisdictions
We expect to launch our planned B2C casino operations in established and high-growth markets where iGaming is gaining traction. Although the Company has not yet determined its first launch jurisdiction, it is currently analyzing high potential jurisdictions to select one or more launch jurisdictions.
Commercialization Strategy and Timeline
The commercialization strategy for launching the Company’s initial online casino is planned to follow a structured approach to ensure a smooth, compliant, and hopefully, profitable rollout. The first step involves the completion of the technology transfer, where all purchased assets, including software and intellectual property representing the Gaming Technology Platform, are securely transferred to the Company’s storage infrastructure. This step has been completed as of the date hereof. When fully operationalized, management anticipates that the Gaming Technology Platform will be hosted on a primary cloud-based server, with a physical server backup. This period is expected to include setting up the necessary server configurations, network security, and data which we anticipate completing by the end of Q2 2025. During this phase, management of the Company will be fully immersed in the technical aspects of the transfer process to ensure a seamless transition.
Concurrently with the technology transfer phase, management will be working to develop and enhance a comprehensive business strategy to guide the Company’s new focus on the iGaming sector. This strategy is planned to include selecting suitable web domains for one or more online casinos; creating relevant websites through the use of experienced casino “front-end” development teams; defining target customer markets and groups, including evaluation and selection of target customer demographics; and evaluation of content offerings, considering target markets and customer segments. Regulatory and compliance reviews are also an essential part of the comprehensive business strategy, as the iGaming business is highly regulated, with a trend to increasing regulatory scrutiny. This will be particularly important as the Company prepares its initial license application (see below) in one or more jurisdictions. Finally, the Company plans to lay the foundation for customer service solutions, which management believes are a critical component of the customer experience with iGaming.
7
Following the technology transfer phase, and running concurrently with the business strategy development phase, management intends to complete a full technology development review, ensuring that the Gaming Technology Platform fully aligns with business goals and meets the latest industry standards in terms of cybersecurity, information protection and reliability. Any potential gaps, such as development needs or cybersecurity updates, will be identified and addressed within this phase. Following the technology review, front-end development will then begin, which will focus on creating a user-friendly and functional gaming site. This phase may involve evaluating possible third-party solutions or self-developed solutions, giving careful consideration to costs and timing. While evaluating front-end solutions, management also intends to simultaneously evaluate potential gaming solutions. In the iGaming sector, games are generally sourced from third-party suppliers on a revenue share basis. Management will evaluate games based on selected target markets and client groups, as well as carefully considering cost and reliability of any potential game offerings.
Evaluation of licensing regimes will also be a critical component of the overall launch strategy, and management plans to focus on securing a reputable and cost-efficient license which will allow the casino to legally operate. Related to licensing, management will also focus on ensuring full compliance with all necessary Anti-Money Laundering (AML) and Know Your Customer (KYC) regulation and requirements, both from an industry perspective but also those unique to the chosen licensing jurisdictions.
The marketing plan development will be another significant milestone, focusing on identifying and attracting customers through affiliate marketing, direct marketing, and other channels. Critical metrics like Customer Lifetime Value (CLV) and Customer Acquisition Costs (CAC) are expected to be evaluated to ensure profitability for each potential market and customer group. The business strategy will also seek to explore how to build a loyal customer base and position the casino against competitors by using the Company’s Gaming Technology Platform, which has a built in customer loyalty functionality.
As the business begins to scale, assembling a growth team of senior management across marketing, technology, compliance, and other areas will be vital. This team will attempt to manage the casino’s expansion and oversee the daily operations. During this phase, establishing internal business processes for anti-money laundering, know your customer, accounting, taxation, and responsible gambling tracking will be key to ensuring long-term success and regulatory compliance. Business continuity, disaster recovery, and cybersecurity protocols are also expected to be implemented during this period.
Before launching, integration and testing will seek to ensure that all components of the casino—front-end, back-end, games, and marketing—work seamlessly together. This testing phase will be necessary to ensure everything is aligned for the official go-live, where the website will launch to the public. During the live testing period following the launch, the team will monitor performance, customer engagement, and feedback, making necessary adjustments to optimize the platform and ensure customer satisfaction.
Management believes that this structured approach, covering technology, business strategy, licensing, game selection, marketing, and internal processes, will provide a robust roadmap to operationalize the Gaming Technology Platform. Management is targeting having the first online casino fully operational and optimized for growth by the end of Q2 2025, although there can be no guarantee that all elements of the commercialization strategy will be completed on this timeline – lack of funding, slower than anticipated evaluation and contracting, and difficulty sourcing one or more key components could all contribute to delays.
Legal and Regulatory
As management evaluates the Company’s first jurisdiction for licensing, it will evaluate multiple options to seek to find the best combination of cost-effectiveness, market acceptability, and high reputation with transparency. The Company currently plans to assess jurisdictions like Curacao, known for its affordable licensing fees and relatively quick approval process, making it attractive for a swift market entry. However, more reputable and highly regulated jurisdictions such as Malta and the Isle of Man will also be considered, as they often offer greater transparency, strong consumer trust, and better access to European markets, though at a higher cost and stricter compliance requirements. Gibraltar could also be an option due to its favorable tax regime and robust regulatory framework.
Management plans to evaluate each jurisdiction based on licensing costs, annual fees, tax rates, regulatory ease, and the ability to serve key target markets. Transparency and oversight will be critical, as the Company seeks to build credibility in the highly competitive iGaming sector. Ultimately, the jurisdiction chosen will not only be picked to offer the best balance of cost and reputation, but to also align most closely with the Company’s target launch markets, ensuring that the license supports our efforts for a successful and compliant entry into those regions.
8
1. Cryptocurrency capability – The Gaming Technology Platform recently acquired by the Company has been designed to offer cryptocurrency capability supported by blockchain technology. Blockchain technology is a database management mechanism that allows transparent information sharing within a business network and is the technology that enables the existence of cryptocurrency. The blockchain component software exists to enhance security and transparency. The Gaming Technology Platform has been designed to allow wagering in cryptocurrency (which is what we plan to initially offer) as well as traditional fiat currencies. Management believes this to be a key focus area for gaming participants, one where potential customers have increasing interest and engagement;
2. Player management and loyalty – As noted above, maintaining customer loyalty is critical in a very competitive market. The Company believes that its technology will allow for significant player engagement, potentially increasing customer value versus customer acquisition cost;
3. Unique content – The Company is currently in early conversations with multiple unique content production and distribution companies in the iGaming sector. If agreements are reached with one or more of such entities (including but not limited to joint-ventures, acquisitions, mergers and various partnerships), the Company believes that this will provide a significant competitive advantage if the Company were to ultimately launch a B2C focused business. The Company hopes to be able to leverage the Gaming Technology Platform to provide potential clients with highly unique, iGaming related content, thus reducing customer acquisition cost and increasing customer lifetime value; and
4. M&A and industry expertise – Management of the Company believes that the management and shareholders of the Company provide a unique blend of capital markets expertise related to negotiating, structuring and executing merger and acquisition (M&A) transactions, plus unique industry knowledge through the involvement of experienced industry executives. Management believes this provides the Company with access to diverse and unique relationships, leading to potential acquisition and partnership opportunities. When combined with M&A expertise, management believes that the Company has a distinct competitive advantage relative to certain peers.
In terms of opportunity, cryptocurrency iGaming casinos have gained significant traction globally, which management believes presents a major opportunity for the Company given the cryptocurrency focus of the Gaming Technology Platform.
Projected Costs
Management believes that the estimated costs to commercialize an online iGaming casino can vary significantly, depending on the jurisdiction and the scale of the operation, but some key expenses are universal (and somewhat fixed), while others are variable and depend on decisions made by management around business strategy. Costs that management expects to incur, given that the Company has already acquired the Gaming Technology Platform, are expected to include the following: first, sourcing a front-end player interface involves either purchasing or developing a customized platform. Off-the-shelf solutions from iGaming software developers typically start at around $100,000, but can increase dramatically for bespoke or highly tailored platforms. Second, partnerships with game providers require initial setup fees, licensing, and revenue-sharing agreements. These costs may range from $10,000 to over $100,000 per game, depending on the scope of the partnership and the range of games offered. Third, management believes that ensuring a smooth user experience, quality design, and strong cybersecurity are essential, which can also add to the initial outlay, with estimated costs of around $500,000 to acquire a sufficient number of games for our planned platform.
9
Based on research undertaken by management to date, an online casino requires a robust marketing budget, often requiring at least $1-2 million in the first year to build brand recognition through digital advertising, influencer partnerships, affiliate programs, and promotions. Establishing legal and regulatory compliance frameworks adds another layer of cost. In regulated markets, obtaining licenses from gaming authorities can range from $50,000 to over $200,000 per jurisdiction, depending on the region, with ongoing costs for renewal. Legal and regulatory consultants, particularly in complex markets, will require ongoing fees, which could be $20,000 to $100,000 annually. Hiring experienced senior staff like a Chief Compliance Officer, legal experts, and marketing heads will further add to the budget. Salaries for top-tier executives can range from $250,000 to $300,000 annually, making human resources one of the largest ongoing expenses. Regulatory assistance to navigate licensing audits and compliance can add up to another $100,000 annually. Together, these elements make the initial commercialization of an online iGaming casino a significant but potentially lucrative investment.
In total, management believes that the initial cost to fully commercialize the Gaming Technology Platform, with a license in one key jurisdiction, ten games and an adequate marketing and administrative budget, ranges from $3 million to more than $5 million.
D. B2B Focus: Blockchain-Enabled Technology Platform
Management is also evaluating the potential future ability to expand into the global online gaming B2B (business-to-business) market by creating and launching a blockchain-enabled platform aimed at providing gaming operators with a cutting-edge, transparent, and secure solution for running their operations. This move would be a potential strategic extension of our newly acquired Gaming Technology Platform, leveraging the benefits of our newly acquired blockchain technology to offer innovative tools that enhance operational efficiency, player trust, and business growth for operators worldwide.
The Gaming Technology Platform, subject to build-out, as discussed above, has the capability to enable B2B customers to launch online casinos with a full suite of iGaming content including sportsbooks, online slots, table games and live casino games. The same is true in the event that the Company pursues a direct B2C product offering, which is the current focus of the Company’s strategy.
Blockchain Technology: Enhancing Trust and Security
At the core of our potential B2B offering is our blockchain technology, which we believe provides advantages for gaming operators over those offerings of our competitors which do not include blockchain technology. Blockchain’s decentralized and immutable nature ensures transparent and secure transaction records, allowing operators to build stronger trust with their players. Every gaming transaction, from deposits and withdrawals to bets and payouts, is recorded on the blockchain, creating an auditable trail that enhances accountability and reduces the risks of fraud or manipulation. Management believes that players are currently demanding increased accountability from gaming operators and have an enhanced focus on potential fraud in the online gaming market globally.
This increased transparency also simplifies regulatory compliance, as operators can provide clear, verifiable records to regulators in real-time. By offering a platform that integrates blockchain-enabled gaming infrastructure, we expect to help allow operators to position themselves as trustworthy and compliant, which is essential in the highly regulated gaming industry. The regulation of online gaming, cryptocurrencies and online transactions generally is converging across jurisdictions, and management believes that our emerging technology has a strong potential to address these key challenges for operators.
Seamless Cryptocurrency and Fiat Currency Support
One of the key features of our Gaming Technology Platform is our currency-agnostic framework, which supports both cryptocurrency and traditional fiat currencies. This flexibility provides operators with the ability to cater to a diverse global audience. As cryptocurrencies like bitcoin and Ethereum continue to gain popularity, especially among younger and tech-savvy players, our platform enables operators to attract this growing demographic by offering seamless crypto-based transactions. According to SOFTSWISS, cryptocurrency based wagering accounted for 27.5% of bets placed in the iGaming industry in 2023, with substantial differences in adoption between various markets.
10
At the same time, our Gaming Technology Platform also supports fiat currency transactions to cater to traditional players who prefer to use government-issued currencies. This dual currency model is expected to enable operators to broaden their player base while offering an enhanced, user-friendly experience that adapts to the evolving preferences of players globally. We believe that the ability to use both crypto and fiat wallets within a single platform would give operators a competitive edge in offering flexible financial options to their users.
Cryptocurrency Transactions and Holdings
Notwithstanding the above, the Gaming Technology Platform’s cryptocurrency and fiat currency holdings will be kept separate and users will not be able to deposit in one crypto currency and withdraw in another, or in a fiat based currency, and vice versa. Additionally, we anticipate our Gaming Technology Platform will initially only allow wagering in cryptocurrency and we plan to focus on bitcoin, Ethereum and Litecoin, which together represent approximately 95% of the popular cryptocurrencies in the iGaming industry, according to an article published in October 2024 at blockonomi.com. In the future, we may also allow wagering in fiat, provided that as discussed above, we do not plan to offer the ability to withdraw a different type of cryptocurrency or currency than deposited or to exchange between cryptocurrencies or currencies. Wagers and payouts will be made only with the same cryptocurrency. For example, if a user places a wager in Ethereum, they can only win additional Ethereum and will only be able to withdraw Ethereum upon the settlement of any wager.
Any cryptocurrencies we hold will be valued from time to time, including as of the end of each calendar quarter, based on then market prices. Users are expected to be able to withdraw deposits and/or winnings at any time, only the type of currency (initially only cryptocurrency) in which they deposited funds.
We plan to hold a sufficient amount of bitcoin, Ethereum and Litecoin to cover wagers in process and expected payouts relating thereto. We plan to purchase, sell and exchange bitcoin, Ethereum and Litecoin from time to time in order to cover wagers and convert net income to cash for operations. All such purchases, sales and exchanges, will be at then market prices, and will be subject to fluctuation with changing market prices.
As of December 31, 2024, we held, and as of the date of this Report, we hold, no cryptocurrencies.
Consumer Relationship Management
The Company’s Gaming Technology Platform also includes sophisticated tools such as a built-in CRM system, which would enable operators to optimize player engagement and retention. By providing data-driven insights and personalized interactions, operators are expected to be able to increase the lifetime value of their players, fostering long-term growth and loyalty.
While management is focused on developing the B2C business as an initial priority, the potential ability to use the Company’s Gaming Technology Platform to expand to B2B sources of revenue in the future is of strong interest, and we believe it has the potential to provide a valuable source of differentiated revenue for the Company down the road.
E. Go-Forward Consolidation Strategy
Finally, management believes that we are uniquely positioned to act as a consolidator in the gaming industry, capitalizing on our Nasdaq listing, highly experienced management team and Board, and robust Gaming Technology Platform. As a publicly-traded company, our Nasdaq listing provides us with access to capital markets and a well-recognized stock currency, which we anticipate will allow us to efficiently fund potential future acquisitions. We hope that this liquidity advantage will allow us to acquire private gaming companies (both B2C and B2B) at attractive valuations, especially in a market where private firms may lack similar access to capital. Management believes that the global iGaming industry is entering a period of rapid consolidation, and that potentially attractive acquisition opportunities in key jurisdictions could become available in the future. At present, management has identified several potential targets, but is still conducting preliminary diligence and has no immediate acquisition plans.
11
We think that management’s experience across multiple industries, combined with a strategic vision for iGaming, enhances our ability to identify and integrate undervalued gaming companies. We plan to work to ensure that future acquisitions are not only executed effectively but also yield synergistic benefits that contribute to accretive growth. By incorporating these companies into our Gaming Technology Platform, either as technology components or users of the technology, we hope to be able to optimize operational efficiencies, expand our geographic and market reach, and enhance our technological offerings, with our goal of positioning the Company as a leader in the evolving iGaming sector.
F. Distribution Methods
Distribution methods depend on whether the chosen commercialization path is via a B2B or B2C offering (or both). The Company has built, and continues to build, significant relationships with a variety of iGaming industry participants, including content producers and providers specific to the industry, content distribution sources, licensed casino operators, technology and marketing providers. Through this network, the Company has already identified a number of potentially significant distribution opportunities for both B2B and B2C product offerings by the Company. In terms of B2B, the Company has already identified potential clients that would like to launch an online blockchain casino (under their own branding) using the Gaming Technology Platform owned by the Company. These leads have been generated by strategic relationships, leading to referrals in the industry. The Company is also in the process of attending a variety of industry trade shows, and meeting with a variety of strategic industry participants, and expects to generate further potential B2B clients as a result of these activities. In terms of B2C distribution channels, should the Company launch its own online casino, the methods to build revenues are fairly standard in the industry. This generally involves acquisition of players via social media, influencers, online advertising such as Facebook & Google and also via affiliate driven traffic (third party companies that specialize in delivering players for commercial benefits). In addition to having identified several groups of assets which could potentially be used by the Company to launch an online casino, the Company is in early conversations with a variety of iGaming related content creators and distributors in addition to affiliate marketing organizations. These conversations are early stage and exploratory – no agreement in principle has been reached with any entity.
G. Projected Costs
Management believes that the estimated costs to commercialize an online iGaming casino can vary significantly, depending on the jurisdiction and the scale of the operation, but some key expenses are universal (and somewhat fixed), while others are variable and depend on decisions made by management around business strategy. Costs that management expects to incur, given that the Company has already acquired the Gaming Technology Platform, are expected to include the following: first, sourcing a front-end player interface involves either purchasing or developing a customized platform. Off-the-shelf solutions from iGaming software developers typically start at around $100,000, but can increase dramatically for bespoke or highly tailored platforms. Second, partnerships with game providers require initial setup fees, licensing, and revenue-sharing agreements. These costs may range from $10,000 to over $100,000 per game, depending on the scope of the partnership and the range of games offered. Third, management believes that ensuring a smooth user experience, quality design, and strong cybersecurity are essential, which can also add to the initial outlay, with estimated costs of around $500,000 to acquire a sufficient number of games for our planned platform.
Based on research undertaken by management to date, an online casino requires a robust marketing budget, often requiring at least $1-2 million in the first year to build brand recognition through digital advertising, influencer partnerships, affiliate programs, and promotions. Establishing legal and regulatory compliance frameworks adds another layer of cost. In regulated markets, obtaining licenses from gaming authorities can range from $50,000 to over $200,000 per jurisdiction, depending on the region, with ongoing costs for renewal. Legal and regulatory consultants, particularly in complex markets, will require ongoing fees, which could be $20,000 to $100,000 annually. Hiring experienced senior staff like a Chief Compliance Officer, legal experts, and marketing heads will further add to the budget. Salaries for top-tier executives can range from $250,000 to $300,000 annually, making human resources one of the largest ongoing expenses. Regulatory assistance to navigate licensing audits and compliance can add up to another $100,000 annually. Together, these elements make the initial commercialization of an online iGaming casino a significant but potentially lucrative investment.
12
In total, management believes that the initial cost to fully commercialize the Gaming Technology Platform, with a license in one key jurisdiction, ten games and an adequate marketing and administrative budget, ranges from $3 million to more than $5 million.
Legacy Biotechnology Programs
We currently have two legacy biotechnology programs that are focused on different diseases or medical conditions, and that target different factors, molecules or proteins. In November 2024, the biotechnology related to alpha 7 nicotinic acetylcholine receptor was returned to Stanford University.
Due to restrictions in the Company’s resources, the Company has slowed down research and development activities significantly in the SCA platform (discussed below) and the anti-TNF platform (discussed below), and the Company as discussed below, the Company’s license agreement relating to the α7nAChR platform (discussed below) was terminated in November 2024.
The Company is currently evaluating all options to monetize its existing life science assets, in addition to exploring other strategic alternatives to maximize value for its stockholders. Our legacy biotechnology programs include:
| ● | fibrosis and anti-tumor necrosis factor (“TNF”); and |
| ● | drugs which are derivatives of cannabidiol (“CBD”) or cannabigerol (“CBG”) analogues (“SCAs”). |
The license agreement with Stanford University covering α7nAChR was terminated effective November 23, 2024. After a careful review of the Company’s intellectual property portfolio as part of the Company’s ongoing strategic review process, the Company decided to move in a different direction and returned the intellectual property to Stanford University.
Fibrosis & Anti-TNF Platform
Our anti-tumor necrosis factor (TNF) platform began at our wholly-owned subsidiary, 180 Therapeutics L.P. (“180 LP”). This platform is focused on studying the molecular mechanism of inflammatory diseases and fibrosis and on the discovery of TNF as a mediator of fibrosis, as well as other immune-driven diseases. This research was first undertaken in the 1980s by our Executive Co-Chairman, Prof. Sir Marc Feldmann, based on analysis of tissue from patients with rheumatoid arthritis (“RA”). We are applying this same approach to the analysis of human disease tissue from patients with active fibrosis, research led by Prof. Jagdeep Nanchahal in Oxford (who is also the Chairman of our Clinical Advisory Board), which has led to the identification of new therapeutic targets and approaches that we are developing. Profs. Nanchahal and Feldmann, in collaboration with other scientists, are leveraging their experience and expertise in developing anti-inflammatories to search for new applications for anti-TNF therapeutics. We are seeking to demonstrate that anti-TNF drugs, such as adalimumab, have a positive effect on new indications such as Dupuytren’s Contracture, frozen shoulder and POCD.
Our first product candidate in clinical development is for the potential treatment of early-stage fibrosis of the hand, Dupuytren’s Contracture, for which there is currently no approved treatment in the U.K. or EU. Collagenase from Clostridium histolyticum has been approved in the USA for late-stage Dupuytren’s Contracture. The proposed treatment will be administered by a local injection of adalimumab, an anti-TNF antibody, into early-stage disease tissue. The results for the Phase 2a clinical trial for Dupuytren’s Contracture, supported by the Wellcome Trust, U.K. Department of Health and us, were published in July 2018. The study demonstrated positive tissue response indicative of anti-fibrotic mechanisms, as well as guiding dosing for follow up trials. Having defined the most efficacious dose and preparation and based on these positive proof of concept data, we, together with the Wellcome Trust and the U.K. Department of Health, initiated a Phase 2b trial in patients with early-stage Dupuytren’s Contracture. The initial plan was to randomize 138 patients in a ratio of 1:1 to receive four injections of adalimumab or placebo at three-month intervals and followed for a total of 18 months from baseline. With additional funding from the Wellcome Trust, the Phase 2b trial completed recruitment of 174 patients in April 2019, having commenced dosing in February 2017. The final patient was enrolled in April 2019. The Phase 2b clinical trial for early-stage Dupuytren’s Contracture has been completed. On December 1, 2021, we announced top line data from the trial, which indicates that the primary end point of nodule hardness and the secondary end point of nodule size on ultrasound scan were met with statistically significant differences. There were no related severe adverse events. As previously reported, the full results of the trial have been published in a peer-reviewed journal. Through this fibrosis and anti-TNF product development platform, we are also performing research for the development of potential treatments of frozen shoulder, liver and lung fibrosis and post-operative cognitive dysfunction/delirium (“POCD”).
13
We have previously obtained regulatory approvals from the U.K. MHRA and the CCMO, as well as from the relevant accredited ethics committees, in order to perform clinical trials in the U.K. and The Netherlands solely for indications under the anti-TNF platform. No marketing applications or requests for marketing approval have been submitted to, the U.S. Food and Drug Administration (“FDA”) for any indications or products under the anti-TNF platform at this time. On March 29, 2022, we submitted a request to the FDA for a Type C Meeting to discuss clinical outcome assessment in clinical trials of the anti-TNF platform for early stage Dupuytren’s disease. On April 26, 2022, the FDA granted the meeting request and agreed to provide written responses in lieu of a meeting. On June 9, 2022, the FDA provided the aforementioned written responses in which the agency questioned whether nodule hardness and size would constitute an appropriate end point in such studies. Specifically, the FDA stated, “The proposed outcome measures of nodule hardness and nodule size do not appear to be clinical outcome measures that measure how a patient feels, functions, or survives, which would be needed to support a demonstration of efficacy in your future registrational studies.”
On February 22, 2023, we announced the closure of recruitment of patients for the feasibility trial for frozen shoulder, for which we have ended such recruitment at nine patients, due to a regulatory request in the U.K. to end slow recruiting trials. The result of the closure of the trial means that another trial will likely need to be undertaken in the future to recruit additional participants. We do not have current plans to conduct another trial and we are seeking a potential sale of such TNF Platform.
In order for the Company, or any acquiror of the Company’s TNF platform, to be able to commercialize the TNF platform in the United States (and likely other jurisdictions), we believe that conducting the follow-on Phase 3 trial of a design that is yet to be determined will likewise be necessary for FDA acceptance and that the Company or an acquiror of the TNF program will have to engage in a further regulatory process with the FDA and/or other regulators internationally. The Company is not pursuing Phase 3 trials or any regulatory dialogue in respect of the TNF program currently.
SCAs Platform
Our SCAs platform began at our wholly-owned subsidiary, CannBioRex Pharmaceuticals Corp. (“CBR Pharma”) with the collaborative work of its founders Prof. Mechoulam, deceased, and Prof. Feldmann. This platform focuses on the development of synthetic pharmaceutical grade molecules close or distant analogs of non-psychoactive cannabinoids such as CBD for the treatment of inflammatory diseases and pain. These development efforts are a result of a 20-year collaboration between Prof. Feldmann, who discovered and commercialized anti-TNF therapy for treatment of RA and subsequently a number of inflammatory diseases, which is currently the best-selling drug class in the world, and Prof. Mechoulam, who was a world leading expert in cannabis chemistry who successfully identified tetrahydrocannabinol (THC) is the primary psychoactive compound found in the cannabis plant, cannabidiol (CBD) a non-psychoactive compound found in the cannabis plant, and, subsequently, the endocannabinoids. We are working with a research team based at the Kennedy Institute at Oxford, consisting of Prof. Feldmann, Prof. Richard Williams and others, and a research team based at Hebrew University, consisting of Prof. Avi Domb, Prof. Amnon Hoffman and others, to generate new drugs, test them, and optimize their uptake and delivery to disease targets. The aim is to develop novel, orally active analgesic and anti-inflammatory medications based on synthetic compounds to target chronic diseases. We term these synthetic compounds generically as “synthetic CBD analogs” (“SCAs”). Our primary development targets are arthritis and chronic and recurrent pain, while our secondary development targets are diabetes/diabetic neuropathy, fibromyalgia, multiple sclerosis, obesity and fatty liver disease. Unfortunately, Dr. Mechoulam passed away in March 2023, and while he will be sorely missed, work with his colleagues will continue as necessary.
14
The Company conducted a research study involving the SCA platform during early 2024 and on July 30, 2024 announced topline results from a clinical pharmacology study (the “Study”), that evaluated the uptake of cannabidiol (CBD) in a solid formulation which can be delivered as a pill orally. The clinical trial in humans, performed with Prof. Avi Domb of the Hebrew University, School of Pharmacy, and with Prof. Elyad Davidson, of Hadassah Hospital, compared two solid formulations of CBD with a U.S. Food and Drug Administration (FDA) approved drug for epilepsy, Epidiolex. The purpose of the Study was to compare the pharmokinetic (PK) profile of a generic approved CBD product, Epidiolex®, with two solid formulations. This type of trial has yet to be examined in a clinical setting. For the Company’s trial, twelve volunteers received all three formulations in a crossover randomized trial. Results of the clinical trial indicate that one of the two solid forms was absorbed faster and exhibited higher maximal levels compared to Epidiolex. Both of the solid formulations were well tolerated.
CBD is a purified product that is not psychoactive, which we believe has potential benefits for treatment of inflammatory processes and pain. A major problem in working with CBD is its low, unpredictable and variable uptake following the most convenient delivery form, by mouth, as a liquid CBD in oil like the approved pharmaceutical Epidiolex, given for epilepsy.
The results of the clinical trial performed on 12 adult males at the Hadassah Hospital, shows that capsules composed of one of the PNL formulations performed better statistically than Epidiolex in terms of the speed of absorption and achievement of maximal levels. The other PNL formulation, also given as a capsule, was statistically equivalent to Epidiolex in terms of speed of absorption and achievement of maximal levels.
We believe that a solid formulation for testing in clinical trials will advance testing CBD in other indications. CBD has shown promise in studies on treatment of pain, post-traumatic stress disorder (PTSD), head trauma, and other indications, yet an oral liquid formulation is undesirable. The new formulations tested by the Company and its collaborators open a potential path for testing solid CBD given by mouth in a capsule.
Full study results are not yet available. The trial results are planned to be submitted for scientific publication at a later date, which has not yet been determined. The Company is currently evaluating the SCA Program, including exploring the potential for disposition of this program pursuant to our previously announced strategic review process.
No regulatory approvals have been sought or obtained from appropriate authorities at this time for any products or indications under the SCAs platform.
15
SCAs Platform
Overview
Cannabinoids are a class of compounds derived from cannabis plants. The two major cannabinoids contained in cannabis are CBD and THC. Although one cannabinoid, THC, is known to cause psychoactive effects associated with the use of herbal cannabis, no other cannabinoid is known to share these properties. In recent decades, there have been major scientific advances that have led to the discovery of new plant-derived cannabinoids and the endocannabinoid system. There are at least two types of cannabinoid receptors in the human endocannabinoid system, cannabinoid receptor 1 (“CB1”) and cannabinoid receptor 2 (“CB2”). CB1 receptors are considered to be among the most widely expressed G protein-coupled receptors in the brain and are particularly abundant in areas of the brain concerned with movement and postural control, pain and sensory perception, memory, cognition, emotion, and autonomic and endocrine function. CB1 receptors are also found in peripheral tissues including peripheral nerves and non-neuronal tissues such as muscle, liver tissues and fat. CB2 receptors are expressed primarily in tissues in the immune system and are believed to mediate the immunological effects of cannabinoids. CBD does not interact with CB1 receptors and is only a weak agonist of CB2 receptors. CBD interacts with other important neurotransmitter and neuromodulatory systems in the human body, including transient receptor potential channels, adenosine uptake and serotonin receptors. The far-reaching and diverse pharmacology of the numerous cannabinoids provides significant potential for development of cannabinoid therapeutics across many indications and disease areas, but also adds to the complexity of the research.
For the SCA program, we have agreements in place with Hebrew University and Oxford, pursuant to which we intend to conduct research to develop and characterize novel SCAs for the treatment of certain target indications, and to perform early-phase clinical trials. Through the Research Agreements with Hebrew University and Oxford, we established research facilities at the Hebrew University and Oxford, in which the development and testing of new cannabinoids designed and synthesized at the Hebrew University will be facilitated. The labs at the Hebrew University will synthesize the chemical compounds and perform preliminary efficacy and safety studies.
Once these initial studies are completed at the Hebrew University, the chemical compounds are sent to Prof. Richard Williams at Oxford, where further evaluation is carried out to identify candidates which have the best potential for clinical efficacy and commercial development. Subsequently, we will support the clinical development of the lead compound(s), culminating in Phase 2 clinical trials to establish clinical utility in chronic pain and inflammatory indications.
The focus of the research is the development of safe and well-tolerated compounds with analgesic and immunomodulatory activity and with the capacity to synergize with current therapies, which target downstream inflammatory processes. After conducting initial research and development, we selected the most promising of the chemical derivatives to move into Phase 1/2 clinical trials, pending successful toxicity studies. In addition, we have identified two lead solid dosage oral formulations of CBD from animal studies, and preparations are underway to facilitate pharmacokinetic analysis in healthy human volunteers.
Product Candidates or Indications
We believe that there are unmet needs for orally available, relatively safe anti-inflammatory drugs, especially those with analgesic properties. We believe that SCAs have the potential to fulfill these needs and we have started to develop novel, orally available and patentable drug candidates to treat certain diseases or conditions such as arthritis, multiple sclerosis, diabetes, psoriasis, obesity and fatty liver, and various painful conditions. Our work on SCAs is currently in the preclinical development stage.
Because medical cannabis is a complex mixture of over 200 compounds from plants, providing a consistent level of the active compound of interest or controlling the level of the other natural compounds is difficult. Accordingly, we are working on orally available SCAs, not derived from plants, to address the deleterious issues of medical cannabis described above. If successful, these SCAs could become approved drug products that offer a robustly consistent and safe dosage that allows patient intake to be carefully controlled.
16
We believe that the development and clinical study of SCAs will reveal that SCAs have several key advantages over medical cannabis, including:
| ● | use of a pure compound (>99.5%) rather than a mixture of compounds; |
| ● | ability to test and control dosing, which in turn controls efficacy and side effect levels; |
| ● | creation of a reproducible product; and |
| ● | ability to engineer novel synthetic analogs to control binding preferences to select receptors, control agonist or antagonist effects of receptor binding (pharmacokinetics and dynamics), modify half-life of the drug in the body, and create pro-drug forms that are only activated in specified tissues, thereby potentially reducing off target side effects. |
In addition to the above advantages, testing SCAs in scientific, double-blind clinical trials would help to allay physicians’ concerns regarding the therapeutic use of marijuana-based compounds. This change could increase the number of patients that have access to these drug therapies. If clinical trials are successful, there are a number of potential markets and indications for SCAs which we could target, which include individuals suffering from chronic and recurrent pain, diabetes, osteoarthritis, obesity and fatty liver disease.
α7nAChR Platform
The license agreement with Stanford University covering α7nAChR was terminated effective November 23, 2024. After a careful review of the Company’s intellectual property portfolio as part of the Company’s ongoing strategic review process, the Company decided to move in a different direction and returned the intellectual property to Stanford University.
Outsourcing and Manufacturing
We are not currently conducting any clinical trials and do not have plans to conduct clinical trials in the future.
Material Agreements
We have entered into material research and licensing agreements (the “Research Agreements”) with various universities and parties in order to conduct research to develop potential product candidates. We have also entered into other material consulting and advisory services agreements with various scientists (the “Consulting Agreements”) to assist with such research.
Overview of Research Agreements
The Research Agreements include agreements with the Hebrew University and Oxford. For the anti-TNF platform, the Research Agreements with Oxford allow us to contribute financially to sponsor the research being conducted for the anti-TNF platform. In return, we will receive an exclusive option to license any intellectual property arising from the Research Agreements. There are also license agreements in place whereby we have exclusively licensed certain intellectual property from Oxford.
For the SCA program, we have agreements in place with Hebrew University and Oxford, pursuant to which we conducted research to develop and characterize novel SCAs for the treatment of certain target indications, and to perform early-phase clinical trials. Through the Research Agreements with Hebrew University and Oxford, we established research facilities at the Hebrew University and Oxford, in which the development and testing of new cannabinoids designed and synthesized at the Hebrew University will be facilitated.
17
The Research Agreements are each described below.
Research Agreements with the Hebrew University
On May 13, 2018, our wholly-owned subsidiary CBR Pharma entered into a research and license agreement (the “2018 Hebrew Agreement”) with Yissum Research Development Company of the Hebrew University of Jerusalem, Ltd. (“Yissum”), pursuant to which Yissum granted CBR Pharma a worldwide exclusive license (the “2018 Hebrew License”) to develop and commercialize certain patents (the “2018 Hebrew Licensed Patents”), know-how and research results (collectively, the “2018 Hebrew Licensed Technology”), in order to develop, manufacture, market, distribute or sell products, all within the use of the 2018 Hebrew Licensed Technology for the treatment of any and all veterinary and human medical conditions, including obesity, pain, inflammation and arthritis (the “2018 Field”).
Pursuant to the 2018 Hebrew Agreement, notwithstanding the grant of the 2018 Hebrew License, Yissum, on behalf of Hebrew University, will retain the right to (i) make, use and practice the 2018 Hebrew Licensed Technology for Hebrew University’s own research and educational purposes; (ii) license or otherwise convey to other academic and not-for-profit research organizations the 2018 Hebrew Licensed Technology for use in non-commercial research; and (iii) license or otherwise convey the 2018 Hebrew Licensed Technology to any third party for research or commercial applications outside the 2018 Field.
The 2018 Hebrew Agreement further provides that CBR Pharma is entitled to grant one or more sublicenses to the 2018 Hebrew Licensed Technology for exploitation in the 2018 Field.
All right, title and interest in and to the 2018 Hebrew Licensed Technology vest solely in Yissum, and CBR Pharma will hold and make use of the rights granted pursuant to the 2018 Hebrew License solely in accordance with the terms of the 2018 Hebrew Agreement.
As consideration for the 2018 Hebrew License, CBR Pharma paid Yissum a license fee of $75,000 and agreed to continue to pay an annual license maintenance fee (the “License Maintenance Fee”) of $50,000, beginning on May 1, 2019 and thereafter on the first day of May each year. The License Maintenance Fee is non-refundable but may be credited each year against royalties on account of net sales of products made from May 1 to April 30 of each year.
Yissum has also agreed to undertake research and to synthesize chemical compounds that will be used by CBR Pharma, through additional research at both Oxford and Hebrew University, to develop orally active analgesic and anti-inflammatory medications. Compounds will be shipped from Hebrew University to Oxford for use in pre-clinical studies to establish efficacy in pain and inflammation.
Upon the achievement of certain milestones in respect of the chemical compounds derived from the 2018 Hebrew Licensed Technology, CBR Pharma is obligated to make certain payments to Yissum, including but not limited to the following:
| Milestone | Milestone Fee | |||
| Submission of the first IND testing for the FDA | $ | 75,000 | ||
| Commencement of one Phase 1/2 trial with the FDA | $ | 100,000 | ||
| Commencement of one Phase 3 trial with the FDA | $ | 150,000 | ||
| For each product market authorization/clearance (maximum of $500,000) | $ | 100,000 | ||
| (maximum of $500,000 | ) | |||
| For every $250 million in accumulated sales of the product until $1 billion in sales is achieved | $ | 250,000 | ||
CBR Pharma will pay Yissum royalties equal to (i) 3% of the net sales for the first annual $500 million of net sales, and (ii) 5% of the net sales after the net sales are at or in excess of $500 million.
18
In the event of a sale by CBR Pharma stockholders of their common shares or the transfer or assignment of the 2018 Hebrew Agreement, CBR Pharma is obligated to pay Yissum a fee of 5% of the consideration received by CBR Pharma pursuant to such corporate transaction. In the event of an initial public offering, or a go-public event, CBR Pharma was obligated to issue registered common shares to Yissum equal to 5% of the issued and outstanding common shares, on a fully-diluted basis, concurrently with the closing of such transaction. The Business Combination that was consummated on November 6, 2020, was considered a go-public event, pursuant to which we issued 12,028 of our common shares to Yissum prior to the closing of the Business Combination.
CBR Pharma has also agreed to reimburse Yissum (to a maximum of $30,000) for costs incurred for patent expenses.
Yissum and CBR Pharma also agreed to establish a research program for which CBR Pharma funded a $400,000 budget for the 12-month period ended May 2019. The Company plans to move forward with the research using a different third party, of which no agreement has been finalized.
The 2018 Hebrew Agreement will terminate upon the occurrence of the later of the following: (i) the expiration of the last of the 2018 Hebrew Licensed Patents; (ii) the expiration of the last exclusivity on any product granted by any regulatory or government body; (iii) the expiration of a continuous period of twenty years during which there was no commercial sale of any product in any country; or (iv) if we elect to obtain an exclusive license to the know-how under the terms of the 2018 Hebrew Agreement, the expiration of such exclusive license.
On November 11, 2019, CBR Pharma entered into an additional research and license agreement (the “2019 Hebrew Agreement”) with Yissum, pursuant to which Yissum granted CBR Pharma a worldwide sole and exclusive license (the “2019 Hebrew License”) to develop and commercialize certain patents (the “2019 Hebrew Licensed Patents”), know-how and research results (collectively, the “2019 Hebrew Licensed Technology,” and together with the 2018 Hebrew Licensed Technology, the “Hebrew Licensed Technology”), in order to develop, manufacture, market, distribute, sell, repair and refurbish products, all within the use of the 2019 Hebrew Licensed Technology for (i) Cannabinoid phenolate metal salts, including mono, di and trivalent metals such as Li, Na, K, Ca, Mg, Zn, Fe and Al and their mixtures with native or synthetic cannabinoids, their pharmaceutical formulations, including for oral and topical administration; and (ii) pharmaceutical formulations, for the administration of cannabinoid chemical derivatives, including any and all veterinary and human medical conditions, including obesity, pain, inflammation and arthritis (the “2019 Field”).
Pursuant to the 2019 Hebrew Agreement, notwithstanding the grant of the 2019 Hebrew License, Yissum, on behalf of Hebrew University, will retain the right to (i) make, use and practice the 2019 Hebrew Licensed Technology for Hebrew University’s own research and educational purposes, but not for commercial purposes, and subject to the maintenance of confidentiality for any know-how or unpublished patent information contained in the 2019 Hebrew Licensed Technology; (ii) license or otherwise convey to other academic and not-for-profit research organizations the 2019 Hebrew Licensed Technology for use in non-commercial research and subject to the maintenance of confidentiality for any know-how or unpublished patent information contained in the 2019 Hebrew Licensed Technology; and (iii) license or otherwise convey the 2019 Hebrew Licensed Technology to any third party for research or commercial applications outside the 2019 Field, subject to the maintenance of confidentiality for any know-how or unpublished patent information contained in the 2019 Hebrew Licensed Technology.
The 2019 Hebrew Agreement further provides that CBR Pharma is entitled to grant one or more sublicenses to the 2019 Hebrew Licensed Technology for exploitation in the 2019 Field.
All right, title and interest in and to the 2019 Hebrew Licensed Technology vests solely in Yissum, and CBR Pharma will hold and make use of the rights granted pursuant to the 2019 Hebrew License solely in accordance with the terms of the 2019 Hebrew Agreement.
19
The 2019 Hebrew Licensed Technology will terminate upon the occurrence of the later of the following: (i) the expiration of the last of the 2019 Hebrew Licensed Patents; (ii) the expiration of the last exclusivity on any product granted by any regulatory or government body; (iii) the expiration of a continuous period of twenty years plus any applicable patent extension period, during which there was no commercial sale of any product in any country; or (iv) if we elect to obtain an exclusive license to the know-how under the terms of the 2019 Hebrew Agreement, the expiration of such exclusive license.
On January 1, 2020, CBR Pharma and Yissum entered into the first amendment to the 2018 Hebrew Agreement (the “First Hebrew Amendment”), which provided for additional research to be done at Yissum on new derivatives of certain molecules. Pursuant to the terms of the First Hebrew Agreement Amendment, we will pay Yissum $200,000 per year plus 35% additional for University overhead for the additional research performed by each professor over an 18-month period, starting May 1, 2019. The additional research ended in April 2021 and further preclinical work is expected to be undertaken following research and development of a potentially successful drug delivery method, which is in its late stage development.
Research Agreements with the University of Oxford
On November 1, 2013, our wholly-owned subsidiary 180 LP entered into an agreement (the “First Oxford Agreement”) with Oxford, pursuant to which 180 LP will sponsor Oxford’s research and development of repurposing anti-TNF for Dupuytren’s Contracture.
Pursuant to the First Oxford Agreement, each payment is to be made to ISIS Innovation (now Oxford University Innovation) at different milestones of the project, outlined below:
| Milestone | Milestone Fee | |||
| Minimum investment completed | £ | 10,000 | ||
| Initiation of Phase 2 trial for a licensed product | £ | 10,000 | ||
| Initiation of Phase 3 trial for a licensed product | £ | 10,000 | ||
| Registerable Phase 3 trial primary endpoint achieved for a licensed product | £ | 20,000 | ||
| Any issued U.S. patent of the licensed intellectual property rights | £ | 5,000 | ||
| Approval by FDA of a New Drug Application (“NDA”) filed by 180 LP or one of its sub-licensees for a licensed product | £ | 30,000 | ||
| Approval by EMA of an MAA filed by 180 LP or one of its sub-licensees for a licensed product | £ | 30,000 | ||
| First commercial sale of a licensed product by 180 LP or any sub-licensee in the U.S. | £ | 50,000 | ||
| First commercial sale of a licensed product by 180 LP or any sub-licensee in the EU | £ | 50,000 | ||
ISIS Innovation is also eligible for royalty payments equal to 0.5% of net sales in any country where there is a valid claim, 0.25% of net sales in other countries and a fee income royalty rate of 7.5% on all up-front, milestone and other one-off payments under or in connection with all sub-licenses and other contracts granted by 180 LP with respect to the licensed technology. The First Oxford Agreement is effective, unless earlier terminated, for so long as the specified patent application remains in effect as an issued patent, pending patent application or supplementary protection certificate or for a term of 20 years, whichever is longer.
On August 15, 2018, CannBioRex Pharma Limited, a company incorporated under the laws of England and Wales (“CannU.K.”) and a wholly-owned subsidiary of our wholly-owned subsidiary CBR Pharma, entered into the Research Agreement (the “Second Oxford Agreement”) with Oxford, pursuant to which CBR Pharma (through CannU.K.) has sponsored Oxford’s research and development of SCAs developed from the Hebrew Licensed Technology. At Oxford, the SCAs generated in the Hebrew University are being tested for analgesic and anti-inflammatory effects in established pre-clinical models.
Pursuant to the Second Oxford Agreement, Oxford undertook a research project (the “Research Project”) based around the clinical development of SCAs that are known to exhibit both anti-inflammatory and immunomodulatory properties. The aim of the Research Project was to develop and characterize chemical compounds that are synthesized at Hebrew University to create treatments for chronic pain, RA and other chronic inflammatory conditions, and to eventually obtain regulatory approval to initiate early-phase clinical trials by mid to late 2022 or as soon as possible thereafter. The Second Oxford Agreement had an initial term of one year beginning on March 22, 2019, but was extended by amendment to March 31, 2020, or any later date agreed to by the parties, unless terminated earlier. The Second Oxford Agreement was not extended any further after March 31, 2020, and CannU.K.’s relationship with Oxford continued with additional agreements with Oxford, as described below.
20
CannU.K., as the sponsor of the Research Project, made the following payments to Oxford pursuant to the Second Oxford Agreement:
| Milestone | Milestone Fee | |||
| Signature of the Oxford Agreement | £ | 166,800 | ||
| 6 months post start of the Research Project | £ | 166,800 | ||
| 9 months post start of the Research Project | £ | 166,800 | ||
| 12 months post start of the Research Project, after report | £ | 55,600 | ||
On September 18, 2020, CannU.K. entered into another research agreement with Oxford (the “Third Oxford Agreement”), pursuant to which CannU.K. sponsors work led by Prof. Nanchahal at the University of Oxford to investigate the mechanisms underlying fibrosis. In connection with the agreement, CannU.K. initially provided $100,000 and then at 6-month intervals further funding to support the salary of Dr. Lynn Williams and consumables.
CannU.K., as the sponsor, agreed to make the following payments to Oxford pursuant to the Third Oxford Agreement:
| Milestone | Amount Due (excluding VAT) | |||
| 30 days post signing of the Third Oxford Agreement | £ | 80,000 | ||
| 6 months post signing of the Third Oxford Agreement | £ | 178,867 | ||
| 12 months post signing of the Third Oxford Agreement | £ | 178,867 | ||
| 24 months post signing of the Third Oxford Agreement | £ | 178,867 | ||
| 36 months post signing of the Third Oxford Agreement | £ | 178,867 | ||
On September 21, 2020, CannU.K. entered into another research agreement with Oxford (the “Fourth Oxford Agreement”), pursuant to which CannU.K. agreed to sponsor work at the University of Oxford to develop and characterize novel cannabinoid derived new chemical entities (NCEs) for the treatment of inflammatory diseases towards initiation of early phase clinical trials in patients within a period of 3 years.
CannU.K., as the sponsor, agreed to make the following payments to Oxford pursuant to the Fourth Oxford Agreement:
| Milestone | Amount Due (excluding VAT) | |||
| 30 days post signing of the Fourth Oxford Agreement | £ | 101,778 | ||
| 6 months post signing of the Fourth Oxford Agreement | £ | 101,778 | ||
| 12 months post signing of the Fourth Oxford Agreement | £ | 101,778 | ||
| 18 months post signing of the Fourth Oxford Agreement | £ | 101,778 | ||
| 24 months post signing of the Fourth Oxford Agreement | £ | 101,778 | ||
21
On March 22, 2022, CannU.K. entered into an amendment to the Fourth Oxford Agreement, to extend the research period to December 31, 2023, at no additional cost to CannU.K.
On May 24, 2021, CannU.K. entered into another research agreement with Oxford (the “Fifth Oxford Agreement”), pursuant to which CannU.K. will sponsor work at the University of Oxford to conduct a multi-center, randomized, double blind, parallel group, feasibility study of anti-TNF injection for the treatment of adults with frozen shoulder during the pain-predominant phase.
CannU.K., as the sponsor, agreed to make the following payments to Oxford pursuant to the Fifth Oxford Agreement:
| Milestone | Amount Due (excluding VAT) | |||
| Upon signing of the Fifth Oxford Agreement | £ | 70,546 | ||
| 6 months post signing of the Fifth Oxford Agreement | £ | 70,546 | ||
| 12 months post signing of the Fifth Oxford Agreement | £ | 70,546 | ||
| 24 months post signing of the Fifth Oxford Agreement | £ | 70,546 | ||
Oxford License Agreement
On November 3, 2021, we entered into an exclusive license agreement with Oxford University Innovation Limited (“Oxford License Agreement”), pursuant to which we were granted the rights to certain patents related to the HMGB1 molecule for liver regeneration.
Pursuant to the Oxford License Agreement, we agreed to the following payment terms:
| Payment | Amount Due | |||
| Past patent costs | £ | 49,207 | ||
| License fee | £ | 10,000 | ||
| Annual maintenance fee | £ | 3,000 | ||
| Milestone | Amount Due | |||
| Submission of IND | £ | 25,000 | ||
| 1st Subject dosed in Phase I studies for each product, each indication | £ | 25,000 | ||
| 1st Subject dosed in Phase II studies for each product, each indication | £ | 100,000 | ||
| 1st Subject dosed in Phase III studies for each product, each indication | £ | 50,000 | ||
| Submission of New Drug Application for each product for each indication | £ | 50,000 | ||
| Issued U.S. patent, per patent | £ | 5,000 | ||
| Receipt of Regulatory Approval in the U.S. for each product for each indication | £ | 1,250,000 | ||
| Receipt of Regulatory Approval in the EU or U.K. for each product for each indication | £ | 550,000 | ||
| Receipt of Regulatory Approval in the Japan for each product for each indication | £ | 150,000 | ||
| Aggregate Net Sales Exceed $5Bn | £ | 10,000,000 | ||
| Aggregate Net Sales Exceed $10Bn | £ | 50,000,000 | ||
| Net Sales (US$) | Royalty Rate | |||
| < $250M | 1.00 | % | ||
| $250M - $1B | 2.00 | % | ||
| $1B - $10B | 3.00 | % | ||
| > $10B | 3.50 | % | ||
22
Due to the ongoing costs of this research program and the need for the Company to focus its resources on the Company’s primary platform to treat fibrosis using anti-TNF (tumor necrosis factor), the Board of Directors of the Company elected to terminate the Company’s HMGB1 license agreement with Oxford on September 22, 2023, and on September 22, 2023, the Company and Oxford entered into a termination letter, formally terminating the License effective September 22, 2023. The termination letter also clarified amounts that we owed after termination of the License, including approximately $20,000 in unbilled fees. No material early termination penalties were incurred by the Company in connection with the termination of the license.
Stanford License Agreement
The license agreement with Stanford University covering α7nAChR was terminated effective November 23, 2024. After a careful review of the Company’s intellectual property portfolio as part of the Company’s ongoing strategic review process, the Company decided to move in a different direction and returned the intellectual property to Stanford University.
Kennedy License Agreement
On September 27, 2019, our wholly-owned subsidiary 180 LP entered into an exclusive license agreement (the “Kennedy License Agreement”) with the Kennedy Trust For Rheumatology Research (“Kennedy”), pursuant to which Kennedy granted to 180 LP an exclusive license in the U.S., Japan and member countries of the EU (including the United Kingdom), to certain licensed patents (the “Kennedy Licensed Patents”), including the right to grant sublicenses, and the right to research, develop, sell or manufacture any pharmaceutical product (i) whose research, development, manufacture, use, importation or sale would infringe on the Kennedy Licensed Patents absent the license granted under the Kennedy License Agreement or (ii) containing an antibody that is a fragment of or derived from an antibody whose research, development, manufacture, use, importation or sale would infringe on the Kennedy Licensed Patents absent the license granted under the Kennedy License Agreement, for all human uses, including the diagnosis, prophylaxis and treatment of diseases and conditions.
Under the Kennedy License Agreement, Kennedy reserves the perpetual, irrevocable, non-exclusive, royalty-free, sublicensable, worldwide right for the Kennedy Licensed Patents and its affiliates, employees, students and other researchers to carry out any acts which would otherwise infringe on the Kennedy Licensed Patents for the purposes of teaching and carrying out research and development, including the right to accept external sponsorship for such research and development and the right to grant sub-licenses for the same purposes.
As consideration for the grant of the Kennedy Licensed Patents, 180 LP paid Kennedy an upfront fee of £60,000, and will also pay Kennedy royalties equal to (i) 1% of the net sales for the first annual $1 billion of net sales, and (ii) 2% of the net sales after the net sales are at or in excess of $1 billion, as well as 25% of all sublicense revenue, provided that the amount of such percentage of sublicense revenue based on amounts which constitute royalties shall not be less than 1% on the first cumulative $1 billion of net sales of the products sold by such sublicenses or their affiliates, and 2% on that portion of the cumulative net sales of the products sold by such sublicenses or their affiliates in excess of $1 billion.
The term of the royalties paid to Kennedy will expire on the later of (i) the last valid claim of a patent included in the Kennedy Licensed Patents which covers or claims the exploitation of a product in the applicable country; (ii) the expiration of regulatory exclusivity for the product in the country; or (iii) 10 years from first commercial sale of the product in the country.
We may terminate the Kennedy License Agreement without cause by providing 90-days’ notice.
23
Kinexum Agreement
On September 9, 2021, we entered into a contract with Kinexum in the ordinary course of business (the “MSA”). Pursuant to the MSA, Kinexum will provide assistance to us in connection with the Conditional Marketing Authorisation (CMA) and Marketing Approval Application (MAA) which we previously planned to submit to the MHRA in connection with our previously planned use of adalimumab to treat progressive early-stage Dupuytren’s disease. The MSA expired on September 9, 2024.
Consulting Agreements
The Consulting Agreements are each described below.
Prof. Jagdeep Nanchahal Consulting Agreement
On February 25, 2021, we (and CannBioRex Pharma Limited, which was added as a party to the agreement later), entered into a Consultancy Agreement dated February 22, 2021, and effective December 1, 2020, with Prof. Jagdeep Nanchahal (as amended, the “Consulting Agreement”). Prof. Nanchahal has been providing services to us and/or our subsidiaries since 2014, was previously a greater than 5% stockholder of the Company and is currently the Chairman of our Clinical Advisory Board.
On March 31, 2021, we entered into a first amendment to Consultancy Agreement with Prof. Jagdeep Nanchahal (the “First Nanchahal Amendment”), which amended the Consultancy Agreement entered into with Prof. Nanchahal on February 25, 2021, to include CannBioRex Pharma Limited, a corporation incorporated and registered in England and Wales (“CannBioRex”), and an indirect wholly-owned subsidiary of the Company, as a party thereto, and to update the prior Consultancy Agreement to provide for cash payments due to Prof. Nanchahal to be paid by CannBioRex, for tax purposes, provide for CannBioRex to be party to certain other provisions of the agreement and to provide for the timing of certain cash bonuses due under the terms of the agreement.
Prof. Nanchahal is a surgeon scientist focusing on defining the molecular mechanisms of common diseases and translating his findings through to early phase clinical trials. He undertook his Ph.D., funded by the U.K. Medical Research Council, whilst a medical student in London and led a lab group funded by external grants throughout his surgical training. After completing fellowships in microsurgery and hand surgery in the USA and Australia, he was appointed as a senior lecturer at Imperial College. His research is focused on promoting tissue regeneration by targeting endogenous stem cells and reducing fibrosis. In 2013 his group identified anti-tumor necrosis factor (TNF) as therapeutic target for Dupuytren’s Contracture, a common fibrotic condition of the hand. He previously lead a Phase 2b clinical trial funded by the Wellcome Trust and Department of Health to assess the efficacy of local administration of anti-TNF in patients with early-stage Dupuytren’s Contracture and a clinical trial for patients with early-stage frozen shoulder. He is a proponent of evidence-based medicine and was the only plastic surgery member of the NICE Guidance Development Groups on complex and non-complex fractures. He was a member of the group that wrote the Standards for the Management of Open Fractures published in 2020. This is an open-source publication to facilitate the care of patients with these severe injuries.
Pursuant to the Consulting Agreement, Prof. Nanchahal agreed, during the term of the agreement, to serve as a consultant to us and provide such services as the Chief Executive Officer and/or our Board shall request from time to time, including but not be limited to: (1) conducting clinical trials in the fields of Dupuytren’s Contracture, frozen shoulder and post-operative delirium/cognitive decline; and (2) conducting laboratory research in other fibrotic disorders, including fibrosis of the liver and lung (collectively, the “Services”).
24
In consideration for providing the Services, we (through CannBioRex Pharma Limited) agreed to pay Prof. Nanchahal 15,000 British Pounds (GBP) per month (approximately $20,800) during the term of the agreement, increasing to GBP 23,000 (approximately $32,000) on the date (a) of publication of the data from the phase 2b clinical trial for Dupuytren’s Contracture (RIDD) and (b) the date that we have successfully raised over $15 million in capital. The fee will increase annually thereafter to reflect progression in other clinical trials and laboratory research as approved by our Board. We also agreed to pay Prof. Nanchahal a bonus (“Bonus 1”) in the sum of GBP 100,000 upon submission of the Dupuytren’s Contracture clinical trial data for publication in a peer-reviewed journal, which submission occurred in December 2021, and which bonus was paid in December 2021. In addition, for prior work performed, including completion of the recruitment to the RIDD (Dupuytren’s) trial, we agreed to pay Prof. Nanchahal GBP 434,673 (approximately $605,000) (“Bonus 2”). At the election of Prof. Nanchahal, Bonus 2 shall be paid at least 50% (fifty percent) or more, as Prof. Nanchahal elects, in shares of our common stock, at a share price of $1,140.00 per share, or the share price on the date of the grant, whichever is lower, with the remainder paid in GBP. Bonus 2 shall be deemed earned and payable upon us raising a minimum of $15 million in additional funding, through the sale of debt or equity, after December 1, 2020 (the “Vesting Date”) and shall not be accrued, due or payable prior to such Vesting Date. Bonus 2 shall be payable by us within 30 calendar days of the Vesting Date. Finally, Prof. Nanchahal shall receive another one-time bonus (“Bonus 3”) of GBP 5,000 (approximately $7,000) on enrollment of the first patient to the phase 2 frozen shoulder trial, and another one-time bonus (“Bonus 4”) of GBP 5,000 (approximately $7,000) for enrollment of the first patient to the phase 2 delirium/POCD trial. On March 30, 2021, we issued Prof. Nanchahal 265 shares of our common stock in lieu of GBP 217,337 and on April 15, 2021, we issued Prof. Nanchahal 99 shares of our common stock in lieu of GBP 82,588. We also waived the requirement for the Company having to raise $15 million in order for Prof. Nanchahal to agree to receive an aggregate of GBP 300,000 via the issuance of shares. Prof. Nanchahal agreed that the remaining GBP 134,673 that is due pursuant to Bonus 2 shall be paid after we have raised a minimum of $15 million in additional funding. On August 23, 2021, at the request of Prof. Nanchahal, we agreed to issue Prof. Nanchahal 161 shares of common stock in consideration for the remaining 31% (or 134,749 GBP, or $184,606) of Bonus 2, based on a $1,140.00 per share price. The shares were issued under our 2020 Omnibus Incentive Plan, which has been approved by stockholders.
Effective on April 27, 2022, we and CannBioRex entered into a Second Amendment to Consulting Agreement with Prof. Jagdeep Nanchahal (the “Second Nanchahal Amendment”). Pursuant to the Second Nanchahal Amendment, Prof. Nanchahal agreed that upon acceptance of the data for the phase 2b clinical trial for Dupuytren’s disease for publication (which occurred March 1, 2022, subject to editing and final approvals), his monthly fee was increased to £23,000, provided that £4,000 of such increase shall be accrued and £19,000 per month of such fees shall be payable per our payroll practices in cash by us starting effective March 1, 2022, and until the earlier of (a) November 1, 2022 or (b) such time as our Board determines that we have sufficient cash on hand to pay such accrued amounts, which we expect will not be until we have raised a minimum of $15,000,000 (the “Funding Determination Date”), at which time all accrued amounts shall be due.
On December 28, 2022, we and CannBioRex, entered into a Third Amendment to Consultancy Agreement with Prof. Nanchahal (the “Third Nanchahal Amendment”). The Third Nanchahal Amendment amended the Consultancy Agreement to provide that the monthly cash fee payable to Prof. Nanchahal pursuant to such agreement would remain at its then current rate, £23,000 per month, through December 31, 2022, and then increase to £35,000 per month during the term of the Consultancy Agreement from January 1, 2023, until the end of the term of the Consultancy Agreement (collectively, the “Fee”). The Third Nanchahal Amendment also provided that the Fee will be adjusted yearly with the recommendation of our Board or the Compensation Committee of the Company, which will consider in its determination of the amount of such increase, the U.K. consumer price index and Prof. Nanchahal’s contributions to advancing our mission, among other things. The Third Nanchahal Amendment also provided that in the event the Consultancy Agreement is terminated by us for any reason other than cause, Prof. Nanchahal is entitled to a lump sum payment of 12 months of his monthly fee as of the date of termination.
Notwithstanding the above, the Board or Compensation Committee of the Company may grant Prof. Nanchahal additional bonuses from time to time in their discretion, in cash, stock or options.
25
The Consulting Agreement has an initial term of three years, and renews thereafter for additional three-year terms, until terminated as provided in the agreement, and currently has a term through December 1, 2026. The Consulting Agreement can be terminated by either party with 12 months prior written notice (provided our right to terminate the agreement may only be exercised if Prof. Nanchahal fails to perform his required duties under the Consulting Agreement), or by us immediately if (a) Prof. Nanchahal fails or neglects efficiently and diligently to perform the Services or is guilty of any breach of its or his obligations under the agreement (including any consent granted under it); (b) Prof. Nanchahal is guilty of any fraud or dishonesty or acts in a manner (whether in the performance of the Services or otherwise) which, in our reasonable opinion, has brought or is likely to bring Prof. Nanchahal, the Company or any of its affiliates into disrepute or is convicted of an arrestable offence (other than a road traffic offence for which a non-custodial penalty is imposed); or (c) Prof. Nanchahal becomes bankrupt or makes any arrangement or composition with his creditors. If the Consulting Agreement is terminated by us for any reason other than cause, Prof. Nanchahal is entitled to a lump sum payment of 12 months of his fee as at the date of termination.
The Consulting Agreement includes a 12 month non-compete and non-solicitation obligation of Prof. Nanchahal, preventing him from competing against us in any part of any country in which he was actively engaged in our business, subject to certain exceptions, including research conducted at the University of Oxford. The Consulting Agreement also includes customary confidentiality and assignment of inventions provisions, in each case subject to our previously existing agreements with various universities, including the University of Oxford, where Prof. Nanchahal serves as a Professor of Hand, Plastic and Reconstructive Surgery.
Service Agreement with Prof. Sir Marc Feldmann
On March 7, 2024, Sir Marc Feldmann, Ph.D. provided notice to the Board of Directors of his resignation as a member of the Board of Directors, effective on the same date.
On January 10, 2024, and effective on January 1, 2024, the Company entered into a Third Amendment to Consulting Agreement with Lawrence Steinman. Pursuant to the amendment, Dr. Steinman, effective as of January 1, 2024, agreed to a reduction of his base salary set forth in his consulting agreement by 100%, to $0 per year, with the amount of such salary reduction ($18,750 per month or $225,000 per year), accruing monthly in arrears, to be paid on the Funding Date, provided that in the event the Funding Date does not occur prior to March 15, 2025, the amounts accrued will be forgiven in their entirety. On May 7, 2024, the Company and Dr. Steinman entered into the Fourth Amendment to Consulting Agreement whereby the Company agreed to pay Dr. Steinman as a regular member of the Board, and Dr. Steinman agreed to release and forgive the Company from any accrued and unpaid fees.
Dr. Steinman is currently a Board member of the Company, and receives compensation of $50,000 per annum in connection with this role. He does not receive any additional compensation from the Company.
Recent Events
Warrant Inducement Agreement and Related Transactions
On October 16, 2024, we entered into a warrant inducement agreement (the “Inducement Agreement”) with a holder (the “Holder”) of warrants to purchase 950,069 shares of common stock of the Company (the “Exercised Warrants”), pursuant to which the Holder agreed to exercise for cash the Exercised Warrants at an exercise price of $3.48 per share ($0.25 greater than the $3.23 per share exercise price of such Exercised Warrants) during the period from the date of the Inducement Agreement until 1:15 p.m., Eastern Time, on October 16, 2024. On October 16 and 17, 2024, the Exercised Warrants were exercised in full for cash by the Holder and the Company received $3,306,240 before deducting financial advisory fees and other expenses payable by us.
In consideration of the Holder’s agreement to exercise the Exercised Warrants in accordance with the Inducement Agreement, the Company agreed to issue new unregistered Warrants to Purchase Shares of Common Stock (the “Inducement Warrants”) to purchase a number of shares of common stock equal to 200% of the number of shares of common stock issued upon exercise of the Exercised Warrants, i.e., warrants to purchase up to 1,900,138 shares of common stock (the “Inducement Warrant Shares”). The Inducement Warrants were immediately exercisable and have a term of exercise of five years.
26
The Company agreed in the Inducement Agreement to file a registration statement on Form S-1 to register the resale of the Inducement Warrant Shares upon exercise of the Inducement Warrants (the “Resale Registration Statement”) by November 15, 2024 (which deadline was met), and to use commercially reasonable efforts to have such Resale Registration Statement declared effective by the Commission within sixty (60) calendar days following the date of filing with the Commission (or ninety (90) calendar days following the date of filing with the Commission in the event of a ‘full review’ by the Commission) and to keep the Resale Registration Statement effective at all times until no holder of the Inducement Warrants owns any Inducement Warrants or Inducement Warrant Shares. In the event that the Company fails to timely deliver to the Holder the Inducement Warrant Shares without restrictive legends, the Company has agreed to pay certain liquidated damages to the Holder. The Resale Registration Statement was declared effective by the SEC within the timeline agreed to by the Company and no penalties were due.
The Company has used, and plans to continue to use, the net proceeds from these transactions to advance the commercialization of the Gaming Technology Platform, and for working capital, and other general corporate purposes.
The Inducement Warrants have an exercise price of $1.50 per share. The exercise price and the number of shares of common stock issuable upon exercise of each Inducement Warrant are subject to appropriate adjustments in the event of certain stock dividends and distributions, stock splits, stock combinations, reclassifications or similar events affecting the common stock. In addition, in certain circumstances, upon a fundamental transaction, a holder of Inducement Warrants will be entitled to receive, upon exercise of the Inducement Warrants, the kind and amount of securities, cash or other property that such holder would have received had they exercised the Inducement Warrants immediately prior to the fundamental transaction.
The Company may not affect the exercise of Inducement Warrants, and the applicable Holder will not be entitled to exercise any portion of any such Inducement Warrant, which, upon giving effect to such exercise, would cause the aggregate number of shares of common stock beneficially owned by the holder of such Inducement Warrant (together with its affiliates) to exceed 4.99% or 9.99%, as applicable, of the number of shares of common stock outstanding immediately after giving effect to the exercise, as such percentage ownership is determined in accordance with the terms of such Inducement Warrants.
The Company engaged A.G.P./Alliance Global Partners (“A.G.P.”) to provide exclusive financial services in connection with the transactions summarized above and, pursuant to a Financial Advisory Agreement between the Company and A.G.P., paid A.G.P. a financial advisory fee of $232,000, and an alternative transaction fee of $100,000. In addition, we reimbursed A.G.P. for its accountable legal expenses in connection with the exercise of the Exercised Warrants and the issuance of the Inducement Warrants of $65,000 and $10,000 non-accountable expenses. In addition, we paid A.G.P. $29,923, half of the financial advisory fees due in connection with a December 2023 warrant inducement. As of the date of this Report, a total of $436,923 related to the aforementioned transaction has been paid to A.G.P and there is no balance outstanding.
Registered Direct and Concurrent Private Offering
On December 27, 2024, we entered into a Securities Purchase Agreement (the “SPA”) with certain accredited investors, including the Holder (the “Investors”). Pursuant to the terms of the SPA, the Company agreed to sell, in a registered direct offering, an aggregate of 1,200,000 shares (the “December 2024 Shares”) of the Company’s common stock and, in a concurrent private placement, warrants (the “December 2024 Warrants”) to purchase up to 1,200,000 shares of common stock (the shares of common stock issuable upon exercise of the December 2024 Warrants, the “December 2024 Warrant Shares”). The combined purchase price per December 2024 Share and December 2024 Warrant was $2.41. The offerings closed on December 30, 2024.
The December 2024 Warrants were immediately exercisable on their grant date at an exercise price of $2.28 per share and expire five and a half years following the initial exercise date.
27
Pursuant to a placement agency agreement (the “Placement Agency Agreement”) dated as of December 27, 2024, between the Company and Maxim Group LLC (the “Placement Agent”), the Company engaged the Placement Agent to act as the Company’s sole placement agent in connection with the registered direct offering. Pursuant to the Placement Agency Agreement, the Company agreed to pay the Placement Agent a cash fee equal to seven percent (7.0%) of the gross proceeds received by the Company from the sale of the December 2024 Shares and December 2024 Warrants and to reimburse the Placement Agent for certain of its expenses in an aggregate amount of $50,000. The Placement Agency Agreement contains customary representations, warranties and agreements by the Company, customary conditions to closing, indemnification obligations of the Company and the Placement Agent, including for liabilities under the Securities Act, other obligations of the parties, and termination provisions.
The Company agreed to file a registration statement under the Securities Act with the SEC, covering the resale of the December 2024 Warrant Shares within 30 calendar days following the date of the SPA, which has been filed to date, and to use commercially reasonable efforts to cause the registration statement to be declared effective by the SEC within 91 days following the closing of the offerings, which was declared effective timely.
The net proceeds to the Company from the registered direct offering and concurrent private placement, after deducting the placement agent’s fees and expenses and the Company’s offering expenses were approximately $2.6 million. The Company intends to use the net proceeds from the transactions for working capital and general corporate purposes, which may include operationalizing and developing the Gaming Technology Platform and capital expenditures.
The December 2024 Shares (but not the December 2024 Warrants or the December 2024 Warrant Shares) were offered and sold by the Company pursuant to a prospectus supplement, dated as of December 30, 2024, which was filed with the SEC in connection with a takedown from the Company’s effective shelf registration statement on Form S-3, which was initially filed with the SEC on June 3, 2022, and subsequently declared effective on June 24, 2024 (File No. 333-265416).
Intellectual Property
Our success depends in significant part on our ability to protect the proprietary elements of our product candidates, technology and know-how, to operate without infringing on the proprietary rights of others, and to defend challenges and oppositions from others and prevent others from infringing on our proprietary rights. We have sought, and will continue to seek, patent protection in the U.S., U.K., Europe and other countries for our proprietary technologies. The Company and its partners hold patents for both of the two remaining biotechnology related assets: ATNF and SCA.
With respect to the ATNF related intellectual property, we are currently involved with “five” patent families as follows:
| 1. | a patent family owned by Oxford University Innovations (OUI) relating to treating Dupuytren’s Disease with a Tumor Necrosis Factor (TNF) Alpha Antagonist which is licensed to the Company; |
| 2. | a patent family owned by the Kennedy Trust for Rheumatology Research relating to Preventing Post-Operative Cognitive Decline (POCD) by administering a TNF Alpha Antagonist during surgery which is licensed to the Company; |
| 3. | a patent family owned by the Company relating to a pre-filled syringe for treating early stage Dupuytren’s Disease with adalimumab or infliximab; |
| 4. | a patent family owned by the Company relating to use of an IL-33 Antagonist to treat a localized fibrotic disorder such as early stage Dupuytren’s Disease; and |
| 5. | a patent family owned by the Company relating to use of a TNF Alpha Antagonist to treat a localized fibrotic disorder such as early stage Dupuytren’s Disease. |
For the second biotechnology related asset, SCA, this patent portfolio consists of one patent family that has entered national stage in Australia, Canada, Europe, Israel, Japan, Korea, Mexico, and USA. All patent applications are pending, and no substantial examination has started in any jurisdiction. In all jurisdictions, at this point, the claims of the applications are directed to novel SCAs and methods for their preparation, compositions comprising them and uses thereof in the treatment of inflammation and/or pain and/or obesity.
Our policy is to seek patent protection for the technology, inventions and improvements that we consider important to the development of our business, but only in those cases where we believe that the costs of obtaining patent protection is justified by the commercial potential of the technology, and typically only in those jurisdictions that we believe present significant commercial opportunities. We also rely on trademarks, trade secrets, know-how and continuing innovation to develop and maintain our competitive position.
The term of individual patents depends upon the countries in which they are obtained. In most countries in which we have filed, the patent term is 20 years from the earliest date of filing a non-provisional patent application. In the U.S., a patent’s term may be lengthened by patent term adjustment, which compensates a patentee for administrative delays by the U.S. Patent and Trademark Office (“USPTO”), in granting a patent, or may be shortened if a patent is terminally disclaimed over another patent.
28
The term of a patent that covers an FDA-approved drug may also be eligible for extension, which permits term restoration as compensation for the term lost during the FDA regulatory review process. The Drug Price Competition and Patent Term Restoration Act of 1984 (the Hatch-Waxman Act) permits an extension of up to five years beyond the expiration of the patent. The length of the patent term extension is related to the length of time the drug is under regulatory review. Extensions cannot extend the remaining term of a patent beyond 14 years from the date of product approval and only one patent applicable to an approved drug may be extended. Similar provisions to extend the term of a patent that covers an approved drug are available in Europe and other non-U.S. jurisdictions.
To protect our rights to any of our issued patents and proprietary information, we may need to litigate against infringing third parties, avail ourselves of the courts or participate in hearings to determine the scope and validity of those patents or other proprietary rights.
We also rely on trade secret protection for our confidential and proprietary information. Our policy requires our employees, consultants, outside scientific collaborators, sponsored researchers and other advisors to execute confidentiality agreements upon the commencement of employment or consulting relationships with us.
From time to time, in the normal course of our operations, we will be a party to litigation and other dispute matters and claims relating to intellectual property.
In September 2024, the Company acquired valuable intellectual property from Elray, consisting of proprietary gaming technology and specialized operational know-how. While this intellectual property is not protected through patents, it remains safeguarded through trade secrets and proprietary operating knowledge, ensuring that the competitive advantage derived from Elray’s innovations remains secure. The acquired intellectual property encompasses critical insights, algorithms, and operational knowledge.
180LS’ Research, Development and License Agreements
180LS has entered into research and licensing agreements with various parties, including the Hebrew University of Jerusalem and Oxford. For information regarding these agreements, see “Material Agreements”, above.
Competition
iGaming Competition
The global iGaming business is highly competitive, driven by rapid technological advancements and the increasing demand for digital entertainment from customers globally, particularly in the mobile format. Major players in the industry compete by offering innovative platforms, diverse game selections, and enticing promotional offers (with correspondingly high customer acquisition costs). The proliferation of mobile gaming and live dealer options has added an extra layer of differentiation, as companies strive to create immersive and user-friendly experiences. Additionally, localization strategies are critical, with operators tailoring games and marketing to suit the preferences and regulatory requirements of specific regions, such as Europe, North America, and emerging markets like Asia and Latin America where much of the growth in iGaming is taking place. As new entrants continually join the market, the competition is further intensified by startups leveraging blockchain technology and cryptocurrency, offering transparency and unique features to attract tech-savvy users and a growing customer base that prefers to transact using cryptocurrencies.
The iGaming industry faces certain external challenges that impact competition, including increasingly complex regulatory environments and shifting consumer preferences. Both B2B and B2C operators must navigate complex licensing and compliance requirements, which vary significantly across jurisdictions, often leading to additional costs and operational hurdles. Furthermore, maintaining customer loyalty in an oversaturated market is increasingly difficult, pushing companies to invest heavily in loyalty programs, responsible gaming initiatives, and cutting-edge technology like artificial intelligence for personalized gaming experiences.
29
Management feels that the Company is very well placed in this competitive environment. Specifically, we believe that there are four key features of the Company and its Gaming Technology Platform that management believes offer a competitive advantage:
1. Full cryptocurrency capability – The Gaming Technology Platform recently acquired by the Company offers full cryptocurrency capability supported by blockchain technology. Management believes this to be a key focus area for gaming participants, one where potential customers have increasing interest and engagement;
2. Player management and loyalty – As noted above, maintaining customer loyalty is critical in a very competitive market. The Company believes that its technology will allow for “best in class” player engagement, potentially increasing customer value versus customer acquisition cost;
3. Unique content – The Company is currently in early conversations with multiple unique content production and distribution companies in the iGaming sector. If agreements are reached with one or more of such entities (including but not limited to joint-ventures, acquisitions, mergers and various partnerships), the Company believes that this will provide a significant competitive advantage if the Company were to ultimately launch a B2C focused business. The Company hopes to be able to leverage the Gaming Technology Platform to provide potential clients with highly unique, iGaming related content, thus reducing customer acquisition cost and increasing customer lifetime value; and
4. M&A and industry expertise – Management of the Company believes that the management and shareholders of the Company provide a unique blend of capital markets expertise related to negotiating, structuring and executing M&A transactions, plus unique industry knowledge through the involvement of experienced industry executives. Management believes this provides the Company with access to diverse and unique relationships, leading to potential acquisition and partnership opportunities. When combined with M&A expertise, management believes that the Company has a distinct competitive advantage relative to certain peers.
In terms of opportunity, cryptocurrency iGaming casinos have gained significant traction globally, which management believes presents a major opportunity for the Company given the cryptocurrency focus of the Gaming Technology Platform.
Life Science Competition
Below is a description of the competitive environment of each of our product candidate development platforms and potential product candidates.
Dupuytren’s Contracture
Our treatment is for early-stage Dupuytren’s Contracture, for which, to our knowledge, there is no approved treatment. Existing treatments focus on late stage Dupuytren’s Contracture, when the fingers are irreversibly curled into the palm. Surgery remains the typical standard treatment, but the relatively long post-operative rehabilitation has driven the reach for less invasive techniques. Xiaflex, a drug developed by Auxilium, has shown effective in treating patients with developed contractures although many patients experience relatively mild side effects. An alternative approach is disruption of the late-stage cords with a needle and data from a comparative clinical trial published in the Journal of Bone and Joint Surgery (American) in 2018 showed similar recurrence rates between collagenase and percutaneous needle fasciotomy at 2 years. A clinical trial funded by the National Institute for Health Research Health Technology Assessment Programme (U.K.) is currently underway in the U.K., comparing the cost efficacy of surgery for Dupuytren’s Contracture with collagenase treatment. The aims of the study are to determine (i) whether collagenase injections are as effective and as safe as surgery for treating this condition and (ii) the costs of both treatments.
30
SCAs
Following the acquisition of GW Pharmaceuticals PLC and its Epidiolex (cannabidiol) and Sativex (THC & CBD) franchises, by Jazz Pharmaceuticals (Ireland), Jazz Pharma has become the prominent player in the cannabidiol space. Epidiolex is an oral cannabidiol solution approved for treating seizures in a range of childhood epileptic diseases, including Dravet’s syndrome (formerly known as severe myoclonic epilepsy of infancy), Rett Syndrome, and Lennox-Gastaut Syndrome. Jazz Pharma is exploring whether Epidiolex is effective in Sturge-Weber Syndrome, in which abnormal development of blood vessels leads to defects in the brain, skin, and eyes from birth, and more broadly in Autism Spectrum Disorder. Clinical trials sponsored by Jazz Pharma testing effectiveness of Epidiolex in autoimmune diseases such as multiple sclerosis, ulcerative colitis, and Crohn’s Disease are ongoing. Collectively, these efforts represent the most extensive cannabidiol clinical program.
To our knowledge, multiple companies are working in the cannabis therapeutic area and are pursuing regulatory approval for their product candidates, including:
| ● | Cardiol Therapeutics (Canada) which is evaluating the effectiveness of their oral CBD liquid formulation on myocardial recovery in patients presenting with acute myocarditis. |
| ● | Zynerba Pharmaceuticals (Pennsylvania) which focuses on pharmaceutically produced transdermal cannabinoid therapies for rare and near-rare neuropsychiatric disorders. Zynerba currently is evaluating ZygelTM, a patent-protected transdermal CBD gel for the treatment of Fragile X syndrome, for which it filed an NDA with the FDA, developmental and epileptic encephalopathies, 22q deletion syndrome, and Autism Spectrum Disorder. |
| ● | Orcosa, (New Jersey) which is testing their CBD, Oravexx (oral disintegrating tablets), to manage pain and inflammation with the hope of reducing clinical reliance on opioids. The particular indication is pain associated with osteoarthritis in the knee in a Phase 2 trial. |
| ● | Stero Biotechs (Israel) which sponsored a phase II trial in GVHD demonstrating that CBD administration (synthetic CBD in olive oil), either enhanced the therapeutic effect of steroids or reduced the steroid dosage while maintaining or improving the steroid’s original therapeutic effect. Additional clinical trials are a Phase Iia, multi centered trial in steroid dependent Crohn’s disease, a Phase Iia trial in chronic urticaia (Hives), and Phase I/II trial in severe Covid-19. |
Government Regulation
iGaming Governmental Regulation
The global iGaming (online casino) industry has experienced exponential growth, driven by increased internet penetration, the popularity of mobile gaming, and the rising adoption of cryptocurrency-based transactions. However, as the sector has expanded, regulatory frameworks worldwide have evolved to address concerns related to responsible gaming, anti-money laundering (AML), data privacy, and player protection. Various jurisdictions have adopted differing approaches, with some fostering innovation while others impose restrictive or protectionist policies. The following is a summary of key iGaming jurisdictions and the evolving regulatory regime in such jurisdictions. By review of such jurisdictions, the Company is not indicating that it is, or is necessarily intending to, operate in such jurisdiction. The Company continues to evaluate potential licensing jurisdictions.
1. Europe: Mature and Evolving Regulatory Regimes
a) United Kingdom (UK)
The UK Gambling Commission (UKGC) remains one of the most stringent regulatory bodies globally, overseeing remote gaming operators. Regulations in the UK include:
| ● | Mandatory adherence to anti-money laundering (AML) and know-your-customer (KYC) protocols. |
| ● | Stringent responsible gambling measures, including affordability checks and limits. |
31
| ● | A 21% remote gaming duty applied to gross gaming revenue (GGR). |
| ● | Recent Developments: |
| ○ | The UK Gambling Act 2005 is under reform, with proposals for affordability checks, maximum stake limits, and tighter controls on online slot games. |
| ○ | The anticipated 2025 White Paper Implementation is expected to introduce stricter advertising regulations and enhanced player protections. |
b) Malta
Malta, regulated by the Malta Gaming Authority (MGA), is a hub for European and global iGaming operators and is considered a high-quality regime for iGaming licensing. The MGA licenses operators offering B2B and B2C services across Europe under a multi-license framework.
| ● | Recent Developments: |
| ○ | In 2024, Malta amended its Gaming Act to enhance AML compliance and introduce stricter oversight for B2B suppliers. |
| ○ | The MGA introduced a voluntary code for responsible gambling and is enhancing oversight of player data protection. |
c) Germany
Germany implemented its Interstate Treaty on Gambling (Glücksspielstaatsvertrag 2021) in July 2021, opening the market to licensed operators but imposing:
| ● | A 5.3% tax on stakes for online slots and poker. |
| ● | Strict advertising restrictions and deposit limits (€1,000 monthly limit per player). |
| ● | Challenges: The slow approval process for licenses and a complex regulatory environment have deterred some operators. |
d) Netherlands
The Dutch Remote Gambling Act (KOA) came into effect in October 2021, allowing online operators to apply for licenses through the Kansspelautoriteit (KSA).
| ● | Recent Developments: |
| ○ | Introduction of enhanced advertising restrictions in 2023 to protect vulnerable players. |
| ○ | Stricter KYC and AML compliance requirements imposed following concerns raised about potential money laundering risks. |
2. United States: State-Level Expansion with Varied Regulatory Approaches
The United States has seen rapid expansion of online gambling following the repeal of PASPA (Professional and Amateur Sports Protection Act) in 2018. However, regulation remains fragmented at the state level, with wide variances between states in terms of permitted wagering activities, enforcement mechanisms and penalties and rate of regulatory change.
a) New Jersey
New Jersey remains the most mature iGaming market in the U.S., with online casinos regulated by the New Jersey Division of Gaming Enforcement (DGE).
| ● | Operators must partner with licensed land-based casinos to offer online services. |
| ● | Strong AML and KYC compliance standards, with regular audits conducted by the DGE. |
32
b) Michigan
Michigan legalized online casinos and sports betting in 2021. The Michigan Gaming Control Board (MGCB) oversees operators.
| ● | Online gambling contributes significantly to state revenue, with a 20-28% tax rate on iGaming revenue. |
| ● | Recent Developments: |
| ○ | Focus on strengthening data privacy measures and enhancing responsible gambling standards. |
c) Pennsylvania
Pennsylvania’s iGaming sector, regulated by the Pennsylvania Gaming Control Board (PGCB), imposes:
| ● | A 54% tax on online slots and a 16% tax on online poker and table games. |
| ● | Stringent compliance checks and responsible gambling standards. |
d) Emerging States
States such as Connecticut, West Virginia, and Delaware have legalized online casinos, while others, including New York and Illinois, are considering legislation to open their markets to iGaming.
3. Canada: Provincial Autonomy with Growing iGaming Adoption
Canada regulates iGaming at the provincial level, allowing provinces to license and regulate online operators.
a) Ontario
Ontario launched a fully regulated iGaming market in April 2022 through iGaming Ontario (iGO), a subsidiary of the Alcohol and Gaming Commission of Ontario (AGCO).
| ● | Ontario allows private operators to enter the market, subject to meeting strict regulatory standards. |
| ● | As of Q1 2025, Ontario’s market has over 70 licensed operators, generating significant tax revenue and offering a competitive market environment. |
b) Other Provinces
| ● | British Columbia (BCLC), Alberta (AGLC), and Quebec (Loto-Québec) continue to operate provincially regulated platforms. |
| ● | There is increasing pressure on other provinces to follow Ontario’s lead by opening their markets to private operators. |
c) Recent Developments
| ● | Discussions are underway regarding a potential federal framework for harmonizing standards across provinces. |
| ● | Concerns about responsible gambling and AML compliance have led to proposed revisions in KYC protocols and player protection measures. |
4. Asia: Rapid Growth Amid Regulatory Uncertainty
Asia remains a high-growth region for iGaming, but regulatory frameworks vary significantly between jurisdictions, ranging from outright illegal, to “grey markets (not legal but tolerated) and fully legal markets.
33
a) Philippines
The Philippines is one of Asia’s most prominent regulated iGaming markets, overseen by the Philippine Amusement and Gaming Corporation (PAGCOR).
| ● | PAGCOR licenses Philippine Offshore Gaming Operators (POGOs), offering services to offshore markets. |
| ● | Regulatory Challenges: |
| ○ | Ongoing crackdowns on illegal operators and increased scrutiny on AML compliance. |
| ○ | POGOs face increasing regulatory pressure and may see stricter oversight in 2025. |
b) Macau
Macau maintains a monopoly on land-based gaming but has historically been resistant to iGaming.
| ● | Recent Developments: |
| ○ | Macau authorities are exploring potential regulatory frameworks to allow limited online gaming in response to declining land-based revenues. |
| ○ | Strict AML compliance measures are likely to be a focal point of any future legislation. |
c) India
India’s regulatory environment is fragmented, with states allowed to regulate or ban online gambling.
| ● | Sikkim and Nagaland have regulated online casinos, but the majority of states have yet to establish a legal framework. |
| ● | Recent Developments: |
| ○ | The Indian government has indicated interest in establishing a national regulatory framework for online gambling, which could unlock significant market potential. |
d) Japan and South Korea
| ● | Japan: Discussions are underway about allowing online casinos to complement the Integrated Resort (IR) developments. However, iGaming remains largely prohibited although there are a number of grey market operators. |
| ● | South Korea: Online gambling is heavily restricted, with only a limited number of domestic platforms allowed to operate. |
Conclusion: Key Trends and Evolving Regulatory Outlook
| 1. | Focus on Responsible Gambling and AML Compliance |
Global regulators are increasingly emphasizing responsible gambling, imposing affordability checks, and enhancing AML frameworks.
| 2. | Increased Regulatory Scrutiny and Licensing Requirements |
Jurisdictions such as the UK, Ontario, and New Jersey are strengthening compliance requirements, leading to higher costs for operators
but improved market integrity.
| 3. | Emerging Jurisdictions and Market Expansion |
As U.S. states and Asian countries explore regulated frameworks, market growth opportunities are increasing, albeit with accompanying
regulatory complexity.
34
| 4. | Rise of Crypto and Blockchain in iGaming |
The integration of cryptocurrencies and blockchain technology presents both regulatory challenges and opportunities, with increased focus
on fraud prevention and enhanced KYC requirements.
| 5. | Convergence of Land-Based and Online Operations |
As more land-based operators expand into online channels, regulators are adapting frameworks to ensure cohesive oversight.
As the iGaming industry continues to grow, a balance between innovation and regulation will be key to maintaining player trust, safeguarding markets, and preventing exploitation.
Life Sciences Government Regulation
We have not held any meetings with, and no applications or requests for approval have been submitted to, the U.S. Food and Drug Administration (“FDA”) for any indications or products under the anti-TNF platform, and do not have any plans to do so at this time.
Employees and Human Capital Management
As of March 25, 2025, we and our subsidiaries had one full-time contractor, (the Chief Executive Officer), one part-time contractor (the Chief Accounting Officer) and one part-time medical research contractor. One of these consultants is located in the U.K., one in Canada, and one in the U.S.
In addition, we employ a limited number of part-time employees on a temporary basis as needed, as well as scientific advisors, consultants and service providers, mainly through academic institutions and contract research organizations.
We have never had a work stoppage and none of our employees are covered by collective bargaining agreements or represented by a labor union. We believe that we have good relationships with our employees.
Our human capital resources objectives include, as applicable, identifying, recruiting, retaining, incentivizing, and integrating our existing and new employees, advisors, and consultants. The principal purposes of our equity and cash incentive plans are to attract, retain and reward personnel through the granting of stock-based and cash-based compensation awards, in order to increase stockholder value and the success of our company by motivating such individuals to perform to the best of their abilities and achieve our objectives.
Corporate History
Formation
We were formed as a blank check company organized under the laws of the State of Delaware on September 7, 2016. We were formed for the purpose of effecting a merger, capital stock exchange, stock purchase, asset acquisition or other similar business combination with one or more operating businesses. Since formation, we focused our efforts on acquiring an operating company in the healthcare and related wellness industry although our efforts in identifying a prospective target business were not limited to a particular industry.
Initial Public Offering
On June 7, 2017, pursuant to our Initial Public Offering (the “IPO”), we sold 11,500,000 Units at a purchase price of $10.00 per Unit, inclusive of 1,500,000 Units sold to the underwriters on June 23, 2017 upon the underwriters’ election to fully exercise their over-allotment option, generating gross proceeds of $115,000,000. Each “Unit” consisted of one-380th of a share of our common stock, one right to receive one-3,800th of one share of our common stock upon the consummation of a business combination (“Right”), and one redeemable warrant to purchase one-seven hundred sixtieth of one share of our common stock (the “Public Warrants”). Each Public Warrant entitles the holder to purchase one- seven hundred sixtieth of one share of common stock at an exercise price of $5.75 per 1/760th of one share ($4,370.00 per whole share), subject to adjustment. No fractional shares will be issued upon exercise of the Public Warrants. The Public Warrants became exercisable 12 months from the closing of the IPO, and expire five years after the completion of the Business Combination (November 6, 2025).
35
We may redeem the Public Warrants, in whole and not in part, at a price of $0.01 per Public Warrant upon 30 days’ notice (“30-day redemption period”), only in the event that the last sale price of the common stock equals or exceeds $6,840.00 per share for any 20 trading days within a 30-trading day period ending on the third trading day prior to the date on which notice of redemption is given, provided there is an effective registration statement with respect to the shares of common stock underlying such Public Warrants and a current prospectus relating to those shares of common stock is available throughout the 30-day redemption period. If we call the Public Warrants for redemption as described above, our management will have the option to require all holders that wish to exercise Public Warrants to do so on a “cashless basis.” In determining whether to require all holders to exercise their Public Warrants on a “cashless basis,” management will consider, among other factors, our cash position, the number of Public Warrants that are outstanding and the dilutive effect on our stockholders of issuing the maximum number of shares of common stock issuable upon the exercise of the Public Warrants. Each holder of a Right received one-3,800th (1/3,800) of one share of common stock upon consummation of the Business Combination. No fractional shares were issued upon exchange of the Rights.
Private Placement
Concurrent with the closing of the IPO, KBL IV Sponsor LLC (the “Sponsor”) and the underwriters purchased an aggregate of 450,000 unregistered Units (“Private Units”) at $10.00 per Unit, generating gross proceeds of $4,500,000 in a private placement. In addition, on June 23, 2017, we consummated the sale of an additional 52,500 Private Units at a price of $10.00 per Unit, which were purchased by the Sponsor and underwriters, generating gross proceeds of $525,000. Of these, 377,500 Private Units were purchased by the Sponsor and 125,000 Private Units were purchased by the underwriters. The proceeds from the Private Units were added to the net proceeds from the IPO held in a Trust Account (the “Trust Account”). The Private Units (including their component securities) were not transferable, assignable or salable until 30 days after the completion of the Business Combination (defined below) and the warrants included in the Private Units (the “Private Placement Warrants”) will be non-redeemable so long as they are held by the Sponsor, the underwriters or their permitted transferees. If the Private Placement Warrants are held by someone other than the Sponsor, the underwriters or their permitted transferees, the Private Placement Warrants will be redeemable by us and exercisable by such holders on the same basis as the warrants included in the Units sold in the IPO. In addition, for as long as the Private Placement Warrants are held by the underwriters or its designees or affiliates, they may not be exercised after five years from the effective date of the registration statement related to the IPO. Otherwise, the Private Placement Warrants have terms and provisions that are identical to those of the warrants sold as part of the Units in the IPO and have no net cash settlement provisions.
Business Combination
On July 25, 2019, we entered into a Business Combination Agreement (as amended from time to time, the “Business Combination Agreement”), with KBL Merger Sub, Inc. (“Merger Sub”), 180 Life Corp. (f/k/a 180 Life Sciences Corp.) (“180”), Katexco Pharmaceuticals Corp. (“Katexco”), CannBioRex Pharmaceuticals Corp. (“CBR Pharma”), 180 Therapeutics L.P. (“180 LP” and together with Katexco and CBR Pharma, the “180 Subsidiaries” and, together with 180 Life Sciences Corp., the “180 Parties”), and Lawrence Pemble, in his capacity as representative of the stockholders of the 180 Parties (the “Stockholder Representative”). The business combination described in the Business Combination Agreement (the “Business Combination”), closed and became effective on November 6, 2020 (the “Combination Closing”). Pursuant to the Business Combination Agreement, among other things, Merger Sub merged with and into 180, with 180 continuing as the surviving entity and a wholly-owned subsidiary of the Company (the “Merger”). In connection with, and prior to, the Combination Closing, 180 Life Sciences Corp. filed a Certificate of Amendment of its Certificate of Incorporation in Delaware to change its name to 180 Life Corp., and our company (which was known as of our entry into the Business Combination as KBL Merger Corp. IV, changed our name to 180 Life Sciences Corp.).
36
180 was incorporated in Delaware on January 28, 2019. Prior to the Combination Closing of the Business Combination, 180 operated through three subsidiaries: 180 LP, a Delaware limited partnership formed on September 6, 2013; Katexco, a company incorporated in British Columbia, Canada on March 7, 2018; and CBR Pharma, a company incorporated in British Columbia, Canada on March 8, 2018.
In July 2019, 180 and each of 180 LP, Katexco and CBR Pharma completed a corporate restructuring, pursuant to which 180 LP, Katexco and CBR Pharma became wholly-owned subsidiaries of 180LS (the “Reorganization”). The corporate restructuring arrangements with respect to Katexco and CBR Pharma were completed under the Business Corporations Act (British Columbia).
On November 6, 2020 (the “Combination Closing Date”), we consummated the Business Combination following a special meeting of stockholders held on November 5, 2020, where the stockholders of the Company considered and approved, among other matters, a proposal to adopt the Business Combination. Pursuant to the Business Combination Agreement, among other things, Merger Sub merged with and into 180, with 180 continuing as the surviving entity and as a wholly-owned subsidiary of the Company. The Merger became effective on November 6, 2020 (such time, the “Effective Time”, and the closing of the Merger being referred to herein as the “Combination Closing”). In connection with, and prior to, the Combination Closing, 180 filed a Certificate of Amendment of its Certificate of Incorporation in Delaware to change its name to 180 Life Corp. and KBL Merger Corp. IV changed its name to 180 Life Sciences Corp.
At the Effective Time, each share of 180 common stock issued and outstanding prior to the Effective Time was automatically converted into the right to receive approximately 0.44310 shares of the common stock, par value $0.0001 per share, of the Company (such shares of common stock issuable to the common stockholders of 180 pursuant to the Business Combination Agreement, the “Merger Consideration Shares”). An aggregate of 46,039 shares of common stock have been issued to date to the common stockholders of 180 as Merger Consideration Shares, including the Escrow Shares (as defined below). Also at the Effective Time, each share underlying the 180 preferred stock issued and outstanding prior to the Effective Time was converted into the right to receive one Class C Special Voting Share of the Company, or one Class K Special Voting Share of the Company, as applicable (such shares, the “Special Voting Shares”). The Special Voting Shares entitled the holder thereof to an aggregate number of votes, on any particular matter, proposition or question, equal to the number of Exchangeable Shares (as defined below) of each of CannBioRex Purchaseco ULC and Katexco Purchaseco ULC, Canadian subsidiaries of 180, respectively, that are outstanding from time to time.
As a result of the Merger, the existing exchangeable shares (collectively, the “Exchangeable Shares”) of CannBioRex Purchaseco ULC and/or Katexco Purchaseco ULC were adjusted in accordance with the share provisions in the articles of CannBioRex Purchaseco ULC or Katexco Purchaseco ULC, as applicable, governing the Exchangeable Shares such that they were multiplied by the exchange ratio for the Merger and became exchangeable into shares of common stock. The Exchangeable Shares entitled the holders to dividends and other rights that are substantially economically equivalent to those of holders of common stock, and holders of Exchangeable Shares have the right to vote at meetings of the stockholders of the Company. All of the Exchangeable Shares have subsequently been converted into common stock of the Company and no Exchangeable Shares are currently outstanding.
Pursuant to the Business Combination Agreement, 2,764 of the Merger Consideration Shares (such shares, the “Escrow Shares”) were deposited into an escrow account (the “Escrow Account”) to serve as security for, and the exclusive source of payment of, our indemnity rights under the Business Combination Agreement, all of which were planned to be released to the same stockholders 12 months following the Combination Closing of the Business Combination, but for a claim made by Dr. Krauss against these shares which is pending.
37
As a result of the Business Combination, the former stockholders of 180 became the controlling stockholders of the Company and 180 became a wholly-owned subsidiary of the Company. The Business Combination was accounted for as a reverse merger, whereby 180 is considered the acquirer for accounting and financial reporting purposes.
In connection with the Combination Closing, we withdrew $9,006,493 of funds from the Trust Account (as defined below) to fund the redemptions of 2,149 shares.
Corporate Structure
The chart below shows our current organizational structure. Note that the Company is currently evaluating and deactivating some inactive subsidiaries:
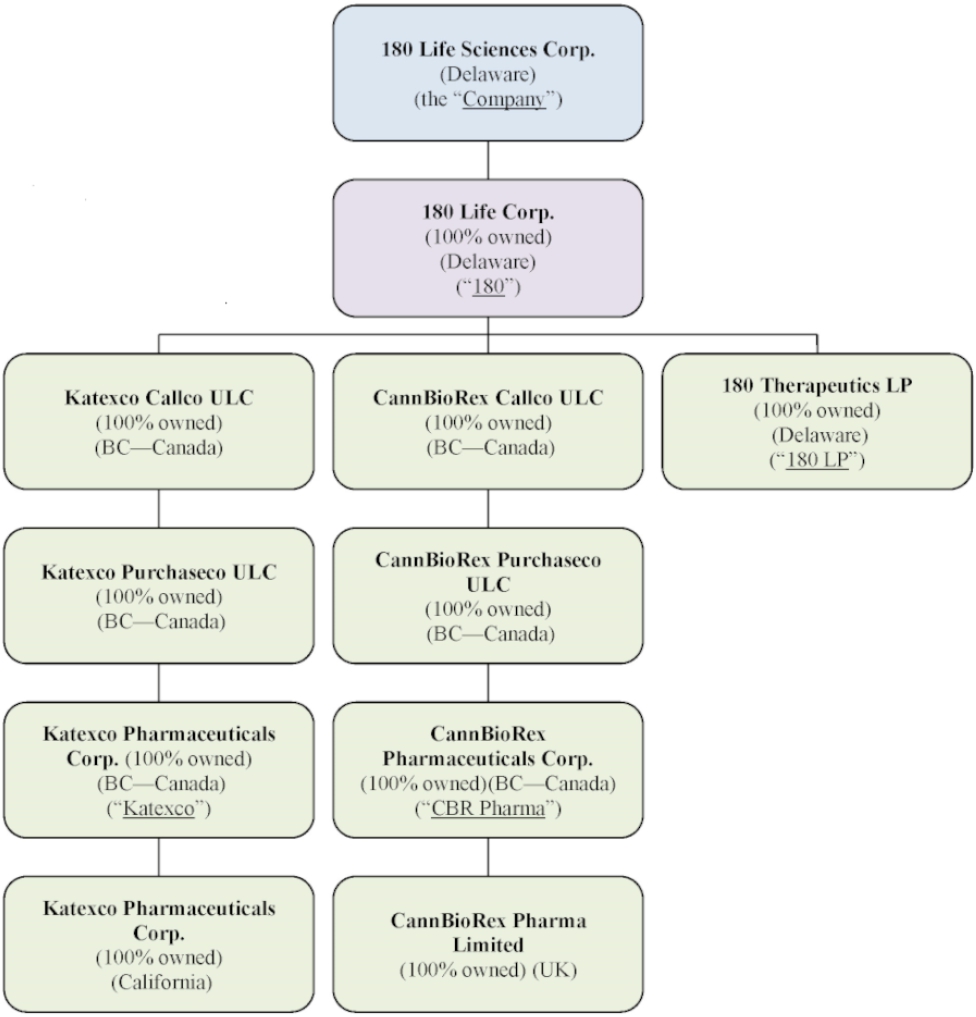
About Us
Our principal executive offices are located at 3000 El Camino Real, Bldg. 4, Suite 200, Palo Alto, CA 94306, and our telephone number is (650) 285-2387. We maintain a website at www.180lifesciences.com. We have not incorporated by reference into this Report the information in, or that can be accessed through, our website, and you should not consider it to be a part of this Report.
38
Item 1A. Risk Factors.
Summary Risk Factors
We face risks and uncertainties related to our business, many of which are beyond our control. In particular, risks associated with our business include:
Risks Relating to Successfully Monetizing Existing Biotech IP
As we are no longer developing or conducting studies regarding our SCA asset and ATNF IP, we may not be able to successfully monetize such assets or intellectual property.
Risks Relating to Our Need For Additional Funding
| ● | Our current cash balance is only expected to be sufficient to fund our planned business operations until approximately December 2025 and we need additional capital which may not be available on commercially acceptable terms, if at all, or may cause dilution, which raises questions about our ability to continue as a going concern. Our accounts payable are significant, and we do not currently have sufficient funds to pay such accounts payable, all of which are past due. |
| ● | We may not receive any additional amounts under our pre-merger directors’ and officers’ insurance policy in connection with certain litigation matters and may be forced to repay $2.57 million previously received. |
Risks Relating to Our Planned Online Casino Operations
| ● | We have no operating history in the gaming industry. We may never become profitable or, if achieved, be able to sustain profitability. The online gaming industry is highly competitive, and competition within the global entertainment and gaming industries is intense, if the Company fails to compete effectively, it could experience price reductions, reduced margins or loss of revenues. |
| ● | The Company will face the risk of fraud, theft, and cheating; we will be reliant on third-party gaming content for our games; we will rely on third party cloud service, electricity and infrastructure, providers, and such providers or services may encounter technical problems and service interruptions. The Company will face cyber security risks that could result in damage to the Company’s reputation and/or subject them to fines, payment of damages, lawsuits and restrictions on the Company’s use of data. |
| ● | The product offerings of the Company must be approved in most regulated jurisdictions in which they are offered. Legislative and regulatory changes could negatively affect the business of the Company and the business of its customers. |
| ● | A reduction in discretionary consumer spending, from an economic downturn or disruption of financial markets or other factors, could negatively impact the financial performance of the Company as it develops its iGaming business. |
39
Risks Relating to Our Plans to Allow Players to Deposit and Withdraw Cryptocurrency
| ● | Bitcoin, Ethereum, Litecoin and other digital assets are novel assets, and are subject to significant legal, commercial, regulatory and technical uncertainty. We may be subject to regulatory developments related to crypto assets and crypto asset markets, which could adversely affect our business, financial condition, and results of operations. |
| ● | Our digital asset holdings are expected to be less liquid and more volatile than our existing cash and cash equivalents and may not be able to serve as a source of liquidity for us to the same extent as cash and cash equivalents. The characteristics of crypto assets have been, and may in the future continue to be, exploited to facilitate illegal activity such as fraud, money laundering, tax evasion and ransomware scams; all of which may have an adverse effect on the market for, and regulation of, crypto assets, and our operations. |
| ● | Incorrect or fraudulent digital asset transactions may be irreversible. The decentralized nature of crypto asset systems may lead to slow or inadequate responses to crises, which may negatively affect our business. |
Risks Related to Our Business Operations
| ● | Our results of operations may be adversely affected by fluctuations in currency values, inflation, and/or economic uncertainty. |
| ● | We depend on our key personnel and our ability to attract and retain employees. Our officers and directors may have conflicts of interest. |
| ● | We may enter into strategic transactions in the future which may result in a material change in our operations and/or a change of control. |
Risks Relating to Our Life Sciences Operations
| ● | We may be unable to monetize our existing life science assets. |
| ● | Our License Agreements with the University of Oxford and other licensors may be terminated in certain circumstances without our consent. |
Risks Relating to Our Ineffective Disclosure Controls and Procedures
| ● | We have in the past, and may in the future, identify material weaknesses in our disclosure controls and procedures and internal control over financial reporting. |
Risks Related to our Intellectual Property
| ● | We may not be able to adequately protect our future product candidates or our proprietary technology in the marketplace. |
Risks Related to our Securities
| ● | We currently have an illiquid and volatile market for our common stock, and the market for our common stock is and may remain illiquid and volatile in the future. Elray Resources, Inc., beneficially owns a significant percentage of our voting stock and as such exercises significant voting control over us, which limits other stockholders’ abilities to influence corporate matters and could delay or prevent a change in corporate control. |
| ● | The exercise of the outstanding options and warrants, and the sale of common stock upon exercise thereof, may adversely affect the trading price of our securities. |
40
General Risk Factors
| ● | Global economic conditions could materially adversely affect our business, results of operations, financial condition and growth. |
| ● | Our proprietary information, or that of our customers, suppliers and business partners, may be lost or we may suffer security breaches. Failure of our information technology systems, including cybersecurity attacks or other data security incidents, could significantly disrupt the operation of our business. |
| ● | If we make any acquisitions, they may disrupt or have a negative impact on our business. |
| ● | We also face other risks and uncertainties, including those described below. |
You should be aware that there are substantial risks for an investment in our common stock. You should carefully consider these risk factors before you decide to invest in our common stock.
If any of the following risks were to occur, our business, financial condition, results of operations or other prospects, could be materially adversely affected, and the occurrence of any of these risks could materially affect our likelihood of success. If that happens, the market price of our common stock, if any, could decline, and prospective investors would lose all or part of their investment in our common stock.
Our business, financial condition and results of operations are subject to various risks and uncertainties, including those described below. This section discusses factors that, individually or in the aggregate, could cause our actual results to differ materially from expected and historical results. Our business, financial condition or results of operations could be materially adversely affected by any of these risks. It is not possible to predict or identify all such factors. Consequently, the following description of Risk Factors is not a complete discussion of all potential risks or uncertainties applicable to our business.
Risks Relating to Our Need For Additional Funding
Our current cash balance is only expected to be sufficient to fund our planned business operations until approximately December 2025. If additional capital is not available, we may not be able to pursue our planned business operations, may be forced to change our planned business operations, or may take other actions that could adversely impact our stockholders, including seeking bankruptcy protection.
We are a clinical stage biotechnology company that is transitioning into the iGaming industry via the acquisition of the Gaming Technology Platform, which platform currently has no revenue. Thus, our business does not generate the cash necessary to finance our planned business operations. We will require significant additional capital to: (i) protect our intellectual property; (ii) attract and retain highly-qualified personnel; (iii) respond effectively to competitive pressures; and (iv) acquire complementary businesses or technologies.
Our future capital needs depend on many factors, including: (i) the scope, duration and expenditures associated with our research, development and commercialization efforts as they relate to iGaming; (ii) the outcome of potential partnering or licensing transactions, if any; (iii) competing technological developments with regards to our iGaming platform; and (iv) protecting and supporting our proprietary patent positions.
41
We will need to raise substantial additional funds through public or private equity offerings, debt financings or strategic alliances and licensing arrangements to finance our planned business operations. We may not be able to obtain additional financing on terms favorable to us, if at all. General market conditions, rising interest rates and inflation, as well as global conflicts such as the ongoing conflict between Ukraine and Russia, and Israel and Hamas, may make it difficult for us to seek financing from the capital markets, and the terms of any financing may adversely affect the holdings or the rights of our stockholders. For example, if we raise additional funds by issuing equity securities, further dilution to our stockholders will result, which may substantially dilute the value of their investment. Any equity financing may also have the effect of reducing the conversion or exercise price of our outstanding convertible or exercisable securities, which could result in the issuance (or potential issuance) of a significant number of additional shares of our common stock. In addition, as a condition to providing additional funds to us, future investors may demand, and may be granted, rights superior to those of existing stockholders. Debt financing, if available, may involve restrictive covenants that could limit our flexibility to conduct future business activities and, in the event of insolvency, could be paid before holders of equity securities received any distribution of our assets. We may be required to relinquish rights to our technologies or product candidates, or grant licenses through alliance, joint venture or agreements on terms that are not favorable to us, in order to raise additional funds. Our current cash balance is only expected to be sufficient to fund our planned business operations until approximately December 2025. If adequate funds are not available, we may have to delay, reduce or eliminate one or more of our planned activities with respect to our business, or terminate our operations, or may be forced to seek bankruptcy protection. These actions would likely reduce the market price of our common stock.
We will need additional capital which may not be available on commercially acceptable terms, if at all, which raises questions about our ability to continue as a going concern.
As of December 31, 2024, we had an accumulated deficit of $141,523,344 and a working capital deficit of $1,636,486, and for the year ended December 31, 2024, a net loss of $6,168,177 and cash used in operating activities for the year ended December 31, 2024, of $1,480,567. As of March 24, 2025, we had cash on hand of approximately $3.8 million. The Company expects to invest a significant amount of capital to commercialize its iGaming assets and fund research and development. On September 29, 2024, the Company acquired certain source code and intellectual property relating to an online blockchain casino and plans to build and launch a fully operational casino operation as part of its future operations. The Company expects to invest a significant amount of capital to fund the development and operation of this business. As a result, the Company expects that its operating expenses will increase significantly, and consequently will require significant revenues to become profitable. Even if the Company does become profitable, it may not be able to sustain or increase profitability on a quarterly or annual basis. The Company cannot predict when, if ever, it will be profitable. As of March 24, 2025, we had cash on hand of approximately $3.8 million, including certain funds with certain restrictions on use (including, restrictions on repaying existing indebtedness) which we expect will last us until December 2025. Our Consolidated Financial Statements included herein have been prepared assuming we will continue as a going concern. As we are not generating revenues, we need to raise a significant amount of capital in order to pay our debts and cover our operating costs. While we have raised funds in the past through debt and the sale of equity, there is no assurance that we will be able to raise additional needed capital or that such capital will be available under favorable terms.
We are subject to all the substantial risks inherent in the development of a new business enterprise within an extremely competitive industry. Due to the absence of a long-standing operating history and the emerging nature of the markets in which we compete, we anticipate operating losses until we can successfully implement our business strategy, which includes all associated revenue streams. We may never achieve profitable operations or generate significant revenues.
We currently have a monthly cash requirement of approximately $252,000. We believe that in the aggregate, we will require significant additional capital funding to support and expand our iGaming assets, the research and development and marketing of our products, fund future clinical trials, repay debt obligations, provide capital expenditures for additional equipment and development costs, payment obligations, office space and systems for managing the business, and cover other operating costs until our planned revenue streams from products are fully-implemented and begin to offset our operating costs, if ever.
Since our inception, we have funded our operations with the proceeds from equity and debt financing. We have experienced liquidity issues due to, among other reasons, our limited ability to raise adequate capital on acceptable terms. We have historically relied upon the sale of equity and debt funding that is convertible into shares of our common stock to fund our operations and have devoted significant efforts to reduce that exposure. We anticipate that we will need to issue equity to fund our operations and fund our operating expenses for the foreseeable future. If we are unable to achieve operational profitability or we are not successful in securing other forms of financing, we will have to evaluate alternative actions to reduce our operating expenses and conserve cash.
42
These conditions raise substantial doubt about our ability to continue as a going concern. The Consolidated Financial Statements included herein have been prepared in accordance with accounting principles generally accepted in the United States on a going concern basis, which contemplates the realization of assets and the satisfaction of liabilities in the normal course of business. Accordingly, the Consolidated Financial Statements included herein do not include any adjustments relating to the recoverability of assets and classification of liabilities that might be necessary should we be unable to continue as a going concern. The Consolidated Financial Statements included herein also include a going concern footnote.
Additionally, wherever possible, the Board of Directors will attempt to use non-cash consideration to satisfy obligations. In many instances, we believe that the non-cash consideration will consist of restricted shares of our common stock, preferred stock or warrants to purchase shares of our common stock. The Board of Directors has authority, without action or vote of the stockholders, but subject to Nasdaq rules and regulations (which generally require stockholder approval for any transactions which would result in the issuance of more than 20% of our then outstanding shares of common stock or voting rights representing over 20% of our then outstanding shares of stock, subject to certain exceptions), to issue all or part of the authorized but unissued shares of common stock, preferred stock or warrants to purchase such shares of common stock. In addition, we may attempt to raise capital by selling shares of our common stock, possibly at a discount to market in the future. These actions will result in dilution of the ownership interests of existing stockholders, may further dilute common stock book value, and that dilution may be material. Such issuances may also serve to enhance existing management’s ability to maintain control of us, because the shares may be issued to parties or entities committed to supporting existing management.
We may not receive any additional amounts under our pre-merger directors’ and officers’ insurance policy in connection with certain litigation matters and may be forced to repay $2.57 million previously received.
On June 29, 2022, AmTrust International Underwriters DAC (“AmTrust”), which was the premerger directors’ and officers’ insurance policy underwriter for KBL, filed a declaratory relief action against the Company in the U.S. District Court for the Northern District of California (the “Declaratory Relief Action”) seeking a declaration that AmTrust is not obligated to reimburse the Company for fees advanced by the Company to Dr. Marlene Krauss and George Hornig, former officer and directors of the Company, under the directors’ and officers’ insurance policy.
On September 20, 2022, the Company filed its Answer and Counterclaims against AmTrust for bad faith breach of AmTrust’s insurance coverage obligations to the Company under the subject insurance policy, seeking at least $2 million in compensatory damages, and punitive damages. In addition, the Company brought a Third-Party Complaint against its excess insurance carrier, Freedom Specialty Insurance Company (“Freedom”) seeking declaratory relief that Freedom will also be required to honor its policy coverage as soon as the amount of AmTrust’s insurance coverage obligations to the Company has been exhausted. On October 25, 2022, AmTrust filed its Answer to the Company’s Counterclaims and, on October 27, 2022, Freedom filed its Answer to the Third-Party Complaint.
On November 22, 2022, the Company filed a Motion for Summary Adjudication against both AmTrust and Freedom. The Motion was fully briefed and a hearing was held on March 9, 2023. The standard to prevail on a Motion for Summary Adjudication in the Court is high to prevail and requires a judge to find that there are no disputed issues of fact so that they can rule on the issues as a matter of law. In this instance the judge found three major issues could be decided as a matter of law in the Company’s favor and that one issue, the Change in Control exclusion, requires further discovery.
43
On April 21, 2023, the Court issued an Order Granting in Part and Denying in Part the Company’s Motion for Partial Summary Judgment.
Specifically, the Court granted summary adjudication in favor of the Company on the following issues: (a) that the Company is, in fact, an insured under both the AmTrust and Freedom insurance policies; (b) that certain SEC subpoena related expenses for defendants Dr. Marlene Krauss, the Company’s former Chief Executive Officer and Director, and George Hornig, the former Chairman of the Board, are within the basic scope of coverage under both the AmTrust and Freedom insurance policies; and (c) that the Insured vs. Insured exclusion relied upon by AmTrust and Freedom is not applicable to bar any such coverage.
The Court also found that there were issues of disputed facts as to the Change in Control exclusion contained within the policies, which therefore precluded the Court from granting the remainder of the Company’s requests for summary adjudication as a matter of law. Accordingly, the Court, at that time, denied the Company’s further requests for summary adjudication and deemed that for the time being, the Change in Control issue is to be determined at the time of trial, in order to find that the policies (i) provide coverage for the fees which the Company has advanced and will advance to Dr. Marlene Krauss and George Hornig; (ii) that AmTrust has breached the policy; (iii) that AmTrust must pay such expenses of the Company; and that, once the AmTrust policy has been exhausted, (iv) Freedom will be obligated to pay such expenses of the Company pursuant to its policy.
On August 4, 2023, the Court granted the Company’s request to file a second motion for partial summary judgment in the case, this one being on the issue of whether AmTrust should be required to advance to the Company the defense costs being incurred by Dr. Marlene Krauss and George Hornig during the pendency of the case. The Motion for Partial Summary Judgment was fully briefed by the parties, and a hearing for such Motion was held on January 11, 2024. After the matter was taken under submission, on February 12, 2024, the Court granted the Company’s Motion for Partial Summary Judgment against both AmTrust and Freedom, and ordered as follows: (a) AmTrust is obligated under its insurance policy with the Company to advance to the Company all defense costs in excess of the deductible that the Company has advanced, or will advance, to Dr. Krauss and Mr. Hornig in connection with certain SEC Subpoenas, and (b) upon exhaustion of the AmTrust insurance policy, Freedom is obligated to do the same pursuant to its excess liability insurance policy with the Company. This Order applies throughout the interim of the case, but does not constitute a final judgment, and both the Company and the two insurers retain their rights to contest all applicable issues at trial.
On April 16, 2024, AmTrust paid the Company $2.27 million in reimbursement of fees which the Company has advanced to Dr. Marlene Krauss and George Hornig, of which the Company received $1.5 million after the payment of attorney’s fees. On May 9, 2024, AmTrust paid the Company $300,140 in reimbursement of fees which the Company had advanced to Dr. Marlene Krauss and George Hornig, and the Company received $200,093 after the payment of attorney’s fees.
The Company, Freedom and Amtrust held a mediation conference on August 21, 2024, during which, the Company agreed to the terms of a settlement with Freedom, pursuant to which Freedom agreed to pay $125,000 to the Company, of which the Company received approximately $83,333 after the payment of legal fees. On September 23, 2024, Freedom paid the Company a further $125,000 in reimbursement of fees advanced by the Company, of which the Company received $76,639 after the payment of attorney’s fees. On September 5, 2024, the Company and Freedom executed a mutual release agreement. The total of $1,789,443 is included in other income on the Company’s unaudited consolidated statements of operations and comprehensive income.
A trial on these matters is currently scheduled for May 12, 2025 and the Company and AmTrust are scheduling mediation on February 27, 2025. A final judgment following trial could potentially confirm these obligations of the insurers or, alternatively, reverse and require the Company to repay all or portions of such advance payments, including up to and including the $2.57 million advanced by AmTrust. There is no assurance at this time as to what the final judgment may entail, or whether settlement efforts will lead to a final conclusion which is acceptable to the Company or AmTrust.
44
While the Company continues to believe it has a strong case against AmTrust, there can be no assurance that the Company will prevail in this action. The final outcome of the litigation is unknown at this time and such final outcome could be materially adverse to the Company. In the event the Company were required to repay the $2.57 million advanced by AmTrust, it could materially adversely effect the Company’s ability to pay its expenses and its cash flows, significantly decrease the amount of funding available to support the Company’s business operations, force us to raise additional capital, which may be dilutive to existing stockholders, or force us to seek bankruptcy protection.
Additional funding may not be available on favorable terms, if at all, causing dilution to our stockholders, restricting our operations or adversely affecting our ability to operate our business.
We may not be able to obtain additional financing on terms favorable to us, if at all, including as a result of macroeconomic conditions such as a severe or prolonged economic downturn. Disruption, uncertainty or volatility in the capital markets could increase our cost of capital or limit our ability to raise funds needed to operate our business. Disruptions could be caused by Federal Reserve policies and actions, currency concerns, inflation, economic downturn or uncertainty, monetary policies, failures of financial institutions, U.S. debt management concerns, tariffs, interest rates, and U.S. debt limit and budget disputes, including government shutdowns, European and worldwide sovereign debt concerns, other global or geopolitical events, or other factors. Current macroeconomic conditions have negatively impacted the U.S. banking sector, including for example, the recent closures and FDIC receiverships of Silicon Valley Bank and Signature Bank. Although we do not have any accounts at or business relationships with these banks, we may be negatively impacted by other disruptions to the U.S. banking system caused by these or similar developments.
Our accounts payable are significant, and we do not currently have sufficient funds to pay such accounts payable, all of which are past due.
We do not currently have adequate cash on hand, and do not expect to generate revenues, sufficient to pay down our significant accounts payable balance, which is currently approximately $2.6 million, all of which is past due as of the date of this Report. While we are actively working with creditors to attempt to satisfy amounts owed in shares of common stock, the issuance of which may cause dilution to existing stockholders, there can be no assurance that such creditors will agree to accept equity in lieu of cash. Such creditors may in the future exercise remedies, including suing the Company for nonpayment or attempting to force the Company into receivership. Creditor litigation may be costly and resource intensive, which could deplete our already limited available cash and/or force us to raise additional funding, which may be dilutive to existing stockholders or seek bankruptcy protection. In the event of a bankruptcy proceeding or insolvency, or restructuring of our capital structure, holders of the Company’s common stock could suffer a total loss of their investment.
Risks Relating to Our Planned Online Casino Operations
We have no operating history in the gaming industry and have incurred significant operating losses since inception. We may never become profitable or, if achieved, be able to sustain profitability.
We have no operating history in the gaming industry upon which to base any assumption as to the likelihood that our gaming operations will prove successful, and we may never achieve profitable operations. We currently expect to incur net losses for the foreseeable future. Even if we do achieve profitability, there can be no guarantee that we will be able to sustain profitability. As a result of the acquisition of certain blockchain casino intellectual property, we plan to focus a portion of our attention on the online gaming industry, and we have limited experience in that industry. If we are unsuccessful in operating our business, it will have a material adverse impact on our business, financial condition and results of operations.
45
The online gaming industry is highly competitive, and if the Company fails to compete effectively, it could experience price reductions, reduced margins or loss of revenues.
The online gaming industry is highly competitive. A number of companies offer products and services that are similar to the Company’s planned online casino. The majority of the Company’s current and potential competitors have longer operating histories, significantly greater financial, technical and marketing resources, greater name recognition, broader or more integrated product offerings, larger technical staffs and a larger installed customer base. These competitors may be able to respond more quickly to new or emerging technologies and changes in customer requirements, develop superior products, and devote greater resources to the development, promotion and sale of online gaming operations than the Company can.
Because of the rapid growth of the gaming industry, and the relatively low capital barriers to entry in the software industry, the Company expects additional competition from other established and emerging companies. Additionally, the Company’s competitors could combine or merge to become more formidable competitors or may adapt more quickly than we can to new technologies, evolving industry trends and changing customer requirements.
Competition within the global entertainment and gaming industries is intense and the future offerings of the Company may not be able to compete against other competing forms of entertainment such as television, movies and sporting events, as well as other entertainment and gaming options on the Internet. If the Company’s offerings are not popular, the Company’s business could be harmed.
The Company plans to begin operating in the global gaming industry and is targeting the second quarter of 2025. The users of the Company’s planned online casino offering will face a vast array of entertainment choices. Other forms of entertainment, such as television, movies, sporting events and in-person casinos, are more well established and may be perceived by users to offer greater variety, affordability, interactivity and enjoyment. The Company’s planned products and services will compete with these other forms of entertainment for the discretionary time and income of end users. If we are unable to sustain sufficient interest in the Company’s planned products, services and offerings in comparison to other forms of entertainment, including new forms of entertainment, the Company’s business model may not be viable.
The Company will face the risk of fraud, theft, and cheating.
The Company will face the risk that third-parties, employees or consultants may attempt or commit fraud or theft or cheat using the Company’s products. Such risks include backdoors, nefarious code and other efforts. Failure to discover such acts or schemes in a timely manner could result in losses in the Company’s operations and those of the Company’s future customers. Negative publicity related to such acts or schemes could have an adverse effect on the Company’s reputation, potentially causing a material adverse effect on the Company’s business.
We will be reliant on third-party gaming content for our games.
We expect to be reliant on third-parties for our gaming content and to have no control over the providers of our content. Our business could be adversely affected if our access to games is limited or delayed.
We expect that in the future our access to gaming content will be controlled by one or a small number of entities, who, if they work together, could materially limit our access to games and other content and available providers may not choose to license games to us. Our business may be adversely affected if our access to games is limited or delayed because of deterioration in our relationships with one or more of these providers or if they choose not to license to us for any other reason.
Even if we are able to secure rights to gaming content from providers or creators, external groups may object and may exert pressure on third parties to discontinue licensing rights to us, hold back content from us, or increase content fees. Content providers also may attempt to take advantage of their market power to demand onerous financial terms from us. If any of these content providers were to not enter into contracts with us, or renew contracts at their expiration, fail to meet their contractual obligations, or cease operations for any reason, and if no suitable alternative providers were available, we could be unable to operate our planned gaming platform. Our inability to retain such third-party providers or find suitable alternate providers in a timely manner could lead to significant costs and disruptions that could prevent us from generating revenues, reduce our future revenue, harm our business reputation, and have a material adverse effect on our financial condition and results of operations.
46
The cost of gaming content may be material and may be greater than our revenues, resulting in future net losses.
To the extent that we are unable to license a large amount of content or the content of certain popular games, our business, operating results, and financial condition could be materially harmed.
The Company will rely on third party cloud services and such providers or services may encounter technical problems and service interruptions.
The Company expects to host its future services on a combination of proprietary and cloud servers. Such servers have in the past and may in the future experience slower response times or interruptions as a result of increased traffic or other reasons. The Company will not have control over the operations of the facilities or infrastructure of the third-party service providers that they use. Such third parties’ facilities are vulnerable to damage or interruption from natural disasters, cybersecurity attacks, terrorist attacks, power outages and similar events or acts of misconduct. We expect that the continuing and uninterrupted performance of the Company’s platform will be critical to our success. The Company may experience interruptions, delays and outages in service and availability from these third-party service providers from time to time due to a variety of factors, including infrastructure changes, human or software errors, website hosting disruptions and capacity constraints. In addition, any changes in these third parties’ service levels may adversely affect our ability to meet the requirements of the Company’s users. Since our platform’s continuing and uninterrupted performance is expected to be critical to our success, sustained or repeated system failures would reduce the attractiveness of our offerings. It may become increasingly difficult to maintain and improve our performance, especially during peak usage times, as the Company expands, and the usage of our offerings increases. Any negative publicity arising from these disruptions could harm our reputation and brand and may adversely affect the usage of our offerings. Any of the above circumstances or events may harm our reputation, reduce the availability or usage of our platform, lead to a significant loss of revenue, increase our costs, and impair our ability to attract new customers, any of which could adversely affect our business, financial condition, and results of operations.
Malfunctions of third-party communications infrastructure, hardware and software may expose the Company to a variety of risks it cannot control.
The business of the Company is expected to depend upon the capacity, reliability and security of the infrastructure owned by third parties over which the Company’s offerings are deployed. The Company has no control over the operation, quality, or maintenance of a significant portion of that infrastructure or whether or not those third parties will upgrade or improve their equipment. The Company instead will depend on these companies to maintain the operational integrity of their connections. If one or more of these companies is unable or unwilling to supply or expand their levels of service in the future, the operations of the Company could be adversely impacted. Also, to the extent the number of users of networks utilizing the Company’s future products and services suddenly increases, the technology platform and secure hosting services which will be required to accommodate a higher volume of traffic may result in slower response times or service interruptions. System interruptions or increases in response time could result in a loss of potential or existing users and, if sustained or repeated, could reduce the appeal of the networks to users. In addition, users depend on real-time communications; outages caused by increased traffic could result in delays and system failures. These types of occurrences could cause users to perceive that the products and services of the Company do not function properly and could therefore adversely affect the Company’s ability to attract and retain licensees, strategic partners, and customers.
The Company’s planned online casino is part of a new and evolving industry, which presents significant uncertainty and business risks.
The gaming platforms, systems and gaming content industries are relatively new and continue to evolve. Whether these industries grow and whether their business will ultimately succeed, will be affected by, among other things, mobile platforms, legal and regulatory developments (such as passing new laws or regulations or extending existing laws or regulations to online gaming and related activities), taxation of gaming activities, data and information privacy and payment processing laws and regulations, and other factors that are unable to be predicted and which are beyond the control of the Company.
47
Given the dynamic evolution of these industries, it can be difficult to plan strategically, including as it relates to product launches in new or existing jurisdictions which may be delayed or denied, and it is possible that competitors will be more successful than the Company is at adapting to change and pursuing business opportunities. Additionally, as the online gaming industry advances, including with respect to regulation in new and existing jurisdictions, the Company may become subject to additional compliance-related costs, including regulatory infractions, licensing, and taxes. If our product offerings do not obtain popularity or maintain popularity, or if we fail to grow in a manner that meets our expectations, or if we cannot offer product offerings in particular jurisdictions that may be material to our business, then our results of operations and financial condition could be harmed.
Additionally, possible future changes in governmental regulations pose material risks to the Company. These changes may include amendments to existing rules or the introduction of new ones, shifts in regulatory focus or policy, or changes in the enforcement or interpretation of current rules and policies. These could lead to increased compliance costs, restrictions or prohibitions on current operations, or required alterations to the way the Company’s then services are offered or marketed, any of which may result in a material adverse effect on the results of operations and financial condition of the Company.
Failure to comply with regulatory requirements in a particular jurisdiction, or the failure to successfully obtain a license or permit applied for in a particular jurisdiction, could impact the ability of the Company to comply with licensing and regulatory requirements in other jurisdictions, or could cause the rejection of license applications or cancellation of existing licenses in other jurisdictions.
Compliance with the various regulations applicable to online gaming is costly and time-consuming. Regulatory authorities at the federal, state and local levels (both in the U.S. and in foreign jurisdictions) have broad powers with respect to the regulation and licensing of real money online gaming operations and may revoke, suspend, condition or limit the licenses of the Company, impose substantial fines on us, and take other actions, any one of which could have a material adverse effect on our business, financial condition, results of operations and prospects. We do not currently hold any licenses and will need to obtain licenses in the future to operate our planned online blockchain casino. Such licenses may not be available in the timeframe we expect, may be more costly, or may require us to undertake various resource intensive actions to obtain. These laws and regulations are dynamic and subject to potentially differing interpretations, and various legislative and regulatory bodies may expand current laws or regulations or enact new laws and regulations regarding these matters. The Company strives to comply with all applicable laws and regulations relating to its business. It is possible, however, that these requirements may be interpreted and applied in a manner that is inconsistent from one jurisdiction to another and may conflict with other rules. Non-compliance with any such law or regulations could expose the Company to claims, proceedings, litigation and investigations by private parties and regulatory authorities, as well as substantial fines and negative publicity, each of which may materially and adversely affect the business of the Company.
The future gaming licenses of the Company could be revoked, suspended or conditioned at any time. The loss of a license in one jurisdiction could trigger the loss of a license or affect our eligibility for such a license in another jurisdiction, and any of such losses, or potential for such loss, could cause the Company to cease offering some or all of its offerings in the impacted jurisdictions. The Company may be unable to obtain or maintain all necessary registrations, licenses, permits or approvals, and could incur fines or experience delays related to the licensing process, which could adversely affect its operations. The delay or failure to obtain or maintain licenses by the Company in any jurisdiction may prevent it from distributing its offerings, increasing its customer base and/or generating revenues. The Company may not be able to obtain and maintain the licenses and related approvals necessary to conduct its operations. Any failure by the Company or its customers to maintain or renew existing licenses, registrations, permits or approvals could have a material adverse effect on the business, financial condition, results of operations and prospects of the Company.
48
The product offerings of the Company must be approved in most regulated jurisdictions in which they are offered; this process cannot be assured or guaranteed.
If the Company fails to obtain necessary gaming licenses in a given jurisdiction, we would likely be prohibited from distributing and providing our product offerings in that particular jurisdiction. If we fail to seek, do not receive, or receive a suspension or revocation of a license in a particular jurisdiction for our product offerings (including any related technology and software) then we cannot offer the same in that jurisdiction and our gaming licenses in other jurisdictions may be impacted. Furthermore, some jurisdictions require license holders to obtain government approval before engaging in some transactions. We may not be able to obtain all necessary licenses in a timely manner, or at all. Delays in regulatory approvals or failure to obtain such approvals may also serve as a barrier to entry to the market for our product offerings. If the Company is unable to overcome the barriers to entry, it will materially affect our results of operations and future prospects.
To the extent new online gaming jurisdictions are established or expanded, the Company cannot guarantee it will be successful in penetrating such new jurisdictions or expanding its business or customer base in line with the growth of existing jurisdictions. As the Company directly or indirectly enters into new markets, it may encounter legal, regulatory and political challenges that are difficult or impossible to foresee and which could result in an unforeseen adverse impact on planned revenues or costs associated with the new market opportunity. If the Company is unable to effectively develop and operate directly or indirectly within these new markets or if its competitors are able to successfully penetrate geographic markets that it cannot access or where it faces other restrictions, then the Company’s business, operating results and financial condition could be impaired. The failure of the Company to obtain or maintain the necessary regulatory approvals in jurisdictions, whether individually or collectively, would have a material adverse effect on its business.
Legislative and regulatory changes could negatively affect the business of the Company and the business of its customers.
Legislative and regulatory changes may affect demand for or place limitations on the Company’s future operations. Such changes could affect the Company in a variety of ways. Legislation or regulation may introduce limitations on their products or opportunities for the use of our products and could foster competitive products. Our business will likely also suffer if our products become obsolete due to changes in laws or the regulatory framework. Moreover, legislation to prohibit, limit or add burdens to our business may be introduced in the future in jurisdictions where gaming has been legalized. In addition, from time to time, legislators and special interest groups have proposed legislation that would expand, restrict or prevent gaming operations or which may otherwise adversely impact our operations in the jurisdictions in which we then operate and will operate in the future.
Legislative or regulatory changes negatively impacting the gaming industry as a whole could also decrease the demand for our future products. Opposition to gaming could result in restrictions or even prohibitions of gaming operations in any jurisdiction or could result in increased taxes on gaming revenues. Tax matters, including changes in state, federal or other tax legislation or assessments by tax authorities could have a negative impact on our business. A reduction in growth of the gaming industry or in the number of gaming jurisdictions or delays in the opening of new or expanded casinos could reduce demand for our products. Changes in current or future laws or regulations or future judicial intervention in any particular jurisdiction may have a material adverse effect on our existing and proposed foreign and domestic operations. Any such adverse change in the legislative or regulatory environment could have a material adverse effect on our business, results of operations or financial condition.
49
The gaming industry is highly regulated, and the Company must adhere to various regulations and maintain applicable licenses to operate. Failure to abide by regulations or maintain applicable licenses could be disruptive to our business and could adversely affect our operations.
The Company and its planned products are, and will be, subject to extensive regulation under federal, state, local and foreign laws, rules and regulations of the jurisdictions in which we do business and our planned products are used. Such entities currently block direct access to wagering on websites from jurisdictions in which they do not have a license to operate through IP address filtering. Individuals are required to enter their age upon gaining access to their platforms and any misrepresentation of such users age will result in the forfeiting of his or her deposit and any withdrawals from such users account requires proof of government issued identification. In addition, payment service providers use their own identity and internet service provider (ISP) verification software. Despite all such measures, it is conceivable that a user, underage, or otherwise could devise a way to evade the Company’s blocking measures and access its website from the United States or any other foreign jurisdiction in which the Company is not then permitted to operate.
Violations of laws in one jurisdiction could result in disciplinary action in other jurisdictions. Licenses, approvals or findings of suitability may be revoked, suspended or conditioned. In sum, the Company may not be able to obtain or maintain all necessary registrations, licenses, permits or approvals. The licensing process may result in delays or adversely affect our operations and our ability to maintain key personnel, and our efforts to comply with any new licensing regulations will increase our costs.
A reduction in discretionary consumer spending, from an economic downturn or disruption of financial markets or other factors, could negatively impact the financial performance of the Company as it develops its iGaming business.
Gaming and other leisure activities that the Company plans to offer represent discretionary expenditures and players’ participation in those activities may decline if discretionary consumer spending declines, including during economic downturns, when consumers generally earn less disposable income. Changes in discretionary consumer spending or consumer preferences are driven by factors beyond the Company’s control, such as:
| ● | perceived or actual general economic conditions; |
| ● | fears of recession and changes in consumer confidence in the economy; |
| ● | high energy, fuel and other commodity costs; |
| ● | tariffs and unemployment; |
| ● | the potential for bank failures or other financial crises; |
| ● | a soft job market; |
| ● | an actual or perceived decrease in disposable consumer income and wealth; |
| ● | increases in taxes, including gaming taxes or fees; and |
| ● | terrorist attacks or other global events. |
During periods of economic contraction, the Company may not be able to generate expected or any revenues, while most of the Company’s costs remain fixed and some costs even increase, resulting in decreased earnings.
50
The Company will face cyber security risks that could result in damage to the Company’s reputation and/or subject them to fines, payment of damages, lawsuits and restrictions on the Company’s use of data.
The information systems and data of the Company, including those the Company maintains with the Company’s third-party service providers, may be subject to cyber security breaches in the future. Computer programmers and hackers may be able to penetrate the Company’s network security and misappropriate, copy or pirate the Company’s confidential information or that of third parties, create system disruptions or cause interruptions or shutdowns of the Company’s internal systems and services. The Company’s websites may become subject to denial-of-service attacks, where a website is bombarded with information requests eventually causing the website to overload, resulting in a delay or disruption of service. Computer programmers and hackers also may be able to develop and deploy viruses, worms and other malicious software programs that attack the Company’s products or otherwise exploit any security vulnerabilities of the Company’s products. Also, there is a growing trend of advanced persistent threats being launched by organized and coordinated groups against corporate networks to breach security for malicious purposes.
The techniques used to obtain unauthorized, improper, or illegal access to the Company’s systems, the Company’s data or customers’ data, disable or degrade service, or sabotage systems are constantly evolving and have become increasingly complex and sophisticated, may be difficult to detect quickly, and often are not recognized or detected until after they have been launched. Although the Company has developed, and plans to develop, systems and processes designed to protect the Company’s data and customer data and to prevent data loss and other security breaches and expects to continue to expend significant resources to bolster these protections, there can be no assurance that these security measures will provide absolute security.
Disruptions in the availability of their computer systems, through cyber-attacks or otherwise, could damage our computer or telecommunications systems, impact our ability to service our customers, adversely affect our operations and results of operations, and have an adverse effect on our reputation. The costs to eliminate or alleviate security problems, bugs, viruses, worms, malicious software programs and security vulnerabilities could be significant, and the efforts to address these problems could result in interruptions, delays, cessation of service and loss of future customers and may impede our sales, distribution and other critical functions. We may also be subject to regulatory penalties and litigation by customers and other parties whose information has been compromised, all of which could have a material adverse effect on our business, results of operations and cash flows.
Risks Relating to Our Plans to Allow Players to Deposit and Withdraw Cryptocurrency
Bitcoin, Ethereum, Litecoin and other digital assets are novel assets, and are subject to significant legal, commercial, regulatory and technical uncertainty.
As described in greater detail above under “Item 1. Business—Our Company— Planned iGaming Casino Operations—D. B2B Focus: Blockchain-Enabled Technology Platform”, we plan to initially offer waging in only bitcoin, Ethereum, and Litecoin. In the future, we may also allow wagering in fiat, provided that we do not plan to offer the ability to withdraw a different type of cryptocurrency or currency than deposited or to exchange between cryptocurrencies or currencies. Wagers and payouts will be made only with the same cryptocurrency. For example, if a user places a wager in Ethereum, they can only win additional Ethereum and will only be able to withdraw Ethereum upon the settlement of any wager.
Bitcoin, Ethereum, Litecoin and other digital assets are relatively novel and are subject to significant uncertainty, which could adversely impact their price. The application of state and federal securities laws and other laws and regulations to digital assets is unclear in certain respects, and it is possible that regulators in the United States or foreign countries may interpret or apply existing laws and regulations in a manner that adversely affects the price of bitcoin, Ethereum, Litecoin and other digital assets.
51
The U.S. federal government, states, regulatory agencies, and foreign countries may also enact new laws and regulations, or pursue regulatory, legislative, enforcement or judicial actions, that could materially impact the price of bitcoin, Ethereum, Litecoin and other digital assets or the ability of individuals or institutions such as us to own or transfer bitcoin, Ethereum, Litecoin and other digital assets. For example, the U.S. executive branch, the SEC, the European Union’s Markets in Crypto Assets Regulation, among others have been active in recent years, and in the U.K., the Financial Services and Markets Act 2023, or FSMA 2023, became law. Additionally, acting Chairman of the SEC, Mark T. Uyeda, in January 2025, launched a cryptocurrency task force to develop “a comprehensive and clear regulatory framework” for cryptocurrency assets and in January 2025, President Trump issued an Executive Order setting forth five high-level policy objectives: (1) protecting the lawful use of blockchain networks, participation in mining and validation, and self-custody of digital assets without unlawful censorship; (2) promoting dollar-backed stablecoins; (3) ensuring fair and open access to banking services; (4) providing “regulatory clarity” for digital assets based on “well-defined jurisdictional regulatory boundaries;” and (5) prohibiting Central Bank Digital Currencies (“CBDC”). Subsequently, on March 6, 2025, President Trump signed an Executive Order establishing a Strategic Bitcoin Reserve, as well as a Digital Asset Stockpile, which will consist of other forms of digital currency forfeited to the federal government as part of criminal or civil proceedings.
It is not possible to predict whether, or when, any of these developments will lead to Congress granting additional authorities to the SEC or other regulators, or whether, or when, any other federal, state or foreign legislative bodies will take any similar actions. It is also not possible to predict the nature of any such additional authorities, how additional legislation or regulatory oversight might impact the ability of digital asset markets to function or the willingness of financial and other institutions to continue to provide services to the digital assets industry, nor how any new regulations or changes to existing regulations might impact the value of digital assets generally and bitcoin specifically. The consequences of increased regulation of digital assets and digital asset activities could adversely affect the market price of bitcoin, Ethereum, Litecoin and other digital assets and therefore the value of our assets, and in turn adversely affect the market price of our common stock.
Moreover, the risks of engaging in a digital assets iGaming strategy are relatively novel and may create complications due to the lack of experience that third parties have with companies engaging in such a strategy, such as increased costs of director and officer liability insurance or the potential inability to obtain such coverage on acceptable terms in the future.
The growth of the digital assets industry in general, and the use and acceptance of bitcoin, Ethereum, Litecoin and other digital assets in particular, may also impact the price of such digital assets and is subject to a high degree of uncertainty. The pace of worldwide growth in the adoption and use of bitcoin, Ethereum, Litecoin and other digital assets may depend, for instance, on public familiarity with digital assets, ease of buying, accessing or gaining exposure to bitcoin, Ethereum, Litecoin and other digital assets, institutional demand for such digital assets as an investment asset, the participation of traditional financial institutions in the digital assets industry, consumer demand for such digital assets as a means of payment, and the availability and popularity of alternatives to bitcoin, Ethereum, Litecoin and other digital assets. Even if growth in adoption of digital assets occurs in the near or medium-term, there is no assurance bitcoin, Ethereum, Litecoin and other digital assets usage will continue to grow over the long-term.
Because bitcoin, Ethereum, Litecoin and other digital assets have no physical existence beyond the record of transactions on the blockchain, a variety of technical factors related to the blockchain could also impact the price of bitcoin, Ethereum, Litecoin and other digital assets. For example, malicious attacks by miners, inadequate mining fees to incentivize validating of digital asset transactions, hard “forks” of the bitcoin blockchain into multiple blockchains, and advances in digital computing, algebraic geometry, and quantum computing could undercut the integrity of the blockchain and negatively affect the price of bitcoin, Ethereum, Litecoin and other digital assets. The liquidity of bitcoin, Ethereum, Litecoin and other digital assets may also be reduced and damage to the public perception of bitcoin, Ethereum, Litecoin and other digital assets may occur, if financial institutions were to deny or limit banking services to businesses that hold bitcoin, Ethereum, Litecoin and other digital assets, provide digital asset-related services or accept digital assets as payment, which could also decrease the price of digital assets. Similarly, the open-source nature of the bitcoin blockchain means the contributors and developers of the bitcoin blockchain are generally not directly compensated for their contributions in maintaining and developing the bitcoin blockchain, and any failure to properly monitor and upgrade the bitcoin blockchain could adversely affect the bitcoin blockchain and negatively affect the price of bitcoin and/or other digital assets.
Recent actions by U.S. banking regulators have reduced the ability of bitcoin-related services providers to gain access to banking services and liquidity of bitcoin may also be impacted to the extent that changes in applicable laws and regulatory requirements negatively impact the ability of exchanges and trading venues to provide services for bitcoin and other digital assets.
52
Regulatory change reclassifying bitcoin or other digital assets as a security could lead to our classification as an “investment company” under the Investment Company Act of 1940, as amended, or the 1940 Act, and could adversely affect the market price of bitcoin, Ethereum, Litecoin and other digital assets and the market price of our common stock.
Under Sections 3(a)(1)(A) and (C) of the 1940 Act, a company generally will be deemed to be an “investment company” for purposes of the 1940 Act if (1) it is, or holds itself out as being, engaged primarily, or proposes to engage primarily, in the business of investing, reinvesting or trading in securities or (2) it engages, or proposes to engage, in the business of investing, reinvesting, owning, holding or trading in securities and it owns or proposes to acquire investment securities having a value exceeding 40% of the value of its total assets (exclusive of U.S. government securities and cash items) on an unconsolidated basis. We do not believe that we are an “investment company,” as such term is defined in the 1940 Act, and are not registered as an “investment company” under the 1940 Act as of the date of this Report. We also do not plan to be an “investment company” or to register under the 1940 Act following the date of this Report.
While senior SEC officials have previously stated their view that bitcoin is not a “security” for purposes of the federal securities laws, a contrary determination by the SEC could lead to our classification as an “investment company” under the 1940 Act in the future, if the portion of our assets consists of investments in bitcoins (or other digital assets) exceeds 40% safe harbor limits prescribed in the 1940 Act, which would subject us to significant additional regulatory controls that could have a material adverse effect on our business and operations and may also require us to change the manner in which we conduct our business.
We plan to monitor our assets and income for compliance under the 1940 Act and seek to conduct our business activities in a manner such that we do not fall within its definitions of “investment company” or that we qualify under one of the exemptions or exclusions provided by the 1940 Act and corresponding SEC regulations. If bitcoin or other digital assets are determined to constitute a security for purposes of the federal securities laws, we would take steps to reduce the percentage of bitcoins (or other digital assets) that constitute investment assets under the 1940 Act. These steps may include, among others, selling bitcoins (or other digital assets) that we might otherwise hold for the long term and deploying our cash in non-investment assets, and we may be forced to sell our bitcoins (or other digital assets) at unattractive prices. We may also seek to acquire additional non-investment assets to maintain compliance with the 1940 Act, and we may need to incur debt, issue additional equity or enter into other financing arrangements that are not otherwise attractive to our business. Any of these actions could have a material adverse effect on our results of operations and financial condition. Moreover, we can make no assurance that we would successfully be able to take the necessary steps to avoid being deemed to be an investment company in accordance with the safe harbor. If we were unsuccessful, and if bitcoin or other digital assets are determined to constitute a security for purposes of the federal securities laws, then we would have to register as an investment company, and the additional regulatory restrictions imposed by 1940 Act could adversely affect the market price of bitcoin (or other digital assets) and in turn adversely affect the market price of our common stock.
A particular crypto asset’s status as a “security” in any relevant jurisdiction is subject to a certain degree of uncertainty and if we are unable to properly characterize a crypto asset, we may be subject to regulatory scrutiny, inquiries, investigations, fines, and other penalties, which may adversely affect our business, operating results, and financial condition.
Given the complexity of the crypto assets in the market, if we are unable to properly characterize a crypto asset, we may be subject to regulatory scrutiny and inquiries. It is possible that a change in the governing administration or the appointment of new SEC commissioners could substantially impact the views of the SEC and its staff. Public statements by senior officials at the SEC indicate that the SEC does not intend to take the position that bitcoin or Ethereum are securities (in their current form). Despite the conclusions we may draw based on our risk-based assessment regarding the likelihood of a particular crypto asset, all other crypto assets may be considered a ‘security’ under applicable laws.
53
Several foreign jurisdictions have taken a broad-based approach to classifying crypto assets as “securities,” while other foreign jurisdictions have adopted a narrower approach. As a result, certain crypto assets may be deemed to be a “security” under the laws of some jurisdictions but not others. Various foreign jurisdictions may, in the future, adopt additional laws, regulations, or directives that affect the characterization of crypto assets as “securities.”
The classification of a crypto asset as a security under applicable law has wide-ranging implications for the regulatory obligations that flow from the offer, sale, trading, and clearing of such assets. For example, a crypto asset that is a security in the United States may generally only be offered or sold in the United States pursuant to a registration statement filed with the SEC or in an offering that qualifies for an exemption from registration. Persons that effect transactions in crypto assets that are securities in the United States may be subject to registration with the SEC as a “broker” or “dealer.” Platforms that bring together purchasers and sellers to trade crypto assets that are securities in the United States are generally subject to registration as national securities exchanges, or must qualify for an exemption. Persons facilitating clearing and settlement of securities may be subject to registration with the SEC as a clearing agency. Foreign jurisdictions may have similar licensing, registration, and qualification requirements.
We plan to put into place procedures to analyze whether crypto assets that we plan to allow users to bet with could be deemed to be a “security” under applicable laws. Our policies and procedures are not expected to constitute a legal standard, but rather to represent a framework for our analysis, which will permit us to make a risk-based assessment regarding the likelihood that a particular crypto asset could be deemed a “security” under applicable laws. The risk-based assessments made by the Company are not a legal standard or binding on any regulatory body or court. Regardless of our conclusions, we could be subject to legal or regulatory action in the event the SEC, a state or foreign regulatory authority, or a court were to determine that a crypto asset supported on our platform is a “security” under applicable laws. We expect our risk assessment policies and procedures to continuously evolve to take into account case law, facts, and developments in technology.
There can be no assurances that we will properly characterize any given crypto asset as a security or non-security for purposes of determining whether our platform will support such crypto asset, or that the SEC, foreign regulatory authority, or a court, if the question was presented to it, would agree with our assessment. If the SEC, state or foreign regulatory authority, or a court were to determine that a supported crypto asset is a security, we may be subject to further additional compliance requirements and/or may be unable to offer customers the right to bet in such digital asset. In addition, we could be subject to judicial or administrative sanctions, which could result in injunctions, cease and desist orders, as well as civil monetary penalties, fines, and disgorgement, criminal liability, and reputational harm. Furthermore, if we remove any digital assets from our platform in the future, our decision may be unpopular with users and may reduce our ability to attract and retain customers.
We may be subject to regulatory developments related to crypto assets and crypto asset markets, which could adversely affect our business, financial condition, and results of operations.
As bitcoin and other digital assets are relatively novel and the application of state and federal securities laws and other laws and regulations to digital assets is unclear in certain respects, and it is possible that regulators in the United States or foreign countries may interpret or apply existing laws and regulations in a manner that adversely affects the price of bitcoin and other digital assets. The U.S. federal government, states, regulatory agencies, and foreign countries may also enact new laws and regulations, or pursue regulatory, legislative, enforcement or judicial actions, that could materially impact the price of bitcoin and other digital assets or the ability of individuals or institutions such as us to own or transfer bitcoin and other digital assets. For examples, see “— Bitcoin, Ethereum, Litecoin and other digital assets are novel assets, and are subject to significant legal, commercial, regulatory and technical uncertainty” above.
54
If bitcoin or other digital assets are determined to constitute a security for purposes of the federal securities laws, the additional regulatory restrictions imposed by such a determination could adversely affect the market price of bitcoin and such other digital assets, the value of our assets, and in turn adversely affect the market price of our common stock. See “— Regulatory change reclassifying bitcoin or other digital assets as a security could lead to our classification as an “investment company” under the Investment Company Act of 1940, as amended, or the 1940 Act, and could adversely affect the market price of bitcoin, Ethereum, Litecoin and other digital assets and the market price of our common stock” above. Moreover, the risks of us engaging in the digital asset iGaming industry create complications due to the lack of experience that third parties have with companies engaging in such a strategy, such as increased costs of director and officer liability insurance or the potential inability to obtain such coverage on acceptable terms in the future.
In the event we hold digital assets, such holdings are expected to be less liquid and more volatile than our existing cash and cash equivalents and may not be able to serve as a source of liquidity for us to the same extent as cash and cash equivalents.
Historically, the crypto currency markets have been characterized by significant volatility in price, limited liquidity and trading volumes compared to sovereign currencies markets, relative anonymity, a developing regulatory landscape, potential susceptibility to market abuse and manipulation, compliance and internal control failures at exchanges, and various other risks inherent in its entirely electronic, virtual form and decentralized network. During times of market instability, we may not be able to sell our bitcoin or other digital assets at favorable prices or at all. For example, a number of bitcoin trading venues temporarily halted deposits and withdrawals in 2022. As a result, our bitcoin holdings may not be able to serve as a source of liquidity for us to the same extent as cash and cash equivalents. Further, bitcoin and other digital assets we hold with our custodians and transact with our trade execution partners do not enjoy the same protections as are available to cash or securities deposited with or transacted by institutions subject to regulation by the Federal Deposit Insurance Corporation or the Securities Investor Protection Corporation. Additionally, we may be unable to enter into term loans or other capital raising transactions collateralized by our unencumbered bitcoin and other digital assets or otherwise generate funds using our bitcoin and other digital holdings, including in particular during times of market instability or when the price of bitcoin (or certain other digital assets) has declined significantly. If we are unable to sell our bitcoin and other digital assets, enter into additional capital raising transactions using bitcoin or our other digital holdings as collateral, or otherwise generate funds using our digital asset holdings, or if we are forced to sell our digital assets at a significant loss, in order to meet our working capital requirements, our business and financial condition could be negatively impacted.
If we or our third-party service providers experience a security breach or cyberattack and unauthorized parties obtain access to our bitcoin and other digital assets, or if our private keys are lost or destroyed, or other similar circumstances or events occur, we may lose some or all of our bitcoin or other digital assets and our financial condition and results of operations could be materially adversely affected.
Substantially all of the bitcoin and other digital assets we plan to own are expected to be held in custody accounts at U.S.-based institutional-grade digital asset custodians, or equivalent international institutional-grade digital asset custodians in developed jurisdictions. Security breaches and cyberattacks will be of particular concern with respect to our bitcoin and digital asset holdings. Bitcoin and other blockchain-based cryptocurrencies and the entities that provide services to participants in the bitcoin ecosystem have been, and may in the future be, subject to security breaches, cyberattacks, or other malicious activities. For example, in October 2021 it was reported that hackers exploited a flaw in the account recovery process and stole from the accounts of at least 6,000 customers of the Coinbase exchange, although the flaw was subsequently fixed and Coinbase reimbursed affected customers. Similarly, in November 2022, hackers exploited weaknesses in the security architecture of the FTX Trading digital asset exchange and reportedly stole over $400 million in digital assets from customers. A successful security breach or cyberattack could result in:
| ● | a partial or total loss of our bitcoin or other digital assets in a manner that may not be covered by insurance or the liability provisions of the custody agreements with the custodians who hold our bitcoin or other digital assets; | |
| ● | harm to our reputation and brand; |
| ● | improper disclosure of data and violations of applicable data privacy and other laws; or |
| ● | significant regulatory scrutiny, investigations, fines, penalties, and other legal, regulatory, contractual and financial exposure. |
55
Further, any actual or perceived data security breach or cybersecurity attack directed at other companies with digital assets or companies that operate digital asset networks, regardless of whether we are directly impacted, could lead to a general loss of confidence in the broader bitcoin blockchain ecosystem or in the use of the bitcoin or other digital assets network to conduct financial transactions, which could negatively impact us.
Attacks upon systems across a variety of industries, including industries related to bitcoin and other digital assets, are increasing in frequency, persistence, and sophistication, and, in many cases, are being conducted by sophisticated, well-funded and organized groups and individuals, including state actors. The techniques used to obtain unauthorized, improper or illegal access to systems and information (including personal data and digital assets), disable or degrade services, or sabotage systems are constantly evolving, may be difficult to detect quickly, and often are not recognized or detected until after they have been launched against a target. These attacks may occur on our systems or those of our third-party service providers or partners. We may experience breaches of our security measures due to human error, malfeasance, insider threats, system errors or vulnerabilities or other irregularities. In particular, we expect that unauthorized parties will attempt to gain access to our systems and facilities, as well as those of our partners and third-party service providers, through various means such as hacking, social engineering, phishing and fraud. Threats can come from a variety of sources, including criminal hackers, hacktivists, state-sponsored intrusions, industrial espionage, and insiders. In addition, certain types of attacks could harm us even if our systems are left undisturbed. For example, certain threats are designed to remain dormant or undetectable, sometimes for extended periods of time, or until launched against a target and we may not be able to implement adequate preventative measures. Further, there has been an increase in such activities due to the increase in work-from-home arrangements. The risk of cyberattacks could also be increased by cyberwarfare in connection with the ongoing Russia-Ukraine and Israel-Hamas conflicts, or other future conflicts, including potential proliferation of malware into systems unrelated to such conflicts. Any future breach of our operations or those of others in the bitcoin industry, including third-party services on which we rely, could materially and adversely affect our financial condition and results of operations.
Intellectual property rights claims may adversely affect the operation of some or all crypto asset networks.
Third parties may assert intellectual property claims relating to the holding and transfer of crypto assets and their source code. Regardless of the merit of any intellectual property or other legal action, any threatened action that reduces confidence in some or all crypto asset networks’ long-term viability or the ability of end-users to hold and transfer crypto assets may adversely affect an investment in us. Additionally, a meritorious intellectual property claim could prevent us and other end-users from accessing some or all crypto asset networks or holding or transferring their crypto assets. As a result, an intellectual property claim against us or other large crypto asset network participants could adversely affect an investment in us.
The impact of geopolitical and economic events on the supply and demand for crypto assets, including bitcoin, is uncertain.
Geopolitical crises may motivate large-scale purchases of bitcoin and other crypto assets, which could increase the price of bitcoin and other crypto assets rapidly, for example, President Trump recently signed an Executive Order establishing a Strategic Bitcoin Reserve, as well as a Digital Asset Stockpile, which will consist of other forms of digital currency forfeited to the federal government as part of criminal or civil proceedings. Each of these may increase the likelihood of a subsequent price decrease as crisis-driven purchasing behavior dissipates, adversely affecting the value of our inventory following such downward adjustment. Such risks are similar, although not identical, to the risks of purchasing commodities in general uncertain times, such as the risk of purchasing, holding or selling gold. Alternatively, as an emerging asset class with limited acceptance as a payment system or commodity, global crises and general economic downturn may discourage investment in bitcoin as investors focus their investment on less volatile asset classes as a means of hedging their investment risk.
56
As an alternative to fiat currencies that are backed by central governments, bitcoin, which is relatively new, is subject to supply and demand forces. How such supply and demand will be impacted by geopolitical events is largely uncertain but could be harmful to us and investors in securities. Political or economic crises may motivate large-scale acquisitions or sales of bitcoin either globally or locally. Such events could have a material adverse effect on our business, prospects or operations and the value of any bitcoin we hold.
The characteristics of crypto assets have been, and may in the future continue to be, exploited to facilitate illegal activity such as fraud, money laundering, tax evasion and ransomware scams; all of which may have an adverse effect on the market for, and regulation of, crypto assets, and our operations.
Digital currencies and the digital currency industry are relatively new and, in many cases, lightly regulated or largely unregulated. Some types of digital currency have characteristics, such as the speed with which digital currency transactions can be conducted, the ability to conduct transactions without the involvement of regulated intermediaries, the ability to engage in transactions across multiple jurisdictions, the irreversible nature of certain digital currency transactions and encryption technology that anonymizes these transactions, that make digital currency particularly susceptible to use in illegal activity such as fraud, money laundering, tax evasion and ransomware scams. Two prominent examples of marketplaces that accepted digital currency payments for illegal activities include Silk Road, an online marketplace on the dark web that, among other things, facilitated the sale of illegal drugs and forged legal documents using digital currencies and AlphaBay, another darknet market that utilized digital currencies to hide the locations of its servers and identities of its users. Both of these marketplaces were investigated and closed by U.S. law enforcement authorities. U.S. regulators, including the SEC, Commodity Futures Trading Commission, and Federal Trade Commission, as well as non-U.S. regulators, have taken legal action against persons alleged to be engaged in Ponzi schemes and other fraudulent schemes involving digital currencies. In addition, in July 2021, the Federal Bureau of Investigation noted the increasing use of digital currency in various ransomware scams.
While we expect that our risk management and compliance framework, which is planned to include thorough reviews as part of our due diligence process (either in connection with onboarding new customers or monitoring existing customers), will be reasonably designed to detect any such illicit activities conducted by our customers, we may not be able to detect any such illegal activity in all instances. Because the speed, irreversibility and anonymity of certain digital currency transactions make them more difficult to track, fraudulent transactions may be more likely to occur, we or our potential banking counterparties may be specifically targeted by individuals seeking to conduct fraudulent transfers, and it may be difficult or impossible for us to detect and avoid such transactions in certain circumstances. Future negative perceptions regarding digital currencies as a result of the above, or future governmental regulation, may have an adverse effect on the market for, and regulation of, crypto assets, and our operations.
Incorrect or fraudulent digital asset transactions may be irreversible.
Digital asset transactions are not, from an administrative perspective, reversible without the consent and active participation of the recipient of the digital assets from the transaction. In theory, digital asset transactions may be reversible with the control or consent of a majority of the processing power on the network, however, we do not expect that we could in the future, possess sufficient processing power to effect this reversal, nor is it likely that sufficient consensus on the relevant network could or would be achieved to enable such a reversal. Once a transaction has been verified and recorded in a block that is added to a blockchain, an incorrect transfer of digital assets or a theft thereof generally will not be reversible, and we may not have sufficient recourse to recover our losses from any such transfer or theft. It is possible that, through computer or human error, or through theft or criminal action, our digital assets could be transferred in incorrect amounts or to unauthorized third parties, or to uncontrolled accounts. In the past, hackers have successfully employed a social engineering attack against certain service providers and misappropriated digital assets. To the extent that we are unable to recover our losses from such action, error or theft, such events could result in significant losses, hurt our brand and reputation, and adversely impact our business.
57
The decentralized nature of crypto asset systems may lead to slow or inadequate responses to crises, which may negatively affect our business.
The decentralized nature of the governance of crypto asset systems may lead to ineffective decision making that slows development or prevents a network from overcoming emergent obstacles. Governance of many crypto asset systems is by voluntary consensus and open competition with no clear leadership structure or authority. To the extent lack of clarity in corporate governance of the bitcoin system leads to ineffective decision making that slows development and growth of bitcoin, the value of our securities may be adversely affected.
It may be illegal now, or in the future, to acquire, own, hold, sell or use bitcoin or other crypto assets, participate in blockchains or utilize similar crypto assets in one or more countries, the ruling of which would adversely affect us.
Although currently crypto assets generally are not regulated or are lightly regulated in most countries, one or more countries such as China and Russia, which have taken harsh regulatory action in the past, may take regulatory actions in the future that could severely restrict the right to acquire, own, hold, sell or use these crypto assets or to exchange for fiat currency. In many nations, particularly in China and Russia, it is illegal to accept payment in bitcoin and other crypto assets for consumer transactions and banking institutions are barred from accepting deposits of bitcoin and in China, crypto trading and mining is banned altogether. Such restrictions may adversely affect us as the large-scale use of bitcoin as a means of exchange is presently confined to certain regions globally. Such circumstances have a material adverse effect on our business, prospects or operations and potentially the value of any digital assets we hold, and harm investors.
The limited rights of legal recourse against us, and our lack of insurance protection expose us and our stockholders to the risk of loss of our crypto assets for which no person is liable.
The crypto assets held by us will not be insured. Therefore, a loss may be suffered with respect to our crypto assets which is not covered by insurance and for which no person is liable in damages which could adversely affect our operations and, consequently, an investment in us.
Digital assets held by us are not subject to FDIC or SIPC protections.
We do not plan to hold our crypto assets with a banking institution or a member of the Federal Deposit Insurance Corporation (“FDIC”) or the Securities Investor Protection Corporation (“SIPC”) and, therefore, our crypto assets are not expected to be subject to the protections enjoyed by depositors with FDIC or SIPC member institutions.
Risks Related to Our Business Operations
Our License Agreements with the University of Oxford and other licensors may be terminated in certain circumstances without our consent.
All of our License Agreements with the University of Oxford and other licensors remain subject to various conditions and covenants, and provide for certain termination rights to the licensors. Those agreements typically allow termination by the licensor for our failure to pay amounts due timely, our failure to cure a material breach under the terms of the applicable license agreement, and our insolvency. As a result, if we are deemed insolvent, or in the event we seek bankruptcy protection, the licensors of our license agreements may terminate their license agreements with us. In the event such license agreements are terminated, we could lose the right to develop all of our platforms and technologies, may lose any investments made towards developing such platforms and technologies, and may be left without any intellectual property, product pathways, or development opportunities. Such terminations may result in the value of our securities declining in value or becoming worthless, the need for us to change our business plan, and may result in the Company seeking bankruptcy protection.
58
We owe a significant amount of money to the University of Oxford, which funds we do not have. The university may take action against us to enforce their rights to payment in the future, which could have a material adverse effect on us and our operations.
Due to recent financial constraints, the Company has been unable to timely pay amounts due to the University of Oxford (“Oxford”), the licensor of the majority of the Company’s licenses and patents and the Company’s research partner. Oxford alleges that an aggregate of approximately $1.4 million is owed from the Company and one of its subsidiaries to Oxford under the terms of licenses and agreements with Oxford and related parties. The Company is currently in ongoing discussions with Oxford to reduce that amount and enter into a payment plan with regards to the amounts owed; however, no definitive terms or extensions have been agreed to date. Oxford has also notified the Company that it is not willing to discuss any new projects or arrangements until all outstanding invoices have been paid or a payment plan has been agreed to; has engaged a law firm to seek the collection of the amounts owed, together with interest; and has threatened legal proceedings against us. While we are hopeful that we can come to mutually agreeable terms regarding a settlement, payment plan, and/or extension, with Oxford, we may not have sufficient funds to pay amounts due to Oxford in the near term, if at all, and Oxford may take action against us, including filing legal proceedings against us seeking amounts due and interest, attempting to terminate their relationship with us, and/or filing a wind-up petition against one of the Company’s subsidiaries in the U.K. If Oxford were to take legal action against us or terminate their relationship with us, we may be forced to scale back our business plan and/or seek bankruptcy protection. We may be subject to litigation and damages for our failure to pay amounts due to Oxford, and may be forced to pay interest and penalties, which funds we do not currently have. We plan to seek to raise funding in the future to support our operations, and to pay amounts due to Oxford, through a combination of equity offerings, debt financing or other capital sources, including potentially collaborations, licenses and other similar arrangements, which may not be available on favorable terms, if at all. The sale of additional equity or debt securities, if accomplished, may result in dilution to our then stockholders. The Company believes that due to the Company’s move into the online gaming/casino industry, Oxford may face certain additional challenges and constraints in continuing its contractual relationship with the Company due to potential Economic, Social and Governance restrictions imposed by Oxford. Such challenges and constraints may result in Oxford terminating or otherwise exiting its contractual relationship with the Company, potentially with limited notice or recourse by the Company, including terminating prior licenses, while seeking immediate repayment of amounts currently due.
Our results of operations may be adversely affected by fluctuations in currency values.
We expend expenses in currencies other than the U.S. dollar. Our reporting currency is the United States dollar. The functional currency of certain subsidiaries is the Canadian Dollar (“CAD”) or British Pound (“£” or “GBP”). The resulting translation adjustments are recognized in stockholders’ equity as a component of accumulated other comprehensive income. Comprehensive income is defined as the change in equity of an entity from all sources other than investments by owners or distributions to owners and includes foreign currency translation adjustments as described above. The Company recognized a $518 foreign currency transaction gain and $2,380 in foreign currency transaction loss for the years ended December 31, 2024 and 2023, respectively.
Changes in the value of the currencies which we pay expenses (and in the future receive revenues), versus each other, and the U.S. dollar, could result in an adverse charge being recorded to our income statement.
Economic uncertainty may affect our access to capital and/or increase the costs of such capital.
Global economic conditions continue to be volatile and uncertain due to, among other things, consumer confidence in future economic conditions, fears of recession and trade wars, tariffs, the price of energy, fluctuating interest rates, the availability and cost of consumer credit, the availability and timing of government stimulus programs, levels of unemployment, increased inflation, tax rates, and the war between Ukraine and Russia which began in February 2022, and Israel and Hamas, which began in October 2023 and which threatens to spread to other Middle Eastern countries. These conditions remain unpredictable and create uncertainties about our ability to raise capital in the future. In the event required capital becomes unavailable in the future, or more costly, it could have a material adverse effect on our business, future results of operations, and financial condition.
59
Tariffs and other non-tariff trade barriers may impact our ability to access capital or the cost of such capital, and our ability to market to customers in certain jurisdictions
We depend on our key personnel and our ability to attract and retain employees.
Our future growth and success depend on our ability to recruit, retain, manage and motivate our employees. We are highly dependent on our current management, including our Chief Executive Officer, Blair Jordan and our Chief Accounting Officer, Eric R. Van Lent. The inability to hire or retain experienced management personnel could adversely affect our ability to execute our business plan and harm our operating results. The competition for qualified personnel is intense and we may be unable to continue to attract and retain qualified personnel necessary for the development of our business or to recruit suitable replacement personnel.
Our employees may have previously engaged, and/or may in the future engage, in misconduct or other improper activities, including noncompliance with regulatory standards and legal requirements.
We are exposed to the risk of employee fraud or other misconduct. Misconduct by employees could include intentional failures to comply with FDA, SEC or Office of Inspector General regulations, or regulations of any other applicable regulatory authority, failure to provide accurate information to the FDA or the SEC, failure to disclose accurate information in SEC filings, failure to comply with applicable manufacturing standards, other federal, state or foreign laws and regulations, report information or data accurately or disclose unauthorized activities. Employee misconduct could also involve the improper use of information, including information obtained in the course of clinical trials, or illegal appropriation of drug product, which could result in government investigations and serious harm to our reputation. Despite our adoption of a Code of Ethics, employee misconduct is not always possible to identify and deter. The precautions we take to detect and prevent these prohibited activities may not be effective in controlling unknown or unmanaged risks or losses or in protecting us from governmental investigations or other actions or lawsuits stemming from a failure to be in compliance with such laws or regulations. If any such actions are instituted against our company, and we are not successful in defending our company or asserting our rights, those actions could have a significant impact on our business, including the imposition of significant fines or other sanctions.
We are subject to the U.S. Foreign Corrupt Practices Act and other anti-corruption laws, as well as export control laws, customs laws, sanctions laws and other laws governing our operations. If we fail to comply with these laws, we could be subject to civil or criminal penalties, other remedial measures, and legal expenses, which could adversely affect our business, results of operations and financial condition.
Our operations are subject to anti-corruption laws, including the U.S. Foreign Corrupt Practices Act (“FCPA”), notwithstanding the fact that President Trump has issued an Executive Order in February 2025, pausing all future investigations and enforcement actions under the FCPA for at least 180 days, and other anti-corruption laws that apply in countries in which we do business. The FCPA and these other laws generally prohibit our company and our employees and intermediaries from bribing, being bribed or making other prohibited payments to government officials or other persons to obtain or retain business or gain some other business advantage. We and our commercial partners operate in a number of jurisdictions that pose a high risk of potential FCPA violations, and we participate in collaborations and relationships with third parties whose actions could potentially subject us to liability under the FCPA (notwithstanding the current pause on FCPA investigations and enforcement action) or local anti-corruption laws. In addition, we cannot predict the nature, scope or effect of future regulatory requirements to which our international operations might be subject or the manner in which existing laws might be administered or interpreted.
60
We are also subject to other laws and regulations governing our international operations, including regulations administered by the governments of the U.S., Canada, Israel, the U.K. and authorities in the EU, including applicable export control regulations, economic sanctions on countries and persons, customs requirements and currency exchange regulations, collectively referred to as the Trade Control Laws.
However, there is no assurance that we will be completely effective in ensuring our compliance with all applicable anti-corruption laws, including the FCPA or other legal requirements, including Trade Control Laws. If we are not in compliance with the FCPA (notwithstanding the current pause on FCPA investigations and enforcement action) and other anti-corruption laws or Trade Control Laws, we may be subject to criminal and civil penalties, disgorgement and other sanctions and remedial measures, and legal expenses, which could have an adverse impact on our business, financial condition, results of operations and liquidity. Likewise, any investigation of any potential violations of the FCPA, other anti-corruption laws by the U.S. or other authorities could also have an adverse impact on our reputation, business, financial condition and results of operations.
Security breaches, loss of data and other disruptions could compromise sensitive information related to our business, prevent us from accessing critical information or expose us to liability, which could adversely affect our business and our reputation.
In the ordinary course of business, we expect to collect and store sensitive data, including legally protected patient health information, credit card information, personally identifiable information about our employees, intellectual property, and proprietary business information. The secure processing, storage, maintenance and transmission of this critical information is vital to our operations and business strategy, and we devote significant resources to protecting such information. Although we take measures to protect sensitive information from unauthorized access or disclosure, our information technology and infrastructure may be vulnerable to attacks by hackers, or viruses, breaches or interruptions due to employee error, malfeasance or other disruptions, or lapses in compliance with privacy and security mandates. Any such virus, breach or interruption could compromise our networks and the information stored there could be accessed by unauthorized parties, publicly disclosed, lost or stolen. We have measures in place that are designed to prevent, and if necessary, to detect and respond to such security incidents and breaches of privacy and security mandates. However, in the future, any such access, disclosure or other loss of information could result in legal claims or proceedings, liability under laws that protect the privacy of personal information, such as the Health Insurance Portability and Accountability Act (HIPAA) and European Union General Data Protection Regulation (“GDPR”), government enforcement actions and regulatory penalties. Unauthorized access, loss or dissemination could also disrupt our operations, including our ability to process samples, provide test results, share and monitor safety data, bill payors or patients, provide customer support services, conduct research and development activities, process and prepare company financial information, manage various general and administrative aspects of our business and may damage our reputation, any of which could adversely affect our business, financial condition and results of operations.
GDPR, which applies to all EU member states includes substantial fines for breaches of the data protection rules and may require us to put in place additional mechanisms ensuring compliance with the new and changing data protection rules. GDPR is a complex law and the regulatory guidance is still evolving, including with respect to how GDPR should be applied in the context of clinical trials or other transactions from which we may gain access to personal data. GDPR increases our costs of compliance and results in greater legal risks.
We may enter into strategic transactions in the future which may result in a material change in our operations and/or a change of control.
In December 2023, we engaged A.G.P./Alliance Global Partners as financial advisor to explore and evaluate strategic alternatives to enhance shareholder value, which engagement concluded in October, 2024. Notwithstanding the conclusion of the A.G.P. engagement, the Company continues to evaluate potential strategic alternatives which may include, but are not limited to, an acquisition, merger, reverse merger, other business combination, sale of assets, licensing or other strategic transactions involving the Company. The Company does not intend to discuss or disclose further developments during any such strategic transaction evaluation process unless and until its Board of Directors has approved a specific action or otherwise determined that further disclosure is appropriate. There is no assurance that any potential evaluation of a strategic transaction will result in the approval or completion of any specific transaction or outcome.
61
The Board of Directors and management team are committed to acting in the best interests of the Company, its stockholders and its stakeholders. There is no deadline or definitive timetable set for completion of the strategic alternatives review process and there can be no assurance that this process will result in the Company pursuing a transaction or any other strategic outcome.
As a result of the above, in the future, we may enter into transactions with parties seeking to merge and/or acquire us and/or our operations. While we have not entered into any agreements or understandings with any such parties to date, in the event that we do enter into such a transaction or transactions in the future, new shares of common stock or preferred stock could be issued resulting in substantial dilution to our then current stockholders and/or a change of control. As a result, our new majority stockholders may change the composition of our Board of Directors and may replace our current management. Any future transaction may also result in a change in our business focus. We have not entered into any agreements relating to any strategic transaction involving the Company as of the date of this Report and may not enter into such agreements in the future. Any future strategic transaction involving the Company or its operations may have a material effect on our operations, cash flows, results of operations, prospects, plan of operations, the listing of our common stock on Nasdaq, our officers, directors and majority stockholder(s), and the value of our securities.
Our directors and officers allocate their time to other businesses thereby causing conflicts of interest in their determination as to how much time to devote to our affairs.
Our directors and officers are not required to, and do not, commit their full time to our affairs, and certain of our directors hold positions, including other directorships and/or consulting roles, with other companies in various industries, which may result in a conflict of interest in allocating their time between our operations and others which they provide services to. If our directors’ and/or officers’ other business affairs require them to devote substantial amounts of time to such affairs in excess of their current commitment levels, it could limit their ability to devote time to our affairs which may have a negative impact on our operations. Additionally, such persons may have conflicts of interest in allocating their time among various business activities. These conflicts may not be resolved in our favor. Additionally, our directors, because of our corporate opportunity waiver, discussed above, may choose to, or be required to, provide corporate opportunities to the other companies which they are affiliated with. Actual or perceived conflicts of interest may have a material adverse effect on our results of operations which may have a material adverse effect on the value of our securities.
Certain of our executive officers and directors may in the future become affiliated with entities engaged in business activities similar to those conducted by us and, accordingly, may have conflicts of interest in determining to which entity a particular business opportunity should be presented.
Our executive officers and directors are, or may in the future become, affiliated with entities that are engaged in business activities similar to those that are conducted by us. Our officers and directors also may become aware of business opportunities which may be appropriate for presentation to us and the other entities to which they owe certain fiduciary or contractual duties. Accordingly, they may have conflicts of interest in determining whether a particular business opportunity should be presented to our company or to another entity. These conflicts may not be resolved in our favor and a potential opportunity may be presented to another entity prior to its presentation to us. Our Certificate of Incorporation provides that we renounce our interest in any corporate opportunity offered to any director or officer unless such opportunity is expressly offered to such person solely in his or her capacity as a director or officer of our company and such opportunity is one we are legally and contractually permitted to undertake and would otherwise be reasonable for us to pursue.
62
Our executive officers, directors, security holders and their respective affiliates may have competitive pecuniary interests that conflict with our interests.
We have not adopted a policy that expressly prohibits our directors, executive officers, security holders or affiliates from having a direct or indirect pecuniary or financial interest in any investment to be acquired or disposed of by us or in any transaction to which we are a party or have an interest. In fact, we may enter into a strategic transaction with a target business that is affiliated with our directors or executive officers. Nor do we have a policy that expressly prohibits any such persons from engaging for their own account in business activities of the types conducted by us. Accordingly, such persons or entities may have a conflict between their interests and ours. Certain of our officers and directors hold positions with companies which may be competitors of us.
Risks Relating to Our Life sciences Operations
We may be unable to monetize our existing life science assets.
With the acquisition of the Gaming Technology Platform in September 2024, the focus of the majority of our operations shifted from the development of therapeutics for unmet medical needs in chronic pain, inflammation and fibrosis by employing innovative research, and, where appropriate, combination therapy, to the creation of a blockchain casino and our planned iGaming operations. Moving forward, we plan to seek to monetize our existing life science assets, whether through outright sales of such assets or partnering with third parties to continue the development of such assets. Acquisition or partnership transactions may not be identified on favorable terms if at all, and we may choose to terminate existing license agreements and/or let certain of our life science intellectual property revert back to the original license holders or expire, if we are unable to come to terms regarding the sale of such assets or the partnership regarding the development of such assets. That may have a material adverse effect on our cash flows and prospects, could have adverse accounting implications, including the loss of net operating loss carryforwards, and could cause the value of our securities to decline in value.
Our License Agreements with the University of Oxford and other licensors may be terminated in certain circumstances without our consent.
All of our License Agreements with the University of Oxford and other licensors remain subject to various conditions and covenants, and provide for certain termination rights to the licensors. Those agreements typically allow termination by the licensor for our failure to pay amounts due timely, our failure to cure a material breach under the terms of the applicable license agreement, our insolvency, and certain changes in our operations, including operating in areas or industries which Oxford deems outside of its guidelines, including the iGaming industry. As a result, if we are deemed insolvent, or in the event we seek bankruptcy protection, or we continue to operate in the iGaming industry, the licensors of our license agreements may terminate their license agreements with us. In the event such license agreements are terminated, we could lose the right to develop all of our platforms and technologies, may lose any investments made towards developing such platforms and technologies, and may be left without any intellectual property, product pathways, or development opportunities. Such terminations may result in the value of our securities declining in value or becoming worthless, the need for us to change our business plan, and may result in the Company seeking bankruptcy protection.
Our ability to generate revenue from any of our potential life sciences products is subject to our ability to obtain regulatory approval and fulfill numerous other requirements and we may never be successful in generating revenues or becoming profitable.
Our ability to generate revenue from life sciences products depends on our ability to generate revenue or execute other business development arrangements. We do not expect to generate significant revenue, if any, from life sciences products unless and until we are able to obtain regulatory approval for, and successfully commercialize the product candidates we are developing or may develop. Successful commercialization, to the extent it occurs, will require achievement of many key milestones, including demonstrating safety and efficacy in clinical trials, obtaining regulatory approval for these product candidates, manufacturing, marketing and selling, or entering into other agreements to commercialize, those products for which we may obtain regulatory approval, satisfying any post-marketing requirements and obtaining reimbursement for our products from private insurance or government payors. Because of the uncertainties and risks associated with these activities, we cannot accurately and precisely predict the timing and amount, if any, of revenues, the extent of any further losses or when we might achieve profitability. We may never succeed in these activities and, even if we do, we may never generate revenues that are sufficient enough for us to achieve profitability. Even if we do achieve profitability, we may not be able to sustain or increase profitability on a quarterly or annual basis.
63
Our failure to become and remain profitable may depress the market price of our common stock and could impair our ability to raise capital, expand our business, diversify our product offerings or continue our operations.
All of our patents in the Anti-TNF and Fibrosis program are method of use patents, which may result in biosimilar drugs being used without our permission.
The success of our most advanced drug development platform depends on the enforceability of our method of use patents, as there are currently many biosimilar anti-TNF drugs in the market. If we are unable to obtain composition of matter patents, and enforce such patents, our ability to generate revenue from the anti-TNF platform may be significantly limited and competitors may be able to use our research to bring competing drugs to market which would reduce our market share.
The majority of our license agreements provide the licensors and/or counter-parties the right to use and/or exploit such licensed intellectual property.
The majority of our license agreements provide the licensors and/or counter-parties the right to use and/or exploit such licensed intellectual property, and in some cases provide them ownership of such intellectual property, know-how and research results. As such, we may be in competition with parties who we have license agreements with, will likely not have the sole right to monetize, sell or distribute our product candidates and may be subject to restrictions on use and territory of sales. Any or all of the above may have a material adverse effect on our results of operations and cash flows and ultimately the value of our securities.
We may not be successful in establishing development and commercialization collaborations which could adversely affect, and potentially prohibit, our ability to develop our product candidates.
Developing pharmaceutical products, conducting clinical trials, obtaining regulatory approval, establishing manufacturing capabilities and marketing approved products is expensive, and therefore we have, and may in the future, seek to enter into collaborations with companies that have more resources and experience in order to continue to develop and commercialize our product candidates. We also may be required due to financial or scientific constraints to enter into additional collaboration agreements to research and/or to develop and commercialize our product candidates. The establishment and realization of such collaborations may not be possible or may be problematic. There can be no assurance that we will be able to establish such additional collaborations on favorable terms, if at all, or that our current or future collaborative arrangements will be successful or maintained for any specific product candidate or indication. If we are unable to reach successful agreements with suitable collaboration partners for the ongoing development and commercialization of our product candidates, we may face increased costs, we may be forced to limit the scope and number of our product candidates we can commercially develop or the territories in which we commercialize such product candidates, and we may be unable to commercialize products or programs for which a suitable collaboration partner cannot be found. If we fail to achieve successful collaborations, our operating results and financial condition will be materially and adversely affected.
In addition, the terms of any collaboration agreements may place restrictions on our activities with respect to other products, including by limiting our ability to grant licenses or develop products with other third parties, or in different indications, diseases or geographical locations, or may place additional obligations on us with respect to development or commercialization of our product candidates. If we fail to comply with or breach any provision of a collaboration agreement, a collaborator may have the right to terminate, in whole or in part, such agreement or to seek damages.
64
Our collaboration and licensing agreements are, and may in the future be, complex and involve sharing or division of ownership of certain data, know-how and intellectual property rights among the various parties. Accordingly, our collaborators could interpret certain provisions differently than we or our other collaborators which could lead to unexpected or inadvertent disputes with collaborators. In addition, these agreements might make additional collaborations, partnering or mergers and acquisitions difficult.
There is no assurance that a collaborator who is acquired by a third party would not attempt to change certain contract provisions that could negatively affect our collaboration. The acquiring company may also not accept the terms or assignment of our contracts and may seek to terminate the agreements. Any one of our collaborators could breach covenants, restrictions and/or sub-license agreement provisions leading us into disputes and potential breaches of our agreements with other partners.
Our and our partner’s operations are subject to risks associated with ongoing and potential future global conflicts, including specifically the operations of Yissum, our research partner.
In February 2022, an armed conflict escalated between Russia and Ukraine. The sanctions announced by the United States and other countries against Russia and Belarus following Russia’s invasion of Ukraine to date include restrictions on selling or importing goods, services, or technology in or from affected regions and travel bans and asset freezes impacting connected individuals and political, military, business, and financial organizations in Russia and Belarus. The United States and other countries could impose wider sanctions and take other actions should the conflict further escalate. Separately, in October 2023, Israel and certain Iranian-backed Palestinian forces began an armed conflict in Israel, the Gaza Strip, and surrounding areas, which spread to other Middle Eastern countries including Lebanon and Syria. There can be no assurance that such conflicts will be resolved, or will not spread to other countries in the region.
These wars are increasingly affecting economic and global financial markets and exacerbating ongoing economic challenges, including issues such as rising inflation and global supply-chain disruption. While we do not believe these conflicts currently have a material impact on our financial accounting and reporting, the degree to which we will be affected in the future largely depends on the nature and duration of uncertain and unpredictable events, and our business could be impacted. Additionally, we currently have agreements and relationships in place with Yissum Research Development Company of the Hebrew University of Jerusalem, Ltd., located in Israel, and Yissum’s operations may be materially affected by the ongoing war in Israel, which may delay, prevent, or materially increase the cost of the ongoing services Yissum is required to provide to the Company. Furthermore, future global conflicts or wars could create further economic challenges, including, but not limited to, increases in inflation and further global supply-chain disruption. Consequently, the ongoing Russia/Ukraine Hamas/Israel conflicts and/or other future global conflicts, could result in an increase in operating expenses and/or a decrease in any future revenue and could further have a material adverse effect on our results of operations and cash flow.
Any failure by our company to comply with existing regulations could harm our reputation and operating results.
We are subject to extensive regulation by U.S. federal and state and foreign governments in each of the U.S., European and Canadian markets, in which we plan to sell our products, or in markets where we have product candidates progressing through the approval process.
65
We must adhere to all regulatory requirements including FDA’s Good Laboratory Practice (GLP), Good Clinical Practice (GCP) and Current Good Manufacturing Practice (CGMP) Regulations, pharmacovigilance requirements, advertising and promotion restrictions, reporting and recordkeeping requirements, and their European equivalents. If we or our suppliers fail to comply with applicable regulations, including FDA pre-or post-approval requirements, then the FDA or other foreign regulatory authorities could sanction our company. Even if a drug is approved by the FDA or other competent authorities, regulatory authorities may impose significant restrictions on a product’s indicated uses or marketing or impose ongoing requirements for potentially costly post-marketing trials. Any of our product candidates which may be approved in the U.S. will be subject to ongoing regulatory requirements for manufacturing, labeling, packaging, storage, distribution, import, export, advertising, promotion, sampling, recordkeeping and submission of safety and other post-market information, including both federal and state requirements. In addition, manufacturers and manufacturers’ facilities are required to comply with extensive FDA requirements, including ensuring that quality control and manufacturing procedures conform to GMP. As such, we and our contract manufacturers (in the event contract manufacturers are appointed in the future) are subject to continual review and periodic inspections to assess compliance with GMP. Accordingly, we and others with whom we work will have to spend time, money and effort in all areas of regulatory compliance, including manufacturing, production, quality control and quality assurance. We will also be required to report certain adverse reactions and production problems, if any, to the FDA, and to comply with requirements concerning advertising and promotion for our products. Promotional communications with respect to prescription drugs are subject to a variety of legal and regulatory restrictions and must be consistent with the information in the product’s approved label. Similar restrictions and requirements exist in the EU and other markets where we operate.
If a regulatory agency discovers previously unknown problems with a product, such as adverse events of unanticipated severity or frequency, or problems with the facility where the product is manufactured, or disagrees with the promotion, marketing or labeling of the product, it may impose restrictions on that product or on our company, including requiring withdrawal of the product from the market. If we fail to comply with applicable regulatory requirements, a regulatory agency or enforcement authority may issue warning letters, impose civil or criminal penalties, suspend regulatory approval, suspend any of our ongoing clinical trials, refuse to approve pending applications or supplements to approved applications submitted by us, impose restrictions on our operations, or seize or detain products or require a product recall.
In addition, any government investigation of alleged violations of law could require us to spend significant time and resources in response and could generate negative publicity. Any failure to comply with ongoing regulatory requirements may significantly and adversely affect our ability to commercialize and generate revenue from our future product candidates. If regulatory sanctions are applied or if regulatory approval is withdrawn, the value of our business and our operating results may be adversely affected.
Any action against us for violation of these laws, even if we successfully defend against it, could cause us to incur significant legal expenses, divert our management’s attention from the operation of our business and damage our reputation. We expect to spend significant resources on compliance efforts and such expenses are unpredictable. Changing laws, regulations and standards might also create uncertainty, higher expenses and increase insurance costs. As a result, we intend to invest all reasonably necessary resources to comply with evolving standards, and this investment might result in increased management and administrative expenses and a diversion of management time and attention from revenue-generating activities to compliance activities.
We are subject to federal, state and foreign healthcare laws and regulations and implementation of or changes to such healthcare laws and regulations could adversely affect our business and results of operations.
In both the U.S. and certain foreign jurisdictions, there have been a number of legislative and regulatory proposals to change the healthcare system in ways that could impact our ability to sell our future product candidates. If we are found to be in violation of any of these laws or any other federal, state or foreign regulations, we may be subject to administrative, civil and/or criminal penalties, damages, fines, individual imprisonment, we from federal health care programs and the restructuring of our operations. Any of these could have a material adverse effect on our business and financial results. Since many of these laws have not been fully interpreted by the courts, there is an increased risk that we may be found in violation of one or more of their provisions. Any action against us for violation of these laws, even if we ultimately are successful in our defense, will cause us to incur significant legal expenses and divert our management’s attention away from the operation of our business. In addition, in many foreign countries, particularly the countries of the EU, the pricing of prescription drugs is subject to government control.
66
In some foreign countries, the proposed pricing for a drug must be approved before it may be lawfully marketed. The requirements governing drug pricing vary widely from country to country.
For example, some European Union (EU) jurisdictions operate positive and negative list systems under which products may only be marketed once a reimbursement price has been agreed. To obtain reimbursement or pricing approval, some of these countries may require the completion of clinical trials that compare the cost-effectiveness of a particular product candidate to currently available therapies. Other member states allow companies to fix their own prices for medicines but monitor and control company profits. Such differences in national pricing regimes may create price differentials between EU member states. There can be no assurance that any country that has price controls or reimbursement limitations for pharmaceutical products will allow favorable reimbursement and pricing arrangements for any of our products. Historically, products launched in the U.K. and EU do not follow price structures of the U.S. In the U.K. and EU, the downward pressure on healthcare costs in general, particularly prescription medicines, has become intense. As a result, barriers to entry of new products are becoming increasingly high and patients are unlikely to use a drug product that is not reimbursed by their government. We may face competition from lower-priced products in foreign countries that have placed price controls on pharmaceutical products. In addition, the importation of foreign products may compete with any future product that we may market, which could negatively impact our profitability.
Specifically, in the U.S., we expect that the Affordable Care Act (ACA), as well as other healthcare reform measures that may be adopted in the future, may result in more rigorous coverage criteria and in additional downward pressure on the price that we may receive for any approved product. There have been judicial challenges to certain aspects of the ACA and numerous legislative attempts to repeal and/or replace the ACA in whole or in part, and we expect there will be additional challenges and amendments to the ACA in the future. At this time, the full effect that the ACA will have on our business in the future remains unclear. An expansion in the government’s role in the U.S. healthcare industry may cause general downward pressure on the prices of prescription drug products, lower reimbursements or any other product for which we obtain regulatory approval, reduce product utilization and adversely affect our business and results of operations. Any reduction in reimbursement from Medicare or other government programs may result in a similar reduction in payments from private payors. The implementation of cost containment measures or other healthcare reforms may prevent us from being able to generate revenue, attain profitability, or commercialize any of our future product candidates for which we may receive regulatory approval.
Risks Relating to Our Ineffective Disclosure Controls and Procedures
We have in the past, and may in the future, identify material weaknesses in our disclosure controls and procedures and internal control over financial reporting. If not remediated, our failure to establish and maintain effective disclosure controls and procedures and internal control over financial reporting could result in material misstatements in our financial statements and a failure to meet our reporting and financial obligations, each of which could have a material adverse effect on our financial condition and the trading price of our securities.
The Company’s management evaluated, with the participation of our principal executive officer and principal financial and accounting officer, the effectiveness of our disclosure controls and procedures as defined in Rules 13a-15(e) and 15d-15(e) of the Exchange Act, as of the end of March 31, 2024, June 30, 2024 and September 30, 2024. Based on their evaluation, our principal executive officer and principal financial and accounting officer concluded that, as of March 31, 2024, June 30, 2024 and September 30, 2024, our disclosure controls and procedures were not effective to provide assurance at a reasonable level that the information we are required to disclose in reports that we file or submit under the Exchange Act is recorded, processed, summarized and reported within the time periods specified in SEC rules and forms, and that such information is accumulated and communicated to our management, including our principal executive officer and principal financial and accounting officer, as appropriate, to allow timely decisions.
67
Management’s evaluation was based on the following material weaknesses in our internal control over financial reporting:
| ● | Ineffective controls: The Company’s review and control procedures did not operate at the appropriate level of precision to determine that reversals of year-end bonus accruals related to a discontinued R&D project should have been recorded as the forgiveness of a liability, and therefore an increase in Other Income on the income statement, as opposed to a reduction to R&D expense. |
A material weakness is a control deficiency or combination of control deficiencies, that results in more than a remote likelihood that a material misstatement of the annual or interim financial statements will not be prevented or detected on a timely basis. As a company with limited accounting resources, a significant amount of management’s time and attention has been and will be diverted from our business to ensure compliance with these regulatory requirements.
The Company has discussed the disclosure procedures and controls with its external accounting advisors, and is currently evaluating steps and processes to remedy such concerns.
Maintaining effective disclosure controls and procedures and effective internal control over financial reporting are necessary for us to produce reliable financial statements and we are committed to remediating our material weaknesses in such controls as promptly as possible. However, there can be no assurance as to when these material weaknesses will be remediated or that additional material weaknesses will not arise in the future. Any failure to remediate the material weaknesses, or the development of new material weaknesses in our internal control over financial reporting, could result in material misstatements in our financial statements and cause us to fail to meet our reporting and financial obligations on a timely and accurate basis. If our financial statements are not accurate, investors may not have a complete understanding of our operations or may lose confidence in our reported financial information. Likewise, if our financial statements are not filed on a timely basis as required by the SEC and Nasdaq, we could face severe consequences from those authorities. Any of these cases could result in a material adverse effect on our business, on our financial condition or have a negative effect on the trading price of our common stock and warrants. Further, if we fail to remedy any future deficiencies or maintain the adequacy of our disclosure controls and procedures and our internal controls, we could be subject to regulatory scrutiny, civil or criminal penalties or stockholder litigation against us or our management.
We can give no assurance that the measures we take in the future will remediate any additional material weaknesses which could be identified, or restatements of our financial statements will not arise in the future due to a failure to implement and maintain adequate internal control over financial reporting or circumvention of those controls.
Further, in the future, if we cannot conclude that we have effective internal control over our financial reporting, or if our independent registered public accounting firm is unable to provide an unqualified opinion regarding the effectiveness of our internal control over financial reporting (to the extent we may be required in the future), investors could lose confidence in the reliability of our financial statements, which could lead to a decline in our stock price. Failure to comply with reporting requirements could also subject us to sanctions and/or investigations by the SEC or Nasdaq, as applicable, or other regulatory authorities.
In addition, even if we are successful in strengthening our controls and procedures, those controls and procedures may not be adequate to prevent or identify irregularities or facilitate the fair presentation of our financial statements or our periodic reports filed with the SEC. This may require us to restate prior financial statements.
Risks Related to our Intellectual Property
We may not be able to adequately protect our future product candidates or our proprietary technology in the marketplace.
Our success will depend, in part, on our ability to obtain patents, protect our trade secrets and operate without infringing on the proprietary rights of others. We rely upon a combination of patents, trade secret protection (i.e., know-how), and confidentiality agreements to protect the intellectual property of our future product candidates. The strengths of patents and intellectual property in the pharmaceutical and iGaming fields involve complex legal and scientific questions and can be uncertain. Where appropriate, we seek patent protection for certain aspects of our products and technologies. Filing, prosecuting and defending patents globally can be prohibitively expensive.
68
Our policy is to look to patent technologies with commercial potential in jurisdictions with significant commercial opportunities. However, patent protection may not be available for some of the products or technology we are developing. If we must spend significant time and money protecting, defending or enforcing our patents, designing around patents held by others or licensing, potentially for large fees, patents or other proprietary rights held by others, our business, results of operations and financial condition may be harmed. We may not develop additional proprietary products that are patentable. As of the date hereof, we have an extensive portfolio of patents, including many granted patents and patents pending approval.
The patent positions of pharmaceutical products and iGaming technology are complex and uncertain. The scope and extent of patent protection for our future product candidates and iGaming technology are particularly uncertain. Our future pharmaceutical product candidates, if any, will be based on medicinal chemistry instead of cannabis plants. While we have sought patent protection, where appropriate, directed to, among other things, composition-of-matter for its specific formulations, their methods of use, and methods of manufacture, we do not have and will not be able to obtain composition of matter protection on these previously known CBD derivatives per se. We anticipate that the products we develop in the future will be based upon synthetic compounds we may discover. Although we have sought, and will continue to seek, patent protection in the U.S., Europe and other countries for our proprietary technologies, future product candidates, their methods of use, and methods of manufacture, any or all of them may not be subject to effective patent protection. If any of our products are approved and marketed for an indication for which we do not have an issued patent, our ability to use our patents to prevent a competitor from commercializing a non-branded version of our commercial products for that non-patented indication could be significantly impaired or even eliminated.
Publication of information related to our future product candidates by our company or others may prevent us from obtaining or enforcing patents relating to these products and product candidates. Furthermore, others may independently develop similar products, may duplicate our products, or may design around our patent rights. In addition, any of our issued patents may be opposed and/or declared invalid or unenforceable. If we fail to adequately protect our intellectual property, we may face competition from companies who attempt to create a generic product to compete with our future product candidates or attempt to market, sell or commercialize iGaming technology to compete with our technology. We may also face competition from companies who develop a substantially similar product to our future product candidates or substantially similar technology to our technology, that is not covered by any of our patents.
Many companies have encountered significant problems in protecting, defending and enforcing intellectual property rights in foreign jurisdictions. The legal systems of certain countries, particularly certain developing countries, do not favor the enforcement of patents and other intellectual property rights, particularly those relating to pharmaceuticals and iGaming technology, which could make it difficult for us to stop the infringement of our patents or marketing of competing products in violation of our proprietary rights generally. Proceedings to enforce our patent rights in foreign jurisdictions could result in substantial costs and divert our efforts and attention from other aspects of our business.
69
If third parties claim that intellectual property used by our company infringes upon their intellectual property, our operating profits could be adversely affected.
There is a substantial amount of litigation, both within and outside the U.S., involving patent and other intellectual property rights in the pharmaceutical and iGaming industries. We may, from time to time, be notified of claims that we are infringing upon patents, trademarks, copyrights or other intellectual property rights owned by third parties, and we cannot provide assurances that other companies will not, in the future, pursue such infringement claims against us, our commercial partners or any third-party proprietary technologies we have licensed. If we were found to infringe upon a patent or other intellectual property right, or if we failed to obtain or renew a license under a patent or other intellectual property right from a third party, or if a third party from whom we were licensing technologies was found to infringe upon a patent or other intellectual property rights of another third party, we may be required to pay damages, including damages of up to three times the damages found or assessed, if the infringement is found to be willful, suspend the manufacture of certain products or use of certain technology, or reengineer or rebrand our products or technology, if feasible, or we may be unable to enter certain new product markets. Any such claims could also be expensive and time consuming to defend and divert management’s attention and resources. Our competitive position could suffer as a result. In addition, if we have declined or failed to enter into a valid non-disclosure or assignment agreement for any reason, we may not own the invention or our intellectual property, and our products may not be adequately protected. Thus, we cannot guarantee that any of our future product candidates, or our commercialization thereof, or our iGaming technology, does not and will not infringe on any third party’s intellectual property.
If we are not able to adequately prevent disclosure of trade secrets and other proprietary information, the value of our technology and products could be significantly diminished.
We rely on trade secrets to protect our proprietary technologies, especially where we do not believe patent protection is appropriate or obtainable. However, trade secrets are difficult to protect. We rely in part on confidentiality agreements with current and former employees, consultants, outside scientific collaborators, sponsored researchers, contract manufacturers, vendors and other advisors to protect our trade secrets and other proprietary information. These agreements may not effectively prevent disclosure of confidential information and may not provide an adequate remedy in the event of unauthorized disclosure of confidential information. In addition, we cannot guarantee that we have executed these agreements with each party that may have or have had access to our trade secrets. Any party with whom we or they have executed such an agreement may breach that agreement and disclose our proprietary information, including our trade secrets, and we may not be able to obtain adequate remedies for such breaches.
Enforcing a claim that a party illegally disclosed or misappropriated a trade secret is difficult, expensive and time-consuming, and the outcome is unpredictable. Also, some courts inside and outside the U.S. are less willing or unwilling to protect trade secrets. If any of our trade secrets were to be lawfully obtained or independently developed by a competitor, we would have no right to prevent them, or those to whom they disclose such trade secrets, from using that technology or information to compete with us. If any of our trade secrets were to be disclosed to or independently developed by a competitor or other third-party, our competitive position would be harmed.
The expiration or loss of patent protection may adversely affect our future revenues and operating earnings.
We rely on patent, trademark, trade secret and other intellectual property protection. In particular, patent protection is important in the development and eventual commercialization of our product candidates. Patents covering our product candidates normally provide market exclusivity, which is important in order to improve the probability that our product candidates are able to become profitable.
One of our patents relating to our Fibrosis and anti-TNF program will expire in 2033; however, the majority of the patent portfolio has a longer lifespan. While we are seeking additional patent coverage which may protect the technology underlying these patents, there can be no assurances that such additional patent protection will be granted, or if granted, that these patents will not be infringed upon or otherwise held enforceable. Even if we are successful in obtaining a patent, patents have a limited lifespan. In the U.S., the natural expiration of a utility patent is generally 20 years after it is filed. Various extensions may be available; however, the life of a patent, and the protection it affords, is limited. Without patent protection of our product candidates, we may be open to competition from generic versions of such methods and compositions.
70
Accounting Risks
We have in the past, and may in the future, impair long-lived assets and intangible assets, including goodwill and acquired in-process research and development.
We review long-lived assets and certain identifiable assets (including intangible assets) for impairment whenever circumstances and situations change such that there is an indication that the carrying amounts may not be recovered. An impairment exists when the carrying value of the long-lived or intangible asset (including goodwill and acquired in-process research and development) is not recoverable and exceeds its estimated fair value. Goodwill represents the difference between the purchase price and the fair value of assets and liabilities acquired in a business combination. We review goodwill yearly, or more frequently whenever circumstances and situations change such that there is an indication that the carrying amounts may not be recovered, for impairment by initially considering qualitative factors to determine whether it is more likely than not that the fair value of a reporting unit is less than its carrying amount, including goodwill, as a basis for determining whether it is necessary to perform a quantitative analysis. If it is determined that it is more likely than not that the fair value of reporting unit is less than its carrying amount, a quantitative analysis is performed to identify goodwill impairment.
Our publicly-traded stock closed at $1,482.00 per share as of December 31, 2021; during 2022, the market value of our single reporting unit significantly declined. As of March 31, 2022, June 30, 2022, September 30, 2022 and December 31, 2022, the market value of our publicly-traded stock fell to $984.20, $322.24, $252.70 and $64.41, per share, respectively, and as such, we elected to conduct a quantitative analysis of goodwill to assess for impairment as of September 30, 2022 and December 31, 2022. We determined the fair market value of our single reporting unit and compared that value with the carrying amount of the reporting unit and determined that goodwill was impaired as of both measurement dates. As of September 30, 2022 and December 31, 2022, the carrying value exceeded the fair market value by $18,872,850 and $14,674,428, respectively. To recognize the impairment of goodwill, we recorded losses for these amounts at the end of the third and fourth quarters, which appear as a loss on goodwill impairment of $33,547,278 on the income statement for the year ended December 31, 2022.
Intangible assets and in-process research and development (“IP R&D”) assets represent the fair value assigned to technologies that were acquired on July 16, 2019 in connection with the Reorganization, which have not reached technological feasibility and have no alternative future use. IP R&D assets are considered to be indefinite-lived until the completion or abandonment of the associated research and development projects. During the period that the IP R&D assets are considered indefinite-lived, they are tested for impairment on an annual basis, or more frequently if we become aware of any events occurring or changes in circumstances that indicate that the fair value of the IP R&D assets are less than their carrying amounts. If and when development is complete, which generally occurs upon regulatory approval, and we are able to commercialize products associated with the IP R&D assets, these assets are then deemed definite-lived and are amortized based on their estimated useful lives at that point in time. If development is terminated or abandoned, we may record a full or partial impairment charge related to the IP R&D assets, calculated as the excess of the carrying value of the IP R&D assets over their estimated fair value.
As of December 31, 2022, the carrying amount of the IP R&D assets on the balance sheet was $12,405,084 (which consists of carrying value of $1,462,084 and $10,943,000 related to the Company’s CBR Pharma subsidiary and its 180 LP subsidiary, respectively). Per the valuation obtained from a third party as of year-end, the fair market value of the Company’s IP R&D assets was determined to be $9,063,000 (which consists of fair market values of $0 and $9,063,000 related to the Company’s CBR Pharma subsidiary and 180 LP subsidiary, respectively). As of this measurement date, the carrying value of the CBR Pharma and 180 LP subsidiaries’ assets exceeded their fair market values by $1,462,084 and $1,880,000, respectively. As such, management determined that the consolidated IP R&D assets were impaired by $3,342,084, and in order to recognize the impairment, the Company recorded a loss for this amount during the fourth quarter of 2022, which appears as a loss on impairment of IP R&D assets on the income statement. This reduced the IP R&D asset balances of its CBR Pharma subsidiary and its 180 LP subsidiary to zero and $9,063,000, respectively, as of December 31, 2022; the total consolidated IP R&D asset balance is $9,063,000 after impairment.
71
As of September 30, 2023, the carrying amount of the IP R&D assets on the balance sheet was $9,063,000 (which consisted of a balance related to the Company’s 180 LP subsidiary); the Company typically assesses asset impairment on an annual basis unless a triggering event or other facts or circumstances indicate that an evaluation should be performed at an earlier date. At the end of the third quarter of 2023, the Company assessed general economic conditions, industry and market considerations, the Company’s financial performance and all relevant legal, regulatory, and political factors that might indicate the possibility of impairment and concluded that, when these factors were collectively evaluated, it was likely that the asset was impaired. The Company recorded a loss in the amount of $9,063,000, which appeared as a loss on impairment to IP R&D assets on the income statement for the three and nine months ended September 30, 2023. As of December 31, 2023, the balance of the IP R&D assets on the balance sheet is $0.
An impairment recognized in one period may not be reversed in a subsequent period, even if the value of our common stock increases in the future. We have in the past and could in the future incur additional impairments of long-lived assets and/or intangible assets, including acquired in-process research and development and goodwill, which may be material.
Risks Related to our Common Stock and Warrants
We currently have an illiquid and volatile market for our common stock, and the market for our common stock is and may remain illiquid and volatile in the future.
We currently have a highly sporadic, illiquid and volatile market for our common stock, which market is anticipated to remain sporadic, illiquid and volatile in the future. During the last 52 weeks our common stock has traded as high as $17.75 per share and as low as $1.02 per share. The market price of our common stock may continue to be highly volatile and subject to wide fluctuations. Our financial performance, government regulatory action, tax laws, interest rates, and market conditions in general could have a significant impact on the future market price of our common stock.
| ● | “short squeezes”; |
| ● | comments by securities analysts or other third parties, including blogs, articles, message boards and social and other media; |
| ● | large stockholders exiting their position in our securities or an increase or decrease in the short interest in our securities; |
| ● | actual or anticipated fluctuations in our financial and operating results; |
| ● | changes in foreign currency exchange rates; |
| ● | the commencement, enrollment or results of our planned or future clinical trials of our product candidates or those of our competitors; |
| ● | the success of competitive drugs or therapies; |
| ● | regulatory or legal developments in the U.S. and other countries; |
| ● | the success of competitive products or technologies; |
| ● | developments or disputes concerning patent applications, issued patents or other proprietary rights; |
| ● | the recruitment or departure of key personnel; |
| ● |
the level of expenses related to our iGaming operations, product candidates or clinical development programs; |
| ● | our ability to economically source third party suppliers for our iGaming operations, including but not limited to content developers (including providers of games of chance used in online casinos, and/or sports book technology providers), marketing, technology and operational services providers; |
72
| ● | litigation matters, including amounts which may or may not be recoverable pursuant to our officer and director insurance policies, regulatory actions affecting the Company and the outcome thereof; |
| ● | actual or anticipated changes in estimates as to financial results, development timelines or recommendations by securities analysts; |
| ● | disputes or other developments relating to proprietary rights, including patents, litigation matters and our ability to obtain patent protection for our technologies; |
| ● | the timing and outcome of our plans to commercialize our iGaming intellectual property; |
| ● | significant lawsuits, including patent or stockholder litigation; |
| ● | variations in our financial results or those of companies that are perceived to be similar to us; |
| ● | changes in the structure of healthcare payment systems, including coverage and adequate reimbursement for any approved drug which could impact our ability to monetize our existing biotechnology assets; |
| ● | market conditions in the pharmaceutical and life sciences sectors; |
| ● | market conditions in the global iGaming sector; |
| ● | regulatory developments in the global iGaming sector, including but not limited to the U.S., Canada and other foreign jurisdictions where the Company does or may seek to do business; |
| ● | changes in taxation regimes in respect of iGaming specifically, and flows of capital more generally, which may impact the ability of the Company to operate, or operate economically, in one or all such locations; |
| ● | general economic, political, and market conditions and overall fluctuations in the financial markets in the U.S. and abroad; and |
| ● | investors’ general perception of us and our business. |
Our common stock is listed on the Nasdaq Capital Market under the symbol “ATNF.” Our stock price may be impacted by factors that are unrelated or disproportionate to our operating performance. The stock markets in general have experienced extreme volatility that has often been unrelated to the operating performance of particular companies. These broad market fluctuations may adversely affect the trading price of our common stock. Additionally, general economic, political and market conditions, such as recessions, inflation, war, interest rates or international currency fluctuations may adversely affect the market price of our common stock. Due to the limited volume of our shares which trade, we believe that our stock prices (bid, ask and closing prices) may not be related to our actual value, and not reflect the actual value of our common stock. You should exercise caution before making an investment in us.
73
Stock markets in general and our stock price in particular have recently experienced extreme price and volume fluctuations that have often been unrelated or disproportionate to the operating performance of those companies and our company. For example, during 2022, the closing sales prices of our common stock ranged from a post-split adjusted high of $1,482.04 per share to a low of $23.56 per share, during fiscal 2023, the closing sales prices of our common stock ranged from a high of $100.70 per share to a low of $3.21 per share and during fiscal 2024, the closing sales prices of our common stock ranged from a post-split adjusted high of $6.47 per share to a low of $1.18 per share. During this time, we do not believe that we have experienced any material changes in our financial condition or results of operations that would explain such price volatility or trading volume; however, we have sold equity which was dilutive to existing stockholders. These broad market fluctuations may adversely affect the trading price of our securities. Additionally, these and other external factors have caused and may continue to cause the market price and demand for our common stock to fluctuate substantially, which may limit or prevent our stockholders from readily selling their shares of our common stock and may otherwise negatively affect the liquidity of our common stock.
Additionally, as a result of the illiquidity of our common stock, investors may not be interested in owning our common stock because of the inability to acquire or sell a substantial block of our common stock at one time. Such illiquidity could have an adverse effect on the market price of our common stock. In addition, a shareholder may not be able to borrow funds using our common stock as collateral because lenders may be unwilling to accept the pledge of securities having such a limited market. An active trading market for our common stock may not develop or, if one develops, may not be sustained.
In the past, many companies that have experienced volatility in the market price of their stock have been subject to securities class action litigation. We may be the target of this type of litigation in the future. Securities litigation against us could result in substantial costs and divert our management’s attention from other business concerns, which could seriously harm our business.
Elray Resources, Inc., beneficially owns a significant percentage of our common stock and as such exercises significant voting control over us, which limits other stockholders’ abilities to influence corporate matters and could delay or prevent a change in corporate control.
Elray beneficially owns 1,318,000 shares of common stock, 25.4% of our total voting shares, and also holds warrants to purchase up to 3,000,000 shares of common stock with an exercise price of $1.68 per share, which if exercised for cash, would result in Elray holding 4,318,000 voting shares, or 52.8% of our then total voting shares. As a result, Elray has significant influence on the stockholder vote. Consequently, it has the ability to influence matters affecting our stockholders and therefore exercise significant control in determining the outcome of a number of corporate transactions or other matters, including (i) making amendments to our certificate of incorporation; (ii) whether to approve the issuance additional shares of common stock and preferred stock, including to itself; (iii) election of directors; and (iv) any merger or significant corporate transactions, including with itself or other related parties. As a potential investor in the Company, you should keep in mind that even if you own shares of our common stock and wish to vote them at annual or special stockholder meetings, your shares will have little effect on the outcome of corporate decisions. The interests of Elray may not coincide with our interests or the interests of other stockholders.
Mr. Jay Goodman, a director of the Company, is the adult son of Mr. Anthony Brian Goodman, who serves as the Chief Executive Officer of Elray and is deemed to beneficially own the securities held by Elray in the Company.
Elray may have interests, with respect to its Company securities, that are different from other investors and the concentration of voting power held by Elray may have an adverse effect on the price of our common stock.
In addition, this concentration of ownership might adversely affect the market price of our common stock by: (1) delaying, deferring or preventing a change of control of our Company; (2) impeding a merger, consolidation, takeover or other business combination involving our Company; or (3) discouraging a potential acquirer from making a tender offer or otherwise attempting to obtain control of our Company.
74
Information available in public media that is published by third parties, including blogs, articles, message boards and social and other media may include statements not attributable to us and may not be reliable or accurate.
We are aware of a large volume of information being disseminated by third parties relating to our operations, including in blogs, message boards and social and other media. Such information as reported by third parties may not be accurate, may lead to significant volatility in our securities and may ultimately result in our common stock or other securities declining in value.
The exercise of the outstanding options and warrants, and the sale of common stock upon exercise thereof, may adversely affect the trading price of our securities.
As of the date of this Report, we had (a) 21,564 shares of common stock issuable upon the exercise of outstanding stock options, with a weighted average exercise price of $23.81 per share; (b) 15,794 shares of common stock issuable upon the exercise of outstanding public warrants exercisable at an exercise price of $4,370.00 per share, (c) 6,748 shares of common stock issuable upon the exercise of certain outstanding private placement warrants at an exercise price of $1,900.00 per share, (d) 6,579 shares of common stock issuable upon the exercise of certain outstanding private placement warrants at an exercise price of $2,850.00 per share, (e) 4,049 shares of common stock issuable upon exercise of certain outstanding warrants to purchase shares of common stock with an exercise price of $3.23 per share, (f) 3,000,000 shares of common stock issuable upon the exercise of warrants to purchase 3,000,000 shares of common stock with an exercise price of $1.68 per share, (g) 168 shares of common stock issuable upon exercise of warrants with an exercise price of $2,006.40 per share, (h) 1,900,138 shares of common stock issuable upon exercise of warrants to purchase shares of common stock with an exercise price of $1.50 per share; and (i) 1,200,000 shares of common stock issuable upon exercise of warrants to purchase shares of common stock with an exercise price of $2.28 per share. For the life of the options and warrants, the holders have the opportunity to profit from a rise in the market price of our common stock without assuming the risk of ownership. The issuance of shares upon the exercise of outstanding securities will also dilute the ownership interests of our existing stockholders.
The availability of these shares for public resale, as well as any actual resales of these shares, could adversely affect the trading price of our common stock. We cannot predict the size of future issuances of our common stock pursuant to the exercise of outstanding options or warrants or conversion of other securities, or the effect, if any, that future issuances and sales of shares of our common stock may have on the market price of our common stock. Sales or distributions of substantial amounts of our common stock (including shares issued in connection with an acquisition), or the perception that such sales could occur, may cause the market price of our common stock to decline.
In addition, the common stock issuable upon exercise/conversion of outstanding convertible securities may represent overhang that may also adversely affect the market price of our common stock. Overhang occurs when there is a greater supply of a company’s stock in the market than there is demand for that stock. When this happens the price of our stock will decrease, and any additional shares which stockholders attempt to sell in the market will only further decrease the share price. If the share volume of our common stock cannot absorb shares sold by holders of our outstanding convertible securities, then the value of our common stock will likely decrease.
Additionally, the warrants to purchase 3,000,000 shares of common stock which we granted effective September 30, 2024, which have an exercise price of $1.68 per share and a term of seven years, have cashless exercise rights, which means that such warrants can be exercised without paying us any cash and instead with the holder netting the value of the exercise price against the value of the shares issuable upon exercise thereof. Additionally, any shares of common stock issuable upon the cashless exercise of such warrants will likely be able to tack the holding period of such shares of common stock with the warrants.
The influx of any of these shares into the public market could potentially have a negative effect on the trading price of our common stock.
Our outstanding public warrants are significantly out of the money.
Each Public Warrant entitles the holder to purchase one-seven hundred sixtieth of one share of common stock at an exercise price of $5.75 per 1/760th of one share ($4,370.00 per whole share), subject to adjustment. No fractional shares will be issued upon exercise of the Public Warrants. The Public Warrants became exercisable 12 months from the closing of the IPO and expire five years after the completion of the Business Combination (November 6, 2025). The Public Warrants are significantly out of the money and because no fractional shares will be issued upon exercise of the Public Warrants, the Public Warrants are only exercisable in multiples of 760. As a result, the Public Warrants may not have any significant value. Additionally, warrant holders not holding at least 760 Public Warrants or who hold Public Warrants which would be exercisable for a fractional share of common stock, must sell any warrants to obtain value from the fractional interest. As a result, the trading of the Public Warrants may be limited or sporadic, and such Public Warrants may not have any significant value. Any holder of Public Warrants holding less than 760 Public Warrants or a number of Public Warrants not evenly divisible by 760 will not receive any common stock upon the exercise of Public Warrants, as no fractional shares of common stock are issuable upon exercise thereof.
75
Provisions of certain outstanding warrants could discourage an acquisition of us by a third party.
Provisions of certain outstanding warrants could make it more difficult or expensive for a third party to acquire us. Certain outstanding warrants prohibit us from engaging in certain transactions constituting “fundamental transactions” unless, among other things, the surviving entity assumes our obligations under each of the outstanding warrants to purchase up to 4,049 shares of common stock with an exercise price of $3.23 per share (warrants to purchase 4,049 shares of common stock), $1,900 per share (warrants to purchase 6,748 shares of common stock) and $2,850 per share (warrants to purchase 6,579 shares of common stock). Further, such outstanding warrants provide that, in the event of certain transactions constituting “fundamental transactions,” with some exception, holders of such warrants will have the right, at their option, to require us to repurchase such warrants at a price described in the applicable warrants (based on the Black Scholes Value of such warrants). These and other provisions of the warrants could prevent or deter a third party from acquiring us even where the acquisition could be beneficial to stockholders.
A significant number of our shares are eligible for sale and their sale or potential sale may depress the market price of our common stock and cause significant dilution to existing stockholders.
Sales of a significant number of shares of our common stock in the public market could harm the market price of our common stock. Most of our common stock is available for immediate resale in the public market, including (a) 21,564 shares of common stock issuable upon the exercise of outstanding stock options, with a weighted average exercise price of $23.81 per share; (b) 15,794 shares of common stock issuable upon the exercise of outstanding public warrants exercisable at an exercise price of $4,370.00 per share, (c) 6,748 shares of common stock issuable upon the exercise of certain outstanding private placement warrants at an exercise price of $1,900.00 per share, (d) 6,579 shares of common stock issuable upon the exercise of certain outstanding private placement warrants at an exercise price of $2,850.00 per share, (e) 4,049 shares of common stock issuable upon exercise of certain outstanding warrants to purchase shares of common stock with an exercise price of $3.23 per share, (f) 3,000,000 shares of common stock issuable upon the exercise of warrants to purchase 3,000,000 shares of common stock with an exercise price of $1.68 per share, (g) 168 shares of common stock issuable upon exercise of warrants with an exercise price of $2,006.40 per share, (h) 1,900,138 shares of common stock issuable upon exercise of warrants to purchase shares of common stock with an exercise price of $1.50 per share; and (i) 1,200,000 shares of common stock issuable upon exercise of warrants to purchase shares of common stock with an exercise price of $2.28 per share. If a significant number of shares were sold, such sales would increase the supply of our common stock, thereby potentially causing a decrease in its price. Some or all of our shares of common stock may be offered from time to time in the open market pursuant to effective registration statements and/or compliance with Rule 144, which sales could have a depressive effect on the market for our shares of common stock. Subject to certain restrictions, a person who has held restricted shares for a period of six months may generally sell common stock into the market. The sale of a significant portion of such shares when such shares are eligible for public sale may cause the value of our common stock to decline in value.
There may not be sufficient liquidity in the market for our securities in order for investors to sell their shares. The market price of our common stock may continue to be volatile.
The market price of our common stock will likely continue to be highly volatile. Some of the factors that may materially affect the market price of our common stock are beyond our control, such as conditions or trends in the industry in which we operate or sales of our common stock. This situation is attributable to a number of factors, including the fact that we are a small company which is relatively unknown to stock analysts, stock brokers, institutional investors and others in the investment community that generate or influence sales volume, and that even if we came to the attention of such persons, they tend to be risk-averse and would be reluctant to follow an unproven company such as ours or purchase or recommend the purchase of our shares until such time as we became more seasoned and viable.
76
As a consequence, there may be periods of several days or more when trading activity in our shares is minimal or non-existent, as compared to a mature issuer which has a large and steady volume of trading activity that will generally support continuous sales without an adverse effect on share price. It is possible that a broader or more active public trading market for our common stock will not develop or be sustained, or that trading levels will not continue. These factors may materially adversely affect the market price of our common stock, regardless of our performance. In addition, the public stock markets have experienced extreme price and trading volume volatility. This volatility has significantly affected the market prices of securities of many companies for reasons frequently unrelated to the operating performance of the specific companies. These broad market fluctuations may adversely affect the market price of our common stock.
We face significant penalties and damages in the event registration statements we have previously filed to register certain securities sold in our prior offerings are subsequently suspended or terminated.
Pursuant to certain prior private offerings of securities, we entered into registration rights agreements which required us to file certain registration statements to register the resale of the privately sold shares and certain securities issuable upon exercise/conversion thereof, and to maintain the effectiveness of such registration statements for certain periods of time. To date, all such required registration statements have been declared effective by the SEC. However, in the event the registration statements are subsequently suspended or terminated, or we otherwise fail to meet certain requirements set forth in the registration rights agreements, we could be required to pay significant penalties which could adversely affect our cash flow and cause the value of our securities to decline in value.
The exercise of such outstanding options or warrants or the conversion of outstanding convertible securities will result in further dilution of your investment. If our existing stockholders sell substantial amounts of our common stock in the public market, or if the public perceives that such sales could occur, this could have an adverse impact on the market price of our common stock, even if there is no relationship between such sales and the performance of our business.
Resales of our common stock in the public market may cause the market price of our common stock to fall.
Sales of a substantial number of shares of our common stock could occur at any time. The issuance of new shares of our common stock could result in resales of our common stock by our current stockholders concerned about the potential ownership dilution of their holdings. In turn, these resales could have the effect of depressing the market price for our common stock.
Future sales of our common stock could cause our stock price to decline.
If our stockholders sell substantial amounts of our common stock in the public market, the market price of our common stock could decrease significantly. The perception in the public market that our stockholders might sell shares of our common stock could also depress the market price of our common stock. Up to $125,000,000 in total aggregate value of securities have been registered by us on a “shelf” registration statement on Form S-3 that we filed with the Commission on June 3, 2022, and which was declared effective on June 24, 2022. However, as of the date of this Report, our public float is less than $75 million, and under SEC regulations for so long as our public float remains less than $75 million, the amount we can raise through primary public offerings of securities in any twelve-month period using our shelf registration statement on Form S-3 is limited to an aggregate of one-third of our public float. At such time as our public float again exceeds $75 million, the number of securities we may sell under a Form S-3 registration statement will no longer be limited by such rules. Additionally, if our existing stockholders sell, or indicate an intention to sell, substantial amounts of our common stock in the public market, the trading price of our common stock could decline significantly. The market price for shares of our common stock may drop significantly when such securities are sold in the public markets. A decline in the price of shares of our common stock might impede our ability to raise capital through the issuance of additional shares of our common stock or other equity securities.
77
As of March 24, 2025, we had 3,837,780 shares of common stock outstanding, all of which shares were, and continue to be, eligible for sale in the public market, subject in some cases to compliance with the requirements of Rule 144, including the volume limitations and manner of sale requirements and lock-up agreements.
Risks Associated with Our Governing Documents and Delaware Law
Our Certificate of Incorporation provides for indemnification of officers and directors at our expense and limits their liability, which may result in a major cost to us and hurt the interests of our stockholders because corporate resources may be expended for the benefit of officers or directors.
Our Certificate of Incorporation provides for indemnification as follows: “To the fullest extent permitted by applicable law, the Corporation is authorized to provide indemnification of, and advancement of expenses to, such agents of the Corporation (and any other persons to which Delaware law permits the Corporation to provide indemnification) through Bylaw provisions, agreements with such agents or other persons, vote of stockholders or disinterested directors or otherwise, in excess of the indemnification and advancement otherwise permitted by Section 145 of the Delaware General Corporation Law, subject only to limits created by applicable Delaware law (statutory or non-statutory), with respect to actions for breach of duty to the Corporation, its stockholders and others.”
We have been advised that, in the opinion of the SEC, indemnification for liabilities arising under federal securities laws is against public policy as expressed in the Securities Act, and is therefore, unenforceable. In the event that a claim for indemnification for liabilities arising under federal securities laws, other than the payment by us of expenses incurred or paid by a director, officer or controlling person in the successful defense of any action, suit or proceeding, is asserted by a director, officer or controlling person in connection with our activities, we will (unless in the opinion of our counsel, the matter has been settled by controlling precedent) submit to a court of appropriate jurisdiction, the question whether indemnification by us is against public policy as expressed in the Securities Act and will be governed by the final adjudication of such issue. The legal process relating to this matter if it were to occur is likely to be very costly and may result in us receiving negative publicity, either of which factors is likely to materially reduce the market and price for our shares.
Our Certificate of Incorporation contains a specific provision that limits the liability of our directors for monetary damages to us and our stockholders and requires us, under certain circumstances, to indemnify officers, directors and employees.
The limitation of monetary liability against our directors, officers and employees under Delaware law and the existence of indemnification rights to them may result in substantial expenditures by us and may discourage lawsuits against our directors, officers and employees.
Our Certificate of Incorporation contains a specific provision that limits the liability of our directors for monetary damages to us and our stockholders. We also have contractual indemnification obligations under our employment and engagement agreements with our executive officers and directors. The foregoing indemnification obligations could result in us incurring substantial expenditures to cover the cost of settlement or damage awards against our directors and officers, which we may be unable to recoup. These provisions and resultant costs may also discourage us from bringing a lawsuit against our directors and officers for breaches of their fiduciary duties and may similarly discourage the filing of derivative litigation by our stockholders against our directors and officers, even though such actions, if successful, might otherwise benefit us and our stockholders.
78
Our directors have the right to authorize the issuance of shares of preferred stock and additional shares of our common stock.
Our directors, within the limitations and restrictions contained in our Certificate of Incorporation and without further action by our stockholders, have the authority to issue shares of preferred stock from time to time in one or more series and to fix the number of shares and the relative rights, conversion rights, voting rights, and terms of redemption, liquidation preferences and any other preferences, special rights and qualifications of any such series. Any issuance of shares of preferred stock could adversely affect the rights of holders of our common stock. Should we issue additional shares of our common stock at a later time, each investor’s ownership interest in our stock would be proportionally reduced.
Anti-takeover provisions in our Second Amended and Restated Certificate of Incorporation, as amended, and our Second Amended and Restated Bylaws, as well as provisions of Delaware law, might discourage, delay or prevent a change in control of our company or changes in our management and, therefore, depress the trading price of our common stock.
Our Second Amended and Restated Certificate of Incorporation, as amended and our Second Amended and Restated Bylaws and Delaware law contain provisions that may discourage, delay or prevent a merger, acquisition or other change in control that stockholders may consider favorable, including transactions in which you might otherwise receive a premium for your shares of our common stock or warrants. These provisions may also prevent or delay attempts by our stockholders to replace or remove our management. Our corporate governance documents include the following provisions:
| ● | a classified board of directors, as a result of which our Board is divided into two classes, with each class serving for staggered two-year terms; |
| ● | the removal of directors only for cause; |
| ● | requiring advance notice of stockholder proposals for business to be conducted at meetings of our stockholders and for nominations of candidates for election to our Board; |
| ● | prohibiting stockholders’ ability to take action via written consents to action; |
| ● | providing that a special meeting of stockholders may be called only by the Chairman of the Board, Chief Executive Officer, or the Board pursuant to a resolution adopted by a majority of the Board; |
| ● | authorizing blank check preferred stock, which could be issued with voting, liquidation, dividend and other rights superior to our common stock; and |
| ● | limiting the liability of, and providing indemnification to, our directors and officers. |
As a Delaware corporation, we are also subject to provisions of Delaware law, including Section 203 of the Delaware General Corporation Law, which limits the ability of stockholders holding shares representing more than 15% of the voting power of our outstanding voting stock from engaging in certain business combinations with us. Any provision of our Second Amended and Restated Certificate of Incorporation, as amended or our Second Amended and Restated Bylaws or Delaware law that has the effect of delaying or deterring a change in control could limit the opportunity for our stockholders to receive a premium for their shares of our common stock and could also affect the price that some investors are willing to pay for our common stock.
The existence of the foregoing provisions and anti-takeover measures could limit the price that investors might be willing to pay in the future for shares of our common stock or warrants. They could also deter potential acquirers of our company, thereby reducing the likelihood that you could receive a premium for your common stock or warrants in an acquisition.
79
Our Second Amended and Restated Certificate of Incorporation, as amended, contains exclusive forum provisions that may discourage lawsuits against us and our directors and officers.
Our Second Amended and Restated Certificate of Incorporation, as amended provides that unless the corporation consents in writing to the selection of an alternative forum, the Court of Chancery of the State of Delaware, will be the sole and exclusive forum for (i) any derivative action or proceeding brought on behalf of us, (ii) any action asserting a claim for breach of a fiduciary duty owed by any current or former director, officer, employee or stockholder of the Company to us or our stockholders, (iii) any action asserting a claim arising pursuant to any provision of the Delaware General Corporation Law, our Second Amended and Restated Certificate of Incorporation or Bylaws, or (iv) any action asserting a claim governed by the internal affairs doctrine.
The choice of forum provision in our Second Amended and Restated Certificate of Incorporation does not waive our compliance with our obligations under the federal securities laws and the rules and regulations thereunder. Moreover, the provision does not apply to suits brought to enforce a duty or liability created by the Exchange Act or by the Securities Act. Section 27 of the Exchange Act creates exclusive federal jurisdiction over all suits brought to enforce any duty or liability created by the Exchange Act or the rules and regulations thereunder, and Section 22 of the Securities Act creates concurrent jurisdiction for federal and state courts with respect to suits brought to enforce a duty or liability created by the Securities Act or the rules and regulations thereunder. Accordingly, both state and federal courts have jurisdiction to entertain claims under the Securities Act.
These exclusive forum provisions may limit the ability of our stockholders to bring a claim in a judicial forum that such stockholders find favorable for disputes with us or our directors or officers, which may discourage such lawsuits against us and our directors and officers. Alternatively, if a court were to find one or more of these exclusive forum provisions inapplicable to, or unenforceable in respect of, one or more of the specified types of actions or proceedings described above, we may incur additional costs associated with resolving such matters in other jurisdictions or forums, which could materially and adversely affect our business, financial condition or results of operations.
Our Second Amended and Restated Certificate of Incorporation, as amended, contains provisions whereby we renounced any interest in any corporate opportunity offered to any director or officer, subject to certain exceptions.
Our Section Amended and Restated Certificate of Incorporation, as amended, provides that to the extent allowed by law, the doctrine of corporate opportunity, or any other analogous doctrine, does not apply with respect to us or any of our officers or directors, or any of their respective affiliates, and that we renounce any expectancy that any of our directors or officers will offer any such corporate opportunity of which he or she may become aware to us, except that the doctrine of corporate opportunity shall apply with respect to any of our directors or officers only with respect to a corporate opportunity (i) that was offered to such person solely in his or her capacity as our director or officer, (ii) that is one we are legally and contractually permitted to undertake and would otherwise be reasonable for us to pursue, and (iii) to the extent the director or officer is permitted to refer such opportunity to us without violating any legal obligation.
Additionally, each of our officers and directors presently has, and any of them in the future may have, additional fiduciary or contractual obligations to other entities pursuant to which such officer or director may be required to present a business opportunity to such entity, subject to his or her fiduciary duties under applicable law. Accordingly, there may arise conflicts of interest in whether to present a potential business combination opportunity to our company. These conflicts may not be resolved in our favor. Our renouncement of corporate opportunities may have a material adverse effect on our results of operations moving forward and/or create conflicts of interest or perceived conflicts of interest which may have a material adverse effect on the value of our securities.
80
Provisions in our Certificate of Incorporation and Delaware law may inhibit a takeover of us, which could limit the price investors might be willing to pay in the future for our common stock and could entrench management.
Our Certificate of Incorporation contains provisions that may discourage unsolicited takeover proposals that stockholders may consider to be in their best interests. These provisions include a staggered board of directors and the ability of our Board to designate the terms of and issue new series of preferred shares, which may make it more difficult for the removal of management and may discourage transactions that otherwise could involve payment of a premium over prevailing market prices for our securities. We are also subject to anti-takeover provisions under Delaware law, which could delay or prevent a change of control of the Company. Together, these provisions may make it more difficult for the removal of management and may discourage transactions that otherwise could involve payment of a premium over prevailing market prices for our securities.
Compliance, Reporting and Listing Risks
We incur significant costs to ensure compliance with U.S. and Nasdaq reporting and corporate governance requirements.
We incur significant costs associated with our public company reporting requirements and with applicable U.S. and Nasdaq corporate governance requirements, including requirements under the Sarbanes-Oxley Act of 2002 and other rules implemented by the SEC and Nasdaq. The rules of Nasdaq include requiring us to maintain independent directors, comply with other corporate governance requirements and pay annual listing and stock issuance fees. All of such SEC and Nasdaq obligations require a commitment of additional resources including, but not limited, to additional expenses, and may result in the diversion of our senior management’s time and attention from our day-to-day operations. We expect all of these applicable rules and regulations to significantly increase our legal and financial compliance costs and to make some activities more time consuming and costly. We also expect that these applicable rules and regulations may make it more difficult and more expensive for us to obtain director and officer liability insurance and we may be required to accept reduced policy limits and coverage or incur substantially higher costs to obtain the same or similar coverage. As a result, it may be more difficult for us to attract and retain qualified individuals to serve on our Board or as executive officers.
We incur increased costs as a result of being a reporting company, and given our limited capital resources, such additional costs may have an adverse impact on our profitability.
We are an SEC-reporting company. The rules and regulations under the Exchange Act require reporting companies to provide periodic reports with interactive data files, which require that we engage legal, accounting and auditing professionals, and eXtensible Business Reporting Language (XBRL) and EDGAR (Electronic Data Gathering, Analysis, and Retrieval) service providers. The engagement of such services can be costly, and we may continue to incur additional losses, which may adversely affect our ability to continue as a going concern. In addition, the Sarbanes-Oxley Act of 2002, as well as a variety of related rules implemented by the SEC, have required changes in corporate governance practices and generally increased the disclosure requirements of public companies. For example, as a result of being a reporting company, we are required to file periodic and current reports and other information with the SEC, and we have adopted policies regarding disclosure controls and procedures and regularly evaluate those controls and procedures.
The additional costs we continue to incur in connection with being a reporting company (expected to be several hundred thousand dollars per year) will continue to further stretch our limited capital resources. Due to our limited resources, we have to allocate resources away from other productive uses in order to continue to comply with our obligations as an SEC reporting company. Further, there is no guarantee that we will have sufficient resources to continue to meet our reporting and filing obligations with the SEC as they come due.
In the past we have not been in compliance with Nasdaq’s continued listing standards and may not be in compliance with such standards in the future, and are currently subject to a one year mandatory panel monitor, and as a result our common stock and public warrants may be delisted from Nasdaq.
Our common stock and public warrants trade on Nasdaq under the symbols “ATNF” and “ATNFW,” respectively. Notwithstanding such listing, there can be no assurance any broker will be interested in trading our securities. Therefore, it may be difficult to sell our securities publicly. There is also no guarantee that we will be able to maintain our listings on Nasdaq for any period of time by perpetually satisfying Nasdaq’s continued listing requirements.
81
We have previously been out of compliance with Nasdaq’s continued listing requirements due to our failure to maintain a minimum bid price of at least $1.00 per share for our common stock, which failure was remedied in March 2024.
Also, on October 11, 2023, the Company received written notice from Nasdaq notifying the Company that it was not in compliance with the shareholder approval requirements set forth in Nasdaq Listing Rule 5635(d), which require prior shareholder approval for transactions, other than public offerings, involving the issuance of 20% or more of the pre-transaction shares outstanding at less than the applicable Minimum Price (as defined in Listing Rule 5635(d)(1)(A)), which non-compliance we remedied in December 2023.
Additionally, on November 15, 2023, the Company received a letter from Nasdaq notifying the Company that it was not in compliance with the minimum stockholders’ equity requirement for continued listing on the Nasdaq Capital Market. Nasdaq Listing Rule 5550(b)(1) (the “Equity Rule”) requires companies listed on the Nasdaq Capital Market to maintain stockholders’ equity of at least $2,500,000. In the Company’s Quarterly Report on Form 10-Q for the quarter ended September 30, 2023, the Company reported a stockholders’ deficit of ($149,327), which was below the minimum stockholders’ equity required for continued listing pursuant to the Equity Rule. Additionally, the Company did not meet the alternative Nasdaq continued listing standards under Nasdaq Listing Rules and did not meet the minimum stockholders’ equity requirement as of December 31, 2023, March 31, 2024, or June 30, 2024.
Nasdaq provided the Company until January 2, 2024 to submit to Nasdaq a plan to regain compliance. We submitted the plan to regain compliance in a timely manner, and on January 11, 2024, Nasdaq advised the Company that it has determined to grant the Company an extension to regain compliance with the Equity Rule, subject to certain requirements, including completing the transactions outlined in the plan of compliance which were intended to increase the Company’s stockholders’ equity to more than $2.5 million.
While the Company was able to undertake some of the transactions described in the compliance plan, it was unable to regain compliance with the Equity Rule prior to the end of the plan period (May 13, 2024). As a result, on May 14, 2024, the Company received a delist determination letter from the Listing Qualifications department of The Nasdaq Stock Market LLC (the “Staff”) advising the Company that the Staff had determined that the Company did not meet the terms of the extension. On May 17, 2024, the Company requested an appeal of the Staff’s delisting determination, and on May 20, 2024, the Staff advised the Company that the delisting action referenced in the Staff’s determination letter had been stayed, pending a final written decision by the Nasdaq Hearings Panel (“Panel”). On July 2, 2024, the Company received notice that the Panel had determined to grant the Company’s request to continue its listing on The Nasdaq Stock Market, subject to the Company meeting certain conditions, including filing on or before July 31, 2024, a public disclosure describing the transactions undertaken by the Company to achieve compliance with Nasdaq’s continued listing rules and demonstrate long-term compliance with the Equity Rule and providing an indication of its equity following those transactions. On July 22, 2024, the Panel granted the Company’s request for additional time to achieve compliance with Nasdaq’s continued listing rules and demonstrate long-term compliance with the Equity Rule. Specifically, the Hearings Panel subsequently extended that deadline to September 30, 2024, and further to October 15, 2024, to regain compliance with the Equity Rule and to allow the continued listing of the Company’s common stock and warrants on The Nasdaq Stock Market through such date, subject to the Company’s compliance with the Equity Rule on or prior to such date.
As a result of the acquisition of the Purchased Assets (discussed above under Item 1. Business), issuance of the 1,000,000 shares of Series B Convertible Preferred Stock and Purchase Warrants as described above, on October 4, 2024, we received a letter from Nasdaq confirming that we had regained compliance with the Equity Rule. Nasdaq also advised the Company that in application of Listing Rule 5815(d)(4)(B), the Company is subject to a mandatory panel monitor for a period of one year from the date of such letter (October 4, 2025). If, within that one-year monitoring period, the Staff finds that the Company is no longer in compliance with the Equity Rule, then, notwithstanding Rule 5810(c)(2), the Company will not be permitted to provide the Staff with a plan of compliance with respect to such deficiency and the Staff will not be permitted to grant additional time for the Company to regain compliance with respect to such deficiency, nor will the Company be afforded an applicable cure or compliance period pursuant to Rule 5810(c)(3). Instead, the Staff will issue a Delist Determination Letter and the Company will have an opportunity to request a new hearing with the initial Panel or a newly convened Hearings Panel if the initial Panel is unavailable. The Company will have the opportunity to respond/present to the Hearings Panel as provided by Listing Rule 5815(d)(4)(C) and the Company’s securities may at that time be delisted from Nasdaq.
82
If the Company’s common stock and public warrants are delisted, it could be more difficult to buy or sell the Company’s common stock and public warrants or to obtain accurate quotations, and the price of the Company’s common stock and public warrants could suffer a material decline. Delisting could also impair the Company’s ability to raise capital and/or trigger defaults and penalties under outstanding agreements or securities of the Company.
Separately, on May 14, 2024, the Staff provided us notice of our non-compliance with the audit committee requirements for continued listing on Nasdaq set forth in Listing Rule 5605(c)(2), which requires that listed companies maintain an audit committee of at least three independent directors. Nasdaq provided the Company a cure period in order to regain compliance, and we regained compliance with this listing rule in December 2024.
Among the conditions required for continued listing on The Nasdaq Capital Market, Nasdaq requires us to maintain at least $2.5 million in stockholders’ equity or $500,000 in net income over the prior two years or two of the prior three years. As of September 30, 2023, December 31, 2023, March 31, 2024, and June 30, 2024, our stockholders’ equity was below $2.5 million and we did not otherwise meet the net income requirements described above, and as such, we were not in compliance with Nasdaq’s continued listing standards relating to minimum stockholders’ equity until September 30, 2024.
Our failure to meet Nasdaq’s continued listing requirements for the reasons above, or any other reason, may result in our securities being delisted from Nasdaq.
Additional conditions required for continued listing on Nasdaq include requiring that we have a majority of independent directors, a two-person compensation committee and a three-member audit committee (each consisting of all independent directors), which three-person audit committee requirement we have not met from time to time in the past. As a result, our common stock and public warrants may be delisted from Nasdaq.
We also have to continue to meet other objective and subjective listing requirements to continue to be listed on The Nasdaq Capital Market. Delisting from The Nasdaq Capital Market could make trading our common stock and public warrants more difficult for investors, potentially leading to declines in our share price and liquidity. Without a Nasdaq Capital Market listing, stockholders may have a difficult time getting a quote for the sale or purchase of our common stock and public warrants, the sale or purchase of our common stock and public warrants would likely be made more difficult, and the trading volume and liquidity of our common stock and public warrants could decline. Delisting from The Nasdaq Capital Market could also result in negative publicity and could also make it more difficult for us to raise additional capital. The absence of such a listing may adversely affect the acceptance of our common stock as currency or the value accorded by other parties. Further, if we are delisted, we would also incur additional costs under state blue sky laws in connection with any sales of our securities. These requirements could severely limit the market liquidity of our common stock and public warrants and the ability of our stockholders and warrant holders to sell our common stock and public warrants in the secondary market. If our common stock and public warrants are delisted by Nasdaq, our common stock and public warrants may be eligible to trade on an over-the-counter quotation system, such as the OTCQB Market or the OTC Pink market, where an investor may find it more difficult to sell our common stock and public warrants or obtain accurate quotations as to the market value of our common stock and public warrants. In the event our common stock and public warrants are delisted from The Nasdaq Capital Market, we may not be able to list our common stock on another national securities exchange or obtain quotation on an over-the counter quotation system.
83
General Risk Factors
Global economic conditions could materially adversely affect our business, results of operations, financial condition and growth.
Adverse macroeconomic conditions, including inflation, slower growth or recession, new or increased tariffs, changes to fiscal and monetary policy, tighter credit, higher interest rates, high unemployment and currency fluctuations, as well as the potential implications of a Congressional impasse over the U.S. debt limit, possible future U.S. governmental shutdowns over budget disagreements and/or the impact of changes in tariff regimes globally, could materially adversely affect our operations, expenses, access to capital and the market for our planned future products. In addition, consumer confidence and spending could be adversely affected in response to financial market volatility, negative financial news, conditions in the real estate and mortgage markets, declines in income or asset values, changes to fuel and other energy costs, labor and healthcare costs and other economic factors.
In addition, uncertainty about, or a decline in, global or regional economic conditions could have a significant impact on our funding sources, suppliers and partners. Potential effects include financial instability; inability to obtain credit to finance operations and purchases of our future planned products; and insolvency.
A downturn in the economic environment could also lead to limitations on our ability to sell equity or issue new debt; reduce liquidity; and result in declines in the fair value of our financial instruments. These and other economic factors could materially adversely affect our business, results of operations, financial condition and growth.
Our proprietary information, or that of our customers, suppliers and business partners, may be lost or we may suffer security breaches.
In the ordinary course of our business, we expect to collect and store sensitive data, including valuable and commercially sensitive intellectual property, clinical trial data, our proprietary business information and that of our future customers, suppliers and business partners, and personally identifiable information of our customers, clinical trial subjects and employees, patients, in our data centers and on our networks. The secure processing, maintenance and transmission of this information is critical to our operations. Despite our security measures, our information technology and infrastructure may be vulnerable to attacks by hackers or breached due to employee error, malfeasance or other disruptions. Any such breach could compromise our networks and the information stored there could be accessed, publicly disclosed, lost or stolen. Any such access, disclosure or other loss of information could result in legal claims or proceedings, liability under laws that protect the privacy of personal information, regulatory penalties, disrupt our operations, damage our reputation, and cause a loss of confidence in our products and our ability to conduct clinical trials, which could adversely affect our business and reputation and lead to delays in gaining regulatory approvals for our future product candidates. Although we maintain business interruption insurance coverage, our insurance might not cover all losses from any future breaches of our systems.
84
Failure of our information technology systems, including cybersecurity attacks or other data security incidents, could significantly disrupt the operation of our business.
Our business increasingly depends on the use of information technologies, which means that certain key areas such as research and development, production and sales are to a large extent dependent on our information systems or those of third-party providers. Our ability to execute our business plan and to comply with regulators’ requirements with respect to data control and data integrity, depends, in part, on the continued and uninterrupted performance of our information technology systems, or internet technology (IT) systems and the IT systems supplied by third-party service providers. As information systems and the use of software and related applications by our company, our business partners, suppliers, and customers become more cloud-based, there has been an increase in global cybersecurity vulnerabilities and threats, including more sophisticated and targeted cyber-related attacks that pose a risk to the security of our information systems and networks and the confidentiality, availability and integrity of data and information. In addition, our IT systems are vulnerable to damage from a variety of sources, including telecommunications or network failures, malicious human acts and natural disasters. Moreover, despite network security and backup measures, some of our servers are potentially vulnerable to physical or electronic break-ins, computer viruses and similar disruptive problems. Despite the precautionary measures we and our third-party service providers have taken to prevent unanticipated problems that could affect our IT systems, a successful cybersecurity attack or other data security incident could result in the misappropriation and/or loss of confidential or personal information, create system interruptions, or deploy malicious software that attacks our systems. It is also possible that a cybersecurity attack might not be noticed for some period of time. In addition, sustained or repeated system failures or problems arising during the upgrade of any of our IT systems that interrupt our ability to generate and maintain data, and in particular to operate our proprietary technology platform, could adversely affect our ability to operate our business. The occurrence of a cybersecurity attack or incident could result in business interruptions from the disruption of our IT systems, or negative publicity resulting in reputational damage with our stockholders and other stakeholders and/or increased costs to prevent, respond to or mitigate cybersecurity events. In addition, the unauthorized dissemination of sensitive personal information or proprietary or confidential information could expose us or other third–parties to regulatory fines or penalties, litigation and potential liability, or otherwise harm our business.
We may acquire other companies which could divert our management’s attention, result in additional dilution to our stockholders, and otherwise disrupt our operations and harm our operating results.
We may in the future seek to acquire businesses, products or technologies that we believe could complement or expand our product offerings, enhance our technical capabilities or otherwise offer growth opportunities. The pursuit of potential acquisitions may divert the attention of management and cause us to incur various expenses in identifying, investigating and pursuing suitable acquisitions, whether or not they are consummated. If we acquire additional businesses, we may not be able to integrate the acquired personnel, operations and technologies successfully, effectively manage the combined business following the acquisition, or realize anticipated cost savings or synergies. We also may not achieve the anticipated benefits from the acquired business due to a number of factors, including:
| ● | incurrence of acquisition-related costs; |
| ● | diversion of management’s attention from other business concerns; |
| ● | unanticipated costs or liabilities associated with the acquisition; |
| ● | harm to our existing business relationships with collaboration partners as a result of the acquisition; |
85
| ● | harm to our brand and reputation; |
| ● | the potential loss of key employees; |
| ● | use of resources that are needed in other parts of our business; and |
| ● | use of substantial portions of our available cash to consummate the acquisition. |
In the future, if our acquisitions do not yield expected returns, we may be required to take charges to our operating results arising from the impairment assessment process. Acquisitions may also result in dilutive issuances of equity securities or the incurrence of debt, which could adversely affect our operating results. In addition, if an acquired business fails to meet our expectations, our business, results of operations and financial condition may be adversely affected.
If we make any acquisitions, they may disrupt or have a negative impact on our business.
If we make acquisitions in the future, funding permitting, which may not be available on favorable terms, if at all, we could have difficulty integrating the acquired company’s assets, personnel and operations with our own. We do not anticipate that any acquisitions or mergers we may enter into in the future would result in a change of control of the Company. In addition, the key personnel of the acquired business may not be willing to work for us. We cannot predict the effect expansion may have on our core business. Regardless of whether we are successful in making an acquisition, the negotiations could disrupt our ongoing business, distract our management and employees and increase our expenses. In addition to the risks described above, acquisitions are accompanied by a number of inherent risks, including, without limitation, the following:
| ● | the difficulty of integrating acquired products, services or operations; |
| ● | the potential disruption of the ongoing businesses and distraction of our management and the management of acquired companies; |
| ● | difficulties in maintaining uniform standards, controls, procedures and policies; |
| ● | the potential impairment of relationships with employees and customers as a result of any integration of new management personnel; |
| ● | the potential inability or failure to achieve additional sales and enhance our customer base through cross-marketing of the products to new and existing customers; |
| ● | the effect of any government regulations which relate to the business acquired; |
| ● | potential unknown liabilities associated with acquired businesses or product lines, or the need to spend significant amounts to retool, reposition or modify the marketing and sales of acquired products or operations, or the defense of any litigation, whether or not successful, resulting from actions of the acquired company prior to our acquisition; and |
| ● | potential expenses under the labor, environmental and other laws of various jurisdictions. |
Our business could be severely impaired if and to the extent that we are unable to succeed in addressing any of these risks or other problems encountered in connection with an acquisition, many of which cannot be presently identified. These risks and problems could disrupt our ongoing business, distract our management and employees, increase our expenses and adversely affect our results of operations.
86
We may apply working capital and future funding to uses that ultimately do not improve our operating results or increase the value of our securities.
In general, we have complete discretion over the use of our working capital and any new investment capital we may obtain in the future. Because of the number and variety of factors that could determine our use of funds, our ultimate expenditure of funds (and their uses) may vary substantially from our current intended operating plan for such funds.
We intend to use existing working capital and future funding for research and development, and general corporate purposes, including the potential expenses related to completing a reverse merger and legal expenses. We will also use capital for general working capital purposes. However, we do not have more specific plans for the use and expenditure of our capital. Our management has broad discretion to use any or all of our available capital reserves. Our capital could be applied in ways that do not improve our operating results or otherwise increase the value of a stockholder’s investment.
We have never paid or declared any dividends on our common stock.
We have never paid or declared any dividends on our common stock or preferred stock. Likewise, we do not anticipate paying, in the near future, dividends or distributions on our common stock. Any future dividends on common stock will be declared at the discretion of our Board and will depend, among other things, on our earnings, our financial requirements for future operations and growth, and other facts as we may then deem appropriate. Since we do not anticipate paying cash dividends on our common stock, return on your investment, if any, will depend solely on an increase, if any, in the market value of our common stock.
Stockholders may be diluted significantly through our efforts to obtain financing and satisfy obligations through the issuance of additional shares of our common stock.
Wherever possible, our Board will attempt to use non-cash consideration to satisfy obligations. In many instances, we believe that the non-cash consideration will consist of restricted shares of our common stock or where shares are to be issued to our officers, directors and applicable consultants. Our Board of Directors has authority, without action or vote of the stockholders, but subject to Nasdaq rules and regulations (which generally require stockholder approval for any transactions which would result in the issuance of more than 20% of our then outstanding shares of common stock or voting rights representing over 20% of our then outstanding shares of stock, subject to certain exceptions), to issue all or part of the authorized but unissued shares of common stock. In addition, we may attempt to raise capital by selling shares of our common stock, possibly at a discount to market. These actions will result in dilution of the ownership interests of existing stockholders, which may further dilute common stock book value, and that dilution may be material. Such issuances may also serve to enhance existing management’s ability to maintain control of the Company because the shares may be issued to parties or entities committed to supporting existing management.
Our growth depends in part on the success of our strategic relationships with third parties.
In order to grow our business, we anticipate that we will need to continue to depend on our relationships with third parties, including our technology providers. Identifying partners, and negotiating and documenting relationships with them, requires significant time and resources. Our competitors may be effective in providing incentives to third parties to favor their products or services, or utilization of, our products and services. In addition, acquisitions of our partners by our competitors could result in a decrease in the number of our current and potential customers. If we are unsuccessful in establishing or maintaining our relationships with third parties, our ability to compete in the marketplace or to grow our revenue could be impaired and our results of operations may suffer. Even if we are successful, we cannot assure you that these relationships will result in increased customer use of our products or increased revenue.
87
Claims, litigation, government investigations, and other proceedings may adversely affect our business and results of operations.
We are currently subject to, and expect to continue to be regularly subject to, actual and threatened claims, litigation, reviews, investigations, and other proceedings. In addition, we have filed lawsuits against certain parties for matters we discovered which related to KBL, prior to the November 6, 2020, business combination. Any of these types of proceedings may have an adverse effect on us because of legal costs, disruption of our operations, diversion of management resources, negative publicity, and other factors. The outcomes of these matters are inherently unpredictable and subject to significant uncertainties. Determining legal reserves and possible losses from such matters involves judgment and may not reflect the full range of uncertainties and unpredictable outcomes. Until the final resolution of such matters, we may be exposed to losses in excess of the amount recorded, and such amounts could be material. Should any of our estimates and assumptions change or prove to have been incorrect, it could have a material effect on our business, consolidated financial position, results of operations, or cash flows. In addition, it is possible that a resolution of one or more such proceedings, including as a result of a settlement, could require us to make substantial future payments, prevent us from offering certain products or services, require us to change our business practices in a manner materially adverse to our business, requiring development of non-infringing or otherwise altered products or technologies, damaging our reputation, or otherwise having a material effect on our operations.
Changes in laws or regulations, or a failure to comply with any laws and regulations, may adversely affect our business, investments and results of operations.
We are subject to laws, regulations and rules enacted by national, regional and local governments. In particular, we are required to comply with certain SEC, Nasdaq and other legal or regulatory requirements. Compliance with, and monitoring of, applicable laws, regulations and rules may be difficult, time consuming and costly. Those laws, regulations and rules and their interpretation and application may also change from time to time and those changes could have a material adverse effect on our business, investments and results of operations. In addition, a failure to comply with applicable laws, regulations and rules, as interpreted and applied, could have a material adverse effect on our business and results of operations.
We may be adversely affected by climate change or by legal, regulatory or market responses to such change.
The long-term effects of climate change are difficult to predict; however, such effects may be widespread. Impacts from climate change may include physical risks (such as rising sea levels or frequency and severity of extreme weather conditions-which may affect our current operations due to among other things, the fact that a majority of our operations we are based in California, which is prone to inclement weather), social and human effects (such as population dislocations or harm to health and well-being), compliance costs and transition risks (such as regulatory or technology changes) and other adverse effects. The effects of climate change could increase the cost of certain products, commodities and energy (including utilities), which in turn may impact our ability to procure goods or services required for the operation of our business. Climate change could also lead to increased costs as a result of physical damage to or destruction of our facilities, loss of inventory, and business interruption due to weather events that may be attributable to climate change. These events and impacts could materially adversely affect our business operations, financial position or results of operation.
We may incur indebtedness in the future which could reduce our financial flexibility, increase interest expense and adversely impact our operations and our costs.
We may incur significant amounts of indebtedness in the future. Our level of indebtedness could affect our operations in several ways, including the following:
| ● | a significant portion of our cash flows is required to be used to service our indebtedness; |
| ● | a high level of debt increases our vulnerability to general adverse economic and industry conditions; |
| ● | covenants contained in the agreements governing our outstanding indebtedness limit our ability to borrow additional funds and provide additional security interests, dispose of assets, pay dividends and make certain investments; |
88
| ● | a high level of debt may place us at a competitive disadvantage compared to our competitors that are less leveraged and, therefore, may be able to take advantage of opportunities that our indebtedness may prevent us from pursuing; and |
| ● | debt covenants may affect our flexibility in planning for, and reacting to, changes in the economy and in our industry. |
A high level of indebtedness increases the risk that we may default on our debt obligations. We may not be able to generate sufficient cash flows to pay the principal or interest on our debt, and future working capital, borrowings or equity financing may not be available to pay or refinance such debt. If we do not have sufficient funds and are otherwise unable to arrange financing, we may have to sell significant assets or have a portion of our assets foreclosed upon which could have a material adverse effect on our business, financial condition and results of operations.
We may be adversely impacted by changes in accounting standards.
Our consolidated financial statements are subject to the application of the accounting principles generally accepted in the United States of America (“U.S. GAAP”), which periodically is revised or reinterpreted. From time to time, we are required to adopt new or revised accounting standards issued by recognized authoritative bodies, including the Financial Accounting Standards Board (“FASB”) and the SEC. It is possible that future accounting standards may require changes to the accounting treatment in our consolidated financial statements and may require us to make significant changes to our financial systems. Such changes might have a materially adverse impact on our financial position or results of operations.
For all of the foregoing reasons and others set forth herein, an investment in our securities involves a high degree of risk.
Item 1B. Unresolved Staff Comments.
None.
Item 1C. Cybersecurity.
The Company understands the
importance of preventing, assessing, identifying, and managing material risks associated with cybersecurity threats. Consultants specializing
in cybersecurity risk were engaged to assess, identify and manage risks from cybersecurity threats separately from the Company’s
other risk assessment processes. The cybersecurity risks assessed include operational risks, fraud, disaster recovery, and violation of
data privacy or security laws. The assessment includes cybersecurity threats associated with the use of
As a result of the cybersecurity risk assessment performed, the Company has implemented certain enhancements to cybersecurity processes and controls. This includes formal monitoring of capital equipment, verifying data encryption and security updates are in place, and enhancing the security of data sharing and transmission. Additional recommendations were made by the consultants, but due to budget constraints they are being considered for future enhancement.
We describe whether and how risks from identified cybersecurity threats, including as a result of any previous cybersecurity incidents, have materially affected or are reasonably likely to materially affect us, including our business strategy, results of operations, or financial condition, under the heading “Failure of our information technology systems, including cybersecurity attacks or other data security incidents, could significantly disrupt the operation of our business”, included as part of our risk factor disclosures at Item 1A of this Annual Report on Form 10-K.
89
Cybersecurity is an important
part of our risk management processes and an area of focus for our Board and management.
As of and for the year ended
December 31, 2024, there have been no cybersecurity incidents that have
Item 2. Properties.
The Company’s headquarters are located in Palo Alto, California. The Company believes its existing leased office space is suitable for the conduct of its business. We believe that this arrangement is suitable for the conduct of our business, but we will secure a physical location for our operations if and when we believe that it becomes necessary.
Item 3. Legal Proceedings.
From time to time, we may be a party to litigation that arises in the ordinary course of our business.
Such current litigation or other legal proceedings are described in, and incorporated by reference in, this “Item 3. Legal Proceedings” of this Annual Report on Form 10-K from, “Note 10 – Commitments and Contingencies”, under the heading “Litigation and Other Loss Contingencies”, in the consolidated financial statements included herein beginning on page F-1 at the end of this Report. The Company believes that the resolution of currently pending matters will not individually or in the aggregate have a material adverse effect on our financial condition or results of operations. However, assessment of the current litigation or other legal claims could change in light of the discovery of facts not presently known to the Company or by judges, juries or other finders of fact, which are not in accord with management’s evaluation of the possible liability or outcome of such litigation or claims.
Additionally, the outcome of litigation is inherently uncertain. If one or more legal matters were resolved against the Company in a reporting period for amounts in excess of management’s expectations, the Company’s financial condition and operating results for that reporting period could be materially adversely affected.
Item 4. Mine Safety Disclosures.
Not applicable.
90
PART II
Item 5. Market for the Registrant’s Common Equity, Related Stockholder Matters and Issuer Purchases of Equity Securities
Market Information
Our common stock and warrants are listed on the Nasdaq Capital Market under the symbols “ATNF” and “ATNFW,” respectively.
Holders
As of March 31, 2025, there were 3,837,780 shares of common stock issued and outstanding held by approximately 122 holders of record, and 15,794 shares of common stock underlying 11,500,000 public warrants outstanding to purchase shares of our common stock. Each public warrant entitles the registered holder to purchase one-seven hundred sixtieth of one share of our common stock at a price of $5.75 per 1/760th of one share or $4,370 per whole share, subject to adjustment as discussed below, at any time commencing on December 6, 2020 (30 days after our initial business combination) and ending on November 6, 2025 (five years after our initial business combination), at 5:00 p.m., New York City time, or earlier upon redemption or liquidation. If a warrant holder holds 760 warrants, such warrants will be exercisable for one share of our common stock. No fractional shares will be issued upon exercise of the warrants and warrants must be exercised for whole shares only.
Dividend Policy
We have never paid or declared any cash dividends on our common stock and do not anticipate paying cash dividends in the foreseeable future. We anticipate that we will retain all of our future earnings for use in the operation of our business and for general corporate purposes. Any determination to pay dividends in the future will be at the discretion of our board of directors. Accordingly, investors must rely on sales of their common stock after price appreciation, which may never occur, as the only way to realize any future gains on their investments.
Recent Sales of Unregistered Securities
The Company has not sold any equity securities during the three months ended December 31, 2024, and through the date of this report, except as has been previously included in a Quarterly Report on Form 10-Q or in a Current Report on Form 8-K.
Issuer Purchases of Equity Securities
None.
Item 6. [Reserved]
91
Item 7. Management’s Discussion and Analysis of Financial Condition and Results of Operations.
The following discussion and analysis of the results of operations and financial condition of 180 Life Sciences Corp. as of and for the years ended December 31, 2024 and 2023 should be read in conjunction with our consolidated financial statements and the notes to those consolidated financial statements that are included elsewhere in this Annual Report. This Management’s Discussion and Analysis of Financial Condition and Results of Operations contains statements that are forward-looking. See “Cautionary Statement Regarding Forward-Looking Information” above. Actual results could differ materially because of the factors discussed in “Risk Factors” elsewhere in this Annual Report, and other factors that we may not know.
As of December 31, 2024, we had an accumulated deficit of $141,523,344 and a working capital deficit of $1,636,486, and for the year ended December 31, 2024, a net loss of $6,168,177 and cash used in operating activities of $1,480,567. The accompanying consolidated financial statements have been prepared assuming the Company will continue as a going concern. As we are not generating revenues, we need to raise a significant amount of capital in order to pay our debts and cover our operating costs. While the Company raised capital in July 2022, December 2022, April 2023, August 2023, November 2023, October 2024 and December 2024, there is no assurance that we will be able to raise additional needed capital or that such capital will be available under favorable terms.
We are subject to all the substantial risks inherent in the development of a new business enterprise within an extremely competitive industry. Due to the absence of a long-standing operating history and the emerging nature of the markets in which we compete, we anticipate operating losses until we can successfully implement our business strategy, which includes all associated revenue streams. We may never ever achieve profitable operations or generate significant revenues.
We currently have a minimum monthly cash requirement spend of approximately $252,000. We believe that in the aggregate, we will require significant additional capital funding to operationalize and commercially launch our Gaming Technology Platform, complete the disposition of our remaining biotechnology assets (either by sale or discontinuation), repay debt obligations, provide capital expenditures for additional equipment and development costs, payment obligations, office space and systems for managing the business, and cover other operating costs until our planned revenue streams from products are fully-implemented and begin to offset our operating costs, if ever.
Since our inception, we have funded our operations with the proceeds from equity and debt financing. We have experienced liquidity issues due to, among other reasons, our limited ability to raise adequate capital on acceptable terms. We have historically relied upon the issuance of equity and promissory notes that are convertible into shares of our common stock to fund our operations and have devoted significant efforts to reduce that exposure. We anticipate that we will need to issue equity to fund our operations and repay our outstanding debt for the foreseeable future. If we are unable to achieve operational profitability, or we are not successful in securing other forms of financing, we will have to evaluate alternative actions to reduce our operating expenses and conserve cash. Our current cash balance is only expected to be sufficient to fund our planned business operations through approximately December 2025. If additional capital is not available, we may not be able to pursue our planned business operations, may be forced to change our planned business operations, or may take other actions that could adversely impact our stockholders, including seeking bankruptcy protection.
The accompanying consolidated financial statements have been prepared in accordance with accounting principles generally accepted in the United States of America on a going concern basis, which contemplates the realization of assets and the satisfaction of liabilities in the normal course of business. Accordingly, the consolidated financial statements do not include any adjustments relating to the recoverability of assets and classification of liabilities that might be necessary should the Company be unable to continue as a going concern. The consolidated financial statements included in this Report also include a going concern footnote.
Additionally, wherever possible, our Board of Directors will attempt to use non-cash consideration to satisfy obligations. In many instances, we believe that the non-cash consideration will consist of restricted shares of our common stock, preferred stock or warrants to purchase shares of our common stock. Our Board of Directors has authority, without action or vote of the shareholders, but subject to Nasdaq rules and regulations (which generally require shareholder approval for any transactions which would result in the issuance of more than 20% of our then outstanding shares of common stock or voting rights representing over 20% of our then outstanding shares of stock), to issue all or part of the authorized but unissued shares of common stock, preferred stock or warrants to purchase such shares of common stock. In addition, we may attempt to raise capital by selling shares of our common stock, possibly at a discount to market in the future. These actions will result in dilution of the ownership interests of existing shareholders, may further dilute the common stock book value, and that dilution may be material. Such issuances may also serve to enhance existing management’s ability to maintain control of us, because the shares may be issued to parties or entities committed to supporting existing management.
92
Organization of MD&A
Our Management’s Discussion and Analysis of Financial Condition and Results of Operations (the “MD&A”) is provided in addition to the accompanying consolidated financial statements and notes to assist readers in understanding our results of operations, financial condition, and cash flows. MD&A is organized as follows:
| ● | Significant Financial Statement Components. A summary of the Company’s significant financial statement components. |
| ● | Results of Operations. An analysis of our financial results comparing the years ended December 31, 2024 and 2023. |
| ● | Liquidity and Capital Resources. An analysis of changes in our balance sheets and cash flows and discussion of our financial condition. |
| ● | Critical Accounting Estimates. Accounting estimates that we believe are important to understanding the assumptions and judgments incorporated in our reported financial results and forecasts. |
Significant Financial Statement Components
Research and Development
To date, 180’s research and development expenses have related primarily to discovery efforts and preclinical and clinical development of its three product platforms: (1) fibrosis and anti-TNF, (2) drugs which are derivatives of CBD, and (3) α7nAChR (which has been transferred back to Stanford University). Research and development expenses consist primarily of costs associated with those three product platforms, which include:
| ● | expenses incurred under agreements with 180’s collaboration partners and third-party contract organizations, investigative clinical trial sites that conduct research and development activities on its behalf, and consultants; |
| ● | costs related to production of clinical materials, including fees paid to contract manufacturers; |
| ● | laboratory and vendor expenses related to the execution of preclinical and clinical trials; |
| ● | employee-related expenses, which include salaries, benefits, and stock-based compensation; and |
| ● | facilities and other expenses, which include expenses for rent and maintenance of facilities, depreciation and amortization expense and other supplies. |
We expense all research and development costs in the periods in which they are incurred. We accrue for costs incurred as services are provided by monitoring the status of each project and the invoices received from our external service providers. We adjust our accrual as actual costs become known. When contingent milestone payments are owed to third parties under research and development arrangements or license agreements, the milestone payment obligations are expensed when the milestone results are achieved.
Research and development activities are important for both our legacy biotechnology assets as well as our new focus on iGaming. For our legacy biotech assets, expenditures are limited to those which are deemed necessary to preserve the value of this legacy intellectual property. This includes, in the case of the SCA asset, having funded a pharmo-kinetic study, while the ATNF IP has been in care and maintenance mode. The costs for ongoing work, if any, on the biotechnology IP are expected to be circa $150,000; management has not yet determined that these additional expenses will be required prior to monetization of this asset. In respect of our new focus on iGaming, the research and development costs relate to evaluation of content for our proposed iGaming operation (populating our front-end), potential development costs associated with the Gaming Technology software acquired, and potential development costs associated with a front-end, when acquired.
93
The duration, costs and timing of the iGaming related research and development will depend on a variety of factors, including, but not limited to, the following:
| ● | availability of “for purchase” front-end user interfaces, and their fitness for the purpose of establishing an operating online casino; |
| ● | availability of suitable content (i.e., games and sports betting books); and |
| ● | the state of regulatory, technological and commercial development in the iGaming industry generally, and specifically in those markets where the Company chooses to operate. |
We will determine which development objectives to pursue and fund in response to the commercial potential of both our proposed online casino, as well as general industry dynamics.
Because the iGaming industry is rapidly evolving, particularly with respect to technological and regulatory change, we cannot estimate the actual amounts necessary to successfully complete the development and commercialization of various products or whether, or when, we may achieve profitability.
General and Administrative
General and administrative expenses consist primarily of salaries and other staff-related costs, including stock-based compensation for shares of common stock issued and options granted to founders, directors, and personnel in executive, commercial, finance, accounting, legal, investor relations, facilities, business development, and human resources functions that include vesting conditions.
Other significant general and administrative costs include costs relating to facilities and overhead costs, legal fees relating to corporate and patent matters, litigation, SEC filings, insurance, investor relations costs, fees for accounting and consulting services, and other general and administrative costs. General and administrative costs are expensed as incurred, and we accrue amounts for services provided by third parties related to the above expenses by monitoring the status of services provided and receiving estimates from our service providers and adjusting our accruals as actual costs become known.
It is expected that the general and administrative expenses will increase over the next several years to support our continued research and development activities related to the iGaming business (as described above), the launch of commercial iGaming operations and the increased costs of operating as a public company. These increases are anticipated to include increased costs related to the hiring of additional personnel, developing commercial infrastructure, fees to outside consultants, lawyers and accountants, and increased costs associated with being a public company, as well as expenses related to services associated with maintaining compliance with Nasdaq listing rules and SEC requirements, insurance and investor relations costs.
Interest Expense
Interest expense consists primarily of interest expense related to debt instruments.
94
Loss on Goodwill Impairment
Loss on goodwill impairment represents an excess of the carrying value of the asset over its estimated fair market value during the reporting period which is not recoverable.
Loss on IP R&D assets impairment
Loss on IP R&D assets impairment represents an excess of the carrying value of the assets over its estimated fair market value during the reporting period which is not recoverable.
Change in Fair Value of IR R&D assets
Change in fair value of IP R&D assets represents the non-cash change in fair value of the assets during the reporting period.
Change in Fair Value of Derivative Liabilities
Change in fair value of derivative liabilities represents the non-cash change in fair value of derivative liabilities during the reporting period. Gains resulting from change in fair value of derivative liabilities during the years ended December 31, 2024 and 2023, were driven by decreases in stock price during the period, resulting in a lower fair value of the underlying liability.
Gain on Issuance of Common Stock for Services
The gain on issuance of common stock for services represents the excess of the price per share paid for services rendered over the fair market value of the shares on the date they were issued.
CONSOLIDATED RESULTS OF OPERATIONS
Consolidated Results of Operations
For the Year Ended December 31, 2024 Compared to the Year Ended December 31, 2023
| For the Years Ended December 31, | ||||||||
| 2024 | 2023 | |||||||
| Operating Expenses: | ||||||||
| Research and development | $ | 1,401,435 | $ | 2,303,751 | ||||
| Research and development – related parties | 567,832 | 480,777 | ||||||
| General and administrative | 4,355,462 | 10,646,417 | ||||||
| General and administrative – related parties | - | 46,555 | ||||||
| Total Operating Expenses | 6,324,729 | 13,477,500 | ||||||
| Loss From Operations | (6,324,729 | ) | (13,477,500 | ) | ||||
| Other (Expense) Income: | ||||||||
| Other income | 2,822,864 | 21,074 | ||||||
| Interest expense | (50,640 | ) | (44,828 | ) | ||||
| Loss on impairment | (1,526,542 | ) | (9,063,000 | ) | ||||
| Change in fair value of derivative liabilities | 58 | 75,323 | ||||||
| Change in fair value of preferred stock liability | (1,577,305 | ) | - | |||||
| Gain on issuance of common stock for services | 192,093 | 204,405 | ||||||
| Total Other Expense, Net | (139,472 | ) | (8,807,026 | ) | ||||
| Loss Before Income Taxes | (6,464,201 | ) | (22,284,526 | ) | ||||
| Income tax benefit | 296,024 | 2,349,414 | ||||||
| Net Loss | (6,168,177 | ) | (19,935,112 | ) | ||||
| Deemed dividend | (8,011,510 | ) | - | |||||
| Net Loss available to Common Stockholders | $ | (14,179,687 | ) | $ | (19,935,112 | ) | ||
95
Research and Development
During the year ended December 31, 2024, we incurred research and development expenses of $1,401,435, compared to $2,303,751 incurred for the year ended December 31, 2023, representing a decrease of $902,316 or 39%. The change is attributable to a decrease in research and development expense includes a) an overall decrease in research and development (R&D) program spending based on Company resource allocation and b) a reduction in stock-based compensation of approximately $298,000.
Research and Development – Related Parties
During the year ended December 31, 2024, we incurred research and development expenses – related parties of $567,832, compared to $480,777 incurred for the year ended December 31, 2023, representing an increase of $87,055 or 18%. The change is mainly attributable to an overall increase in R&D program spending based on Company resource allocation.
General and Administrative
During the year ended December 31, 2024, we incurred general and administrative expenses of $4,355,462, compared to $10,646,417 incurred for the year ended December 31, 2023, representing a decrease of $6,290,955 or 59%. The change resulted from reduced legal fees of approximately $1.2 million from lower activity related to ongoing litigation in the current period, reduced professional fees of approximately $2.3 million, from lower consulting and accounting fees, and reduced salaries expense of approximately $528,000 from a reduction in headcount and a reduction in stock-based compensation expense of approximately $1.7 million. The decline in stock-based compensation is primarily due to previous awards being fully vested in 2023 and no significant new grants during 2024.
General and Administrative – Related Parties
During the year ended December 31, 2024, we incurred general and administrative expenses – related parties of $0 compared to $46,555 incurred for the year ended December 31, 2023, representing a decrease of $46,555, or 100%. The decrease is related to a decrease in related party consulting expenses of $46,555.
Other Expenses, Net
During the year ended December 31, 2024, we incurred other income, net of $139,472, compared to other expenses, net of $8,807,026 for the year ended December 31, 2023, representing a decrease in other expenses of $8,667,554 or 98%. The decrease is primarily attributable to an impairment to IP R&D assets in the prior year of approximately $9.1 million (see “Note 3 – Summary of Significant Accounting Policies— Intangible Assets and In-Process Research and Development (“IP R&D”)” compared to a loss on impairment of patents and technology of $1.5 million, in the notes to consolidated financial statements included at the end of this Report, for further information), $1,712,804 in proceeds received from AmTrust our pre-Merger officer and director insurance carrier (see “Note 10 – Commitments and Contingencies—Legal Matters”, in the notes to consolidated financial statements included at the end of this Report, for further information), a gain on settlement of liabilities with a lender of $156,928, and a gain of $1,039,364 from settlements with certain R&D contractors (see “Note 10 – Commitments and Contingencies—Legal Matters”, in the notes to consolidated financial statements included at the end of this Report, for further information).
96
Net Loss
We had a net loss of $6,168,177 for the year ended December 31, 2024, compared to a net loss of $19,935,112 for the year ended December 31, 2023, a decrease in net loss $13,766,935 or 69% for the reasons discussed above.
Deemed Dividend
During the year ended December 31, 2024, the Company recognized a deemed dividend of $8,011,510 in connection with its October 2024 Warrant Inducement related to the new warrants issued to the investor.
Liquidity and Capital Resources
As of December 31, 2024 and 2023, we had cash balances of $4,585,141 and $1,975,799, respectively, and a working capital deficit of $1,636,486 and $1,422,710, respectively.
For the years ended December 31, 2024 and 2023, cash used in operating activities was $1,480,567 and $10,922,223, respectively. Our cash used in operations for the year ended December 31, 2024 was primarily attributable to our net loss of $6,168,177, adjusted for non-cash expenses in the aggregate amount of $2,955,016, as well as $1,732,594 of net cash used in changes in the levels of operating assets and liabilities. Our cash used in operations for the year ended December 31, 2023 was primarily attributable to our net loss of $19,935,112, adjusted for non-cash expenses in the aggregate amount of $9,026,993, as well as $14,104 of net cash used in changes in the levels of operating assets and liabilities. A significant portion of the non-cash expenses during the year relates to $9.1 million of non-recurring expenses associated with the impairment of IP R&D assets (see “Note 5 – Intangible Assets and Impairment of Long-lived Assets”, in the notes to consolidated financial statements included at the end of this Report, for more information).
For the years ended December 31, 2024 and 2023, there was no cash provided by investing activities.
For the years ended December 31, 2024 and 2023, cash provided by financing activities was $4,037,715 and $5,907,887, respectively. Cash provided by financing activities during the year ended December 31, 2024, was primarily comprised of i) proceeds from exercise of pre-funded warrants sold in November 2023 of $489 ii) proceeds from sale of stock and warrants in December 2024 of $2,572,769 and iii) proceeds from the exercise of warrants of $2,797,936, partially offset by iv) the repayment of loans payable of $1,333,499. Cash provided by financing activities during the year ended December 31, 2023, was primarily comprised of i) proceeds from the sale of common stock and common stock warrants from the April and August 2023 Offerings (discussed below) of $5,999,488 and ii) proceeds from the repricing of pre-funded warrants and common warrants from the August 2023 Offering (discussed below) of $830,769, partially offset by iii) aggregate offering costs paid in connection with our April and August 2023 Offerings and the repricing of the August 2023 Offering of $634,816, as well as iv) proceeds from and repayments of loans of $969,322 and $1,257,389, respectively.
Our drug product candidates may never achieve commercialization (in the form of a sale of the intellectual property), and our iGaming business may never achieve full commercialization; we anticipate that we will continue to incur losses for the foreseeable future. We expect that our research and development expenses, general and administrative expenses, and capital expenditures will continue to increase. As a result, until such time, if ever, as we are able to generate substantial product revenue, we expect to finance our cash needs through a combination of equity offerings, debt financings or other capital sources, including potentially collaborations, licenses and other similar arrangements, which may not be available on favorable terms, if at all. The sale of additional equity or debt securities, if accomplished, may result in dilution to our then stockholders. Our primary uses of capital are, and we expect will continue to be, compensation and related expenses, certain third-party clinical research and development services, certain third party service providers, including but not limited to marketing, content, and management services in respect of our iGaming operations, certain license payments, intellectual property maintenance costs or milestone obligations that may arise, laboratory and related supplies, clinical costs, potential manufacturing costs, legal and other regulatory expenses and general overhead costs.
Our material cash requirements and time periods of such requirements from known contractual and other obligations include milestone and royalty payments related to license agreements with Oxford University and Yissum, payments related to the D&O insurance, payments to consultants and payments related to outside consulting firms, such as legal counsel, auditors, accountants, etc.
97
Further, our operating plans may change, and we may need additional funds to meet operational needs and capital requirements for launching our iGaming operations, and research and development activities related to both certain biotechnology assets as well as in respect of our proposed iGaming operations. We currently have no credit facility or committed sources of capital. Because of the numerous risks and uncertainties associated with the development and commercialization of our iGaming operations, and in terms of monetizing our biotechnology product candidates, we are unable to estimate the amounts of increased capital outlays and operating expenditures associated with our current and anticipated product development programs.
We have not yet achieved profitability and expect to continue to incur cash outflows from operations. It is expected that our research and development and general and administrative expenses will continue to increase and, as a result, we will eventually need to raise additional capital to fund our operations. If we are unable to obtain adequate funds on reasonable terms, we may be required to significantly curtail or discontinue operations or obtain funds by entering into financing agreements on unattractive terms. Our operating needs include the planned costs to operate our business, including amounts required to fund working capital and capital expenditures. As of December 31, 2024, the conditions outlined above indicated that there was a substantial doubt about our ability to continue as a going concern within one year after the financial statement issuance date. However, in April 2023, August 2023, November 2023, October 2024 and December 2024, the Company raised additional capital of approximately $2.7 million, $2.7 million, $0.8 million, $3.3 million, and $2.6 million, respectively, and with current cash on hand of approximately $3.8 million as of March 25, 2025, the Company expects to be able to continue as a going concern through December 2025.
Our consolidated financial statements have been prepared in conformity with accounting principles generally accepted in the United States of America (“U.S. GAAP”), which contemplate continuation of the Company as a going concern and the realization of assets and satisfaction of liabilities in the normal course of business. The carrying amounts of assets and liabilities presented in the consolidated financial statements do not necessarily purport to represent realizable or settlement values. The consolidated financial statements do not include any adjustment that might result from the outcome of this uncertainty.
Prior Financings
December 2022 Offering
On December 20, 2022, the Company entered into a Securities Purchase Agreement with certain purchasers, pursuant to which the Company agreed to sell an aggregate of 11,316 shares of common stock, pre-funded warrants to purchase up to an aggregate of 78,910 shares of common stock (“December 2022 Pre-Funded Warrants”), and common stock warrants to purchase up to an aggregate of 135,339 shares of common stock (the “December 2022 Common Warrants”), at a combined purchase price of $66.50 per share and warrant (the “December 2022 Offering”). Aggregate gross proceeds from the December 2022 Offering were $5,999,851. Net proceeds to the Company from the offering, after deducting the placement agent fees and other estimated offering expenses payable by the Company, were approximately $5.5 million. The placement agent fees and offering expenses of approximately $500,000 were accounted for as a reduction of additional paid in capital. The December 2022 Offering closed on December 22, 2022.
98
April 2023 Offering
On April 5, 2023, the Company entered into a Securities Purchase Agreement with certain purchasers, pursuant to which the Company agreed to sell an aggregate of 21,053 shares of common stock, pre-funded warrants to purchase up to an aggregate of 61,615 shares of common stock (“April 2023 Pre-Funded Warrants”), and common stock warrants to purchase up to an aggregate of 82,668 shares of common stock (the “April 2023 Common Warrants”), at a combined purchase price of $36.29 per share and warrant (the “April 2023 Offering”). Aggregate gross proceeds from the April 2023 Offering were $2,999,882. Net proceeds to the Company from the offering, after deducting the placement agent fees and other estimated offering expenses payable by the Company, were approximately $2.7 million. The placement agent fees and offering expenses of approximately $300,000 were accounted for as a reduction of additional paid in capital. The April 2023 Offering closed on April 10, 2023.
August 2023 Offering
On August 9, 2023, the Company entered into a Securities Purchase Agreement with an accredited investor, in addition to certain purchasers who relied on the Company’s registration statement filed with the SEC on July 25, 2023, which became effective on August 9, 2023, pursuant to which the Company agreed to sell an aggregate of 35,102 shares of common stock, pre-funded warrants to purchase up to an aggregate of 207,814 shares of common stock (“August 2023 Pre-Funded Warrants”), and common stock warrants to purchase up to an aggregate of 242,915 shares of common stock (the “August 2023 Common Warrants”), at a combined purchase price of $12.35 per share and warrant (the “August 2023 Offering”). Aggregate gross proceeds from the August 2023 Offering were $2,999,606. Net proceeds to the Company from the offering, after deducting the placement agent fees and other estimated offering expenses payable by the Company, were approximately $2.7 million. The placement agent fees and offering expenses of approximately $300,000 were accounted for as a reduction of additional paid in capital. The August 2023 Offering closed on August 14, 2023.
Amendment to August 2023 Offering
On October 11, 2023, the Company received a letter from Nasdaq, stating that Nasdaq had determined that the Company failed to comply with Nasdaq’s shareholder approval requirements set forth in Listing Rule 5635(d), which requires prior shareholder approval for transactions, other than public offerings, involving the issuance of 20% or more of the pre-transaction shares outstanding at less than the Minimum Price. The letter from Nasdaq referred to the August 2023 Offering and requested the Company to submit a plan of compliance no later than November 27, 2023. On November 9, 2023, the Company submitted a plan of compliance to Nasdaq, and on November 14, 2023, Nasdaq provided a letter of extension to the Company.
Pursuant to the compliance plan submitted to Nasdaq, on November 28, 2023, the Company entered into an amendment to the August 2023 Offering (“Amendment to the August 2023 Offering”), whereby (i) the main purchaser (the “Purchaser”) agreed to pay an additional $830,769 in connection with the repricing of the August 2023 shares of common stock and August 2023 Pre-Funded Warrants (the “Repricing Amount”), (ii) the Company agreed to issue to the Purchaser additional pre-funded warrants to purchase up to 257,205 shares of common stock, with an exercise price of $0.0019 per share (the “Additional Pre- Funded Warrants”), and warrants to purchase up to 477,058 shares of common stock, with an exercise price of $3.23 per share (the “Additional Common Warrants”, collectively the “Additional Warrants”), and (iii) the Company and the Purchaser agreed to enter into the warrant amendment agreement. Net proceeds to the Company from the repricing, after deducting the placement agent fees and other estimated offering expenses payable by the Company, were approximately $0.8 million. The placement agent fees and offering expenses of approximately $60,000 were accounted for as a reduction of additional paid in capital. The Amendment to the August 2023 Offering closed on December 1, 2023.
99
In accordance with the Amendment to the August 2023 Offering, the Company entered into a warrant amendment agreement to amend the following outstanding warrants held by the Purchaser: (i) warrants to purchase up to 16,138 shares of common stock issued in July 2022; (ii) warrants to purchase up to 135,339 shares of common stock issued in December 2022; (iii) warrants to purchase up to 82,668 shares of common stock issued in April 2023; and (iv) warrants to purchase up to 242,915 shares of common stock issued in August 2023 (collectively, the “Existing Common Warrants”). Pursuant to the warrant amendment agreement, the Existing Common Warrants were not exercisable until the Company obtained stockholder approval for the issuance of up to 477,058 shares of common stock upon exercise of the Existing Common Warrants. The Existing Common Warrants have an exercise price equal to $3.23 per share, and the Existing Common Warrants will expire on the fifth anniversary of the stockholder approval date. The stockholder approval date occurred on February 16, 2024.
Warrant Inducement Agreement and Related Transactions
On October 16, 2024, we entered into a warrant inducement agreement with a holder of 477,058 Additional Common Warrants and 473,011 of the Existing Common Warrants, pursuant to which the Holder agreed to exercise for cash the Exercised Warrants to purchase 950,069 shares of common stock at an exercise price of $3.48 per share ($0.25 greater than the $3.23 per share exercise price of such Exercised Warrants) during the period from the date of the Inducement Agreement until 1:15 p.m., Eastern Time, on October 16, 2024. On October 16 and 17, 2024, the Exercised Warrants were exercised in full for cash by the Holder and the Company received $3,306,240 before deducting financial advisory fees and other expenses payable by us.
In consideration of the Holder’s agreement to exercise the Exercised Warrants in accordance with the Inducement Agreement, the Company agreed to issue new unregistered Warrants to Purchase Shares of Common Stock to purchase a number of shares of common stock equal to 200% of the number of shares of common stock issued upon exercise of the Exercised Warrants, i.e., warrants to purchase up to 1,900,138 shares of common stock. The Inducement Warrants were immediately exercisable and have a term of exercise of five years.
The Company agreed in the Inducement Agreement to file a registration statement on Form S-1 to register the resale of the Inducement Warrant Shares upon exercise of the Inducement Warrants by November 15, 2024, and to use commercially reasonable efforts to have such Resale Registration Statement declared effective by the Commission within sixty (60) calendar days following the date of filing with the Commission (or ninety (90) calendar days following the date of filing with the Commission in the event of a ‘full review’ by the Commission) and to keep the Resale Registration Statement effective at all times until no holder of the Inducement Warrants owns any Inducement Warrants or Inducement Warrant Shares. In the event that the Company fails to timely deliver to the Holder the Inducement Warrant Shares without restrictive legends, the Company has agreed to pay certain liquidated damages to the Holder. The Resale Registration Statement was timely filed and timely declared effective by the SEC.
The Company has used, and expects to use, the net proceeds from these transactions to advance the commercialization of the technology gaming platform recently acquired by the Company, and for working capital, and other general corporate purposes.
The Inducement Warrants will have an exercise price of $1.50 per share. The exercise price and the number of shares of common stock issuable upon exercise of each Inducement Warrant are subject to appropriate adjustments in the event of certain stock dividends and distributions, stock splits, stock combinations, reclassifications or similar events affecting the common stock. In addition, in certain circumstances, upon a fundamental transaction, a holder of Inducement Warrants will be entitled to receive, upon exercise of the Inducement Warrants, the kind and amount of securities, cash or other property that such holder would have received had they exercised the Inducement Warrants immediately prior to the fundamental transaction.
100
The Company may not affect the exercise of Inducement Warrants, and the applicable Holder will not be entitled to exercise any portion of any such Inducement Warrant, which, upon giving effect to such exercise, would cause the aggregate number of shares of common stock beneficially owned by the holder of such Inducement Warrant (together with its affiliates) to exceed 4.99% or 9.99%, as applicable, of the number of shares of common stock outstanding immediately after giving effect to the exercise, as such percentage ownership is determined in accordance with the terms of such Inducement Warrants.
The Company engaged A.G.P./Alliance Global Partners (“A.G.P.”) to provide exclusive financial services in connection with the transactions summarized above and, pursuant to a Financial Advisory Agreement between the Company and A.G.P., paid A.G.P. a financial advisory fee of $232,000, and an alternative transaction fee of $100,000. In addition, we reimbursed A.G.P. for its accountable legal expenses in connection with the exercise of the Exercised Warrants and the issuance of the Inducement Warrants of $65,000 and $10,000 non-accountable expenses. In addition, we paid A.G.P. $29,923, half of the financial advisory fees due in connection with a December 2023 warrant inducement. As of October 31, 2024, a total of $436,923 related to the aforementioned transaction has been paid to A.G.P and there is no balance outstanding.
December 2024 Offering
On December 27, 2024, we entered into a Securities Purchase Agreement with certain accredited investors, including the Holder. Pursuant to the terms of the SPA, the Company agreed to sell, in a registered direct offering, an aggregate of 1,200,000 shares of the Company’s common stock and, in a concurrent private placement, warrants to purchase up to 1,200,000 shares of common stock. The combined purchase price per December 2024 Share and December 2024 Warrant was $2.41. The offerings closed on December 30, 2024.
The December 2024 Warrants were immediately exercisable on their grant date at an exercise price of $2.28 per share and expire five and a half years following the initial exercise date.
Pursuant to a placement agency agreement dated as of December 27, 2024, between the Company and Maxim Group LLC, the Company engaged the Placement Agent to act as the Company’s sole placement agent in connection with the registered direct offering. Pursuant to the Placement Agency Agreement, the Company agreed to pay the Placement Agent a cash fee equal to seven percent (7.0%) of the gross proceeds received by the Company from the sale of the December 2024 Shares and December 2024 Warrants and to reimburse the Placement Agent for certain of its expenses in an aggregate amount of $50,000. The Placement Agency Agreement contains customary representations, warranties and agreements by the Company, customary conditions to closing, indemnification obligations of the Company and the Placement Agent, including for liabilities under the Securities Act, other obligations of the parties, and termination provisions.
The Company agreed to file a registration statement under the Securities Act with the SEC, covering the resale of the December 2024 Warrant Shares within 30 calendar days following the date of the SPA, which has been filed to date, and to use commercially reasonable efforts to cause the registration statement to be declared effective by the SEC within 91 days following the closing of the offerings, which was declared effective timely.
The net proceeds to the Company from the registered direct offering and concurrent private placement, after deducting the placement agent’s fees and expenses and the Company’s offering expenses were approximately $2.6 million. The Company intends to use the net proceeds from the transactions for working capital and general corporate purposes, which may include operationalizing and developing the Gaming Technology Platform and capital expenditures.
101
Critical Accounting Estimates
The Company’s consolidated financial statements are prepared in accordance with accounting principles that are generally accepted in the United States. The preparation of these consolidated financial statements requires management to make estimates and assumptions that affect the reported amounts of its assets, liabilities, revenue and expenses. The Company has identified certain estimates as critical to its business operations and the understanding of its past or present results of operations related to impairment of long-lived assets. These estimates are considered critical because they had a material impact, or they have the potential to have a material impact, on the Company’s consolidated financial statements and because they require management to make significant judgments, assumptions or estimates. The Company believes that the estimates, judgments and assumptions made when accounting for the items described below were reasonable, based on information available at the time they were made. However, actual results may differ from those estimates, and these differences may be material.
Long-Lived Asset Impairment
We assess our long-lived assets, including intangible assets, for impairment at least annually, or earlier if there are indications that the fair value of the Company’s reporting units may not exceed their carrying value. This assessment involves significant judgment regarding future cash flow, discount rates, market conditions and other factors. If our estimates are not accurate, they could result in material adjustments to the consolidated financial statements. During the year ended December 31, 2024, the Company determined that its licensed R&D patents and related technology assets were fully impaired due to the Company’s shift in strategy to focus on its blockchain casino technology assets acquired in September 2024. Accordingly, we recognized an impairment loss of $1,526,542 during the year ended December 31, 2024.
Recently Issued Accounting Pronouncements
See Note 3 – Summary of Significant Accounting Policies of our consolidated financial statements included within this Annual Report for a summary of recently issued and adopted accounting pronouncements.
Item 7A. Quantitative and Qualitative Disclosures About Market Risk.
Pursuant to Item 305 of Regulation S-K (§ 229.305(e)), the Company is not required to provide the information required by this Item as it is a “smaller reporting company,” as defined by Rule 229.10(f)(1), however, the Company has provided the following information below relating to interest rate risk.
Interest Rate Risk
We are exposed to market risks in the ordinary course of its business. These risks primarily include interest rate sensitivities. As of December 31, 2024, we had $4,572,983 in cash and cash equivalents. We intend to hold our cash in interest-bearing money market accounts. Our primary exposure to market risk is interest rate sensitivity, which is affected by changes in the general level of U.S. interest rates. Due to the short-term maturities of our cash equivalents and the low risk profile of its investments, an immediate 100 basis point change in interest rates would not have a material effect on the fair market value of our cash equivalents.
102
Item 8. Financial Statements and Supplemental Data.
The information required by this Item is included in this Report as set forth in the “Index to Consolidated Financial Statements” which appear on page F-1 of this Annual Report on Form 10-K, after the signature pages of this Report, and is incorporated by reference herein.
Item 9. Changes in and Disagreements with Accountants on Accounting and Financial Disclosure.
None.
Item 9a. Controls and Procedures.
Evaluation of Disclosure Controls and Procedures
Our management, with the participation of our principal executive officer and principal financial officer, evaluated the effectiveness of our disclosure controls and procedures, as such term is defined in Rules 13a-15(e) and 15d-15(e) promulgated under the Securities and Exchange Act as of December 31, 2024. As noted above with respect to the ineffective disclosure controls (for which management is evaluating processes to address these deficiencies), based on the evaluation, the principal executive officer and the principal financial officer have concluded that the Company’s disclosure controls and procedures were not effective as of December 31, 2024.
Management’s Report on Internal Control over Financial Reporting
Management of 180 Life Sciences Corp. is responsible for establishing and maintaining adequate internal control over financial reporting, as such term is defined in Exchange Act Rules 13a-15(f) and 15d-15(f). Internal control over financial reporting is a process designed by, or under the supervision of, our principal executive officer and principal financial officer, or persons performing similar functions, and effected by our board of directors to provide reasonable assurance regarding the reliability of financial reporting and the preparation of financial statements in accordance with generally accepted accounting principles. A company’s internal control over financial reporting includes those policies and procedures that: (i) pertain to the maintenance of records that, in reasonable detail, accurately and fairly reflect the transactions and dispositions of the assets of the Company; (ii) provide reasonable assurance that transactions are recorded as necessary to permit preparation of financial statements in accordance with generally accepted accounting principles, and that receipts and expenditures of the Company are being made only in accordance with authorizations of management and directors of the Company; and (iii) provide reasonable assurance regarding prevention or timely detection of unauthorized acquisition, use, or disposition of the Company’s assets that could have a material effect on the financial statements.
Because of its inherent limitations, internal control over financial reporting may not prevent or detect misstatements. Therefore, even those systems determined to be effective can provide only reasonable assurance with respect to financial statement preparation and presentation, projections of any evaluation of effectiveness to future periods are subject to the risk that controls may become inadequate because of changes in conditions or that the degree of compliance with the policies or procedures may deteriorate.
A material weakness is a control deficiency or combination of control deficiencies, that results in more than a remote likelihood that a material misstatement of the annual or interim financial statements will not be prevented or detected on a timely basis. As a company with limited accounting resources, a significant amount of management’s time and attention has been and will be diverted from our business to ensure compliance with these regulatory requirements.
103
We are a “smaller reporting company” as defined in Item 10(f)(1) of Regulation S-K under the Securities Act. For as long as we continue to be a smaller reporting company, we may take advantage of exemptions from various reporting requirements that are applicable to other public companies that are not smaller reporting companies.
Management of 180 Life Sciences Corp., including our principal financial officer, conducted an evaluation of the effectiveness of the Company’s internal control over financial reporting as of December 31, 2024 using the criteria set forth by the Committee of Sponsoring Organizations of the Treadway Commission (“COSO”) in “Internal Control - Integrated Framework” (2013).
Management concluded that the Company’s internal control over financial reporting was not effective as of December 31, 2024, based on those criteria.
Limitations on Effectiveness of Controls and Procedures
In designing and evaluating the disclosure controls and procedures, management recognizes that any controls and procedures, no matter how well designed and operated, can provide only reasonable assurance of achieving the desired control objectives. In addition, the design of disclosure controls and procedures must reflect the fact that there are resource constraints and that management is required to apply its judgment in evaluating the benefits of possible controls and procedures relative to their costs.
Changes in Internal Control over Financial Reporting
There have been no changes in the Company’s internal control over financial reporting during the quarter ended December 31, 2024, that materially affected, or are reasonably likely to materially affect, the Company’s internal control over financial reporting.
Item 9B. Other Information.
(a) Form 8-K Information. Because this Annual Report on Form 10-K is being filed within four business days from the date of the reportable event discussed below, we have elected to make the following disclosures in this Annual Report on Form 10-K instead of in a Current Report on Form 8-K under Item 3.02:
Item 3.02 Unregistered Sales of Equity Securities.
On March 27, 2025, Elray Resources, Inc., the sole holder of the Series B Convertible Preferred Stock of the Company, converted all 1,000,000 outstanding shares of Series B Convertible Preferred Stock of the Company into 1,318,000 shares of common stock (the “Conversion Shares”)(1.318 shares of common stock for each share of Series B Convertible Preferred Stock converted), in accordance with the terms of such preferred stock and the optional conversion right set forth therein.
We claim an exemption from registration provided by Section 3(a)(9) of the Securities Act for the issuance of the Conversion Shares, as the securities were exchanged by us with our existing security holder in a transaction where no commission or other remuneration was paid or given directly or indirectly for soliciting such exchange.
Following the issuance of the Conversion Shares, we will have an aggregate of 5,185,780 shares of common stock outstanding.
(b) Rule 10b5-1 Trading
Plans. During the quarter ended December 31, 2024, none of the Company’s directors or officers (as defined in Rule 16a-1(f))
Item 9C. Disclosure Regarding Foreign Jurisdictions That Prevent Inspections.
None.
104
PART III
Information required by Items 10, 11, 12, 13 and 14 of Part III is omitted from this Annual Report and will be filed in a definitive proxy statement or by an amendment to this Annual Report not later than 120 days after the end of the fiscal year covered by this Annual Report.
Item 10. Directors, Executive Officers and Corporate Governance.
The information required by this Item is included under the headings “Election of Directors”, “Executive Officers”, “Corporate Governance”, “Code of Ethics”, “Board Committee Membership”, “Insider Trading/Anti-Hedging Policies” and “Delinquent Section 16(a) Reports” (to the extent applicable and warranted) in the Company’s 2025 Proxy Statement to be filed with the SEC within 120 days after December 31, 2024, in connection with the solicitation of proxies for the Company’s 2025 annual meeting of stockholders and is incorporated herein by reference.
Item 11. Executive Compensation.
The information required by this Item is included under the headings “Executive and Director Compensation”, “Executive Compensation”, “Director Compensation”, “Outstanding Equity Awards at Fiscal Year-End”, “Compensation Committee Interlocks and Insider Participation”, “Policy on Timing of Award Grants”, and “Compensation Committee Report” (to the extent required), in the Company’s 2025 Proxy Statement to be filed with the SEC within 120 days after December 31, 2024 and is incorporated herein by reference.
Item 12. Security Ownership of Certain Beneficial Owners and Management and Related Stockholder Matters.
The information required by this Item is included under the heading “Voting Rights and Principal Stockholders” and “Equity Compensation Plan Information” in the Company’s 2025 Proxy Statement to be filed with the SEC within 120 days after December 31, 2024, and is incorporated herein by reference.
Item 13. Certain Relationships and Related Transactions, and Director Independence.
The information required by this Item is included under the headings “Certain Relationships and Related Transactions” and “Corporate Governance” - “Director Independence” in the Company’s 2025 Proxy Statement to be filed with the SEC within 120 days after December 31, 2024 and is incorporated herein by reference.
Item 14. Principal Accountant Fees and Services.
Our independent public accounting firm during the year ended December 31, 2023 was Marcum LLP, San Francisco, CA, PCAOB Auditor ID Auditor Firm Id: 688.
Our independent public accounting firm during the year ended December 31, 2024 is M&K CPAS, PLLC, Houston, TX, PCAOB Auditor ID Auditor Firm Id: 2738.
The information required by this Item is included under the heading “Ratification of Appointment of Auditors”-“Audit Fees” in the Company’s 2025 Proxy Statement to be filed with the SEC within 120 days after December 31, 2024 and is incorporated herein by reference.
105
PART IV
Item 15. Exhibits, Financial Statements and Schedules
| (1) | (a) Documents filed as part of this Annual Report: |
The following is an index of the financial statements, schedules and exhibits included in this Form 10-K or incorporated herein by reference.
All Financial Statements
(2) Consolidated Financial Statement Schedules
Except as provided above, all financial statement schedules have been omitted, since the required information is not applicable or is not present in amounts sufficient to require submission of the schedule, or because the information required is included in the consolidated financial statements and notes thereto included in this Form 10-K.
(3) Exhibits
EXHIBIT INDEX
106
107
108
109
110
111
112
| * | Filed herewith. | |
| ** | Furnished herewith. | |
| # | Management contract or compensatory plans or arrangements. | |
| £ | Certain schedules, annexes and similar attachments have been omitted pursuant to Item 601(a)(5) of Regulation S-K. A copy of any omitted schedule or exhibit will be furnished supplementally to the Securities and Exchange Commission upon request; provided, however that 180 Life Sciences Corp. may request confidential treatment pursuant to Rule 24b-2 of the Securities Exchange Act of 1934, as amended, for any schedule or exhibit so furnished. |
Item 16. Form 10–K Summary
None.
113
SIGNATURES
Pursuant to the requirements of Section 13 or 15(d) of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned, thereunto duly authorized.
| 180 LIFE SCIENCES CORP. | ||
| Date: March 31, 2025 | /s/ Blair Jordan | |
| By: | Blair Jordan, Chief Executive Officer (Principal Executive Officer) | |
| Date: March 31, 2025 | /s/ Eric R. Van Lent | |
| By: | Eric R. Van Lent, Chief Accounting Officer (Principal Financial and Accounting Officer) | |
Pursuant to the requirements of the Securities Exchange Act of 1934, this report has been signed below by the following persons on behalf of the registrant and in the capacities and on the dates indicated.
| Signature | Title | Date | ||
| /s/ Blair Jordan | Chief Executive Officer and Director | March 31, 2025 | ||
| Blair Jordan | (Principal Executive Officer) | |||
| /s/ Eric R. Van Lent | Chief Accounting Officer | March 31, 2025 | ||
| Eric R. Van Lent | (Principal Financial and Accounting Officer) | |||
| /s/ Lawrence Steinman | Director | March 31, 2025 | ||
| Lawrence Steinman | ||||
| /s/ Ryan L. Smith | Lead Director | March 31, 2025 | ||
| Ryan L. Smith | ||||
| /s/ Jay Goodman | Director | March 31, 2025 | ||
| Jay Goodman | ||||
| /s/ Stephen Shoemaker | Director | March 31, 2025 | ||
| Stephen Shoemaker |
114
180 LIFE SCIENCES CORP. AND SUBSIDIARIES
CONSOLIDATED FINANCIAL STATEMENTS AS OF DECEMBER 31, 2024 AND 2023
TABLE OF CONTENTS
F-1

REPORT OF INDEPENDENT REGISTERED PUBLIC ACCOUNTING FIRM
To the Board of Directors and Stockholders of 180 Life Sciences Corp and Subsidiaries
Opinion on the Consolidated Financial Statements
We have audited the accompanying consolidated balance sheets of 180 Life Sciences Corp. (the Company) as of December 31, 2024 and the related consolidated statements of operations and comprehensive loss, changes in stockholders’ equity (deficit), and cash flows for the year-ended December 31, 2024 and the related notes (collectively referred to as the "financial statements"). We have audited note 3 in regards to the presentation of segment information for the year-ended December 31, 2023. In our opinion, the consolidated financial statements referred to above present fairly, in all material respects, the financial position of the Company as of December 31, 2024, and the results of its operations and its cash flows for the year-ended December 31, 2024, in conformity with accounting principles generally accepted in the United States of America. The financial statements of 180 Life Sciences Corp., as of December 31, 2023, were audited by other auditors whose report dated March 22, 2024, expressed an unqualified opinion on those financial statements.
Going Concern
The accompanying consolidated financial statements have been prepared assuming that the Company will continue as a going concern. As discussed in Note 2 to the consolidated financial statements, the Company suffered a net loss from operations and has insufficient revenues and income to fully fund the operations, which raises substantial doubt about its ability to continue as a going concern. Management’s plans regarding those matters are also described in Note 2. The consolidated financial statements do not include any adjustments that might result from the outcome of this uncertainty.
Basis for Opinion
These consolidated financial statements are the responsibility of the Company’s management. Our responsibility is to express an opinion on the Company’s consolidated financial statements based on our audits. We are a public accounting firm registered with the Public Company Accounting Oversight Board (United States) (PCAOB) and are required to be independent with respect to the Company in accordance with the U.S. federal securities laws and the applicable rules and regulations of the Securities and Exchange Commission and the PCAOB.
We conducted our audits in accordance with the standards of the PCAOB. Those standards require that we plan and perform the audits to obtain reasonable assurance about whether the consolidated financial statements are free of material misstatement, whether due to error or fraud. The Company is not required to have, nor were we engaged to perform, an audit of its internal control over financial reporting. As part of our audits, we are required to obtain an understanding of internal control over financial reporting, but not for the purpose of expressing an opinion on the effectiveness of the Company’s internal control over financial reporting. Accordingly, we express no such opinion.
Our audits included performing procedures to assess the risks of material misstatement of the consolidated financial statements, whether due to error or fraud, and performing procedures that respond to those risks. Such procedures included examining, on a test basis, evidence regarding the amounts and disclosures in the consolidated financial statements. Our audits also included evaluating the accounting principles used and the significant estimates made by management, as well as evaluating the overall presentation of the consolidated financial statements. We believe our audits provide a reasonable basis for our opinion.
F-2
Critical Audit Matter
The critical audit matter communicated below is a matter arising from the current period audits of the consolidated financial statements that were communicated, or required to be communicated, to the audit committee and that: (1) relate to accounts or disclosures that are material to the consolidated financial statements and (2) involved our especially challenging, subjective, or complex judgments. The communication of critical audit matter does not alter in any way our opinion on the consolidated financial statements, taken as a whole, and we are not, by communicating the critical audit matter below, providing separate opinion on the critical audit matter or on the accounts or disclosures to which they relate.
Evaluation of Intangible Assets
As discussed in Note 3 and 5 to the consolidated financial statements, the Company had intellectual property that was being amortized and had indications of impairment. The Company was required to assess for impairment based on the facts and circumstances at that time. Auditing management’s evaluation of intangible assets can be a significant judgment given the fact that the Company uses management estimates on future revenues and expenses which are not easily able to be substantiated.
Given these factors and due to significant judgements made by management, the related audit effort in evaluating management’s judgments in evaluation of intangible assets required a high degree of auditor judgment.
The procedures performed included evaluation of the methods and assumptions used by the Company, tests of the data used and an evaluation of the findings. We evaluated and tested the Company’s significant judgments that determine the impairment evaluation of intangible assets.
/s/ M&K CPAS, PLLC
We have served as the Company’s auditor since 2024
The Woodlands, Texas
March 31, 2025
PCAOB ID #2738
F-3
REPORT OF INDEPENDENT REGISTERED PUBLIC ACCOUNTING FIRM
To the Shareholders and Board of Directors of
180 Life Sciences Corp.
Opinion on the Financial Statements
We have audited the accompanying consolidated balance sheet of 180 Life Sciences Corp. (the “Company”) as of December 31, 2023, the related consolidated statements of operations and comprehensive loss, changes in stockholders’ equity (deficit) and cash flows for the year ended December 31, 2023, and the related notes (collectively referred to as the “financial statements”). In our opinion, the financial statements present fairly, in all material respects, the financial position of the Company as of December 31, 2023, and the results of its operations and its cash flows for the year ended December 31, 2023, in conformity with accounting principles generally accepted in the United States of America.
Explanatory Paragraph – Going Concern
The accompanying financial statements have been prepared assuming that the Company will continue as a going concern. As more fully described in Note 2, the Company has a significant working capital deficiency, has incurred significant losses and needs to raise additional funds to meet its obligations and sustain its operations. These conditions raise substantial doubt about the Company's ability to continue as a going concern. Management's plans in regard to these matters are also described in Note 2. The financial statements do not include any adjustments that might result from the outcome of this uncertainty.
Basis for Opinion
These financial statements are the responsibility of the Company's management. Our responsibility is to express an opinion on the Company's financial statements based on our audit. We are a public accounting firm registered with the Public Company Accounting Oversight Board (United States) ("PCAOB") and are required to be independent with respect to the Company in accordance with the U.S. federal securities laws and the applicable rules and regulations of the Securities and Exchange Commission and the PCAOB.
We conducted our audit in accordance with the standards of the PCAOB. Those standards require that we plan and perform the audit to obtain reasonable assurance about whether the financial statements are free of material misstatement, whether due to error or fraud. The Company is not required to have, nor were we engaged to perform, an audit of its internal control over financial reporting. As part of our audit we are required to obtain an understanding of internal control over financial reporting but not for the purpose of expressing an opinion on the effectiveness of the Company's internal control over financial reporting. Accordingly, we express no such opinion.
Our audit included performing procedures to assess the risks of material misstatement of the financial statements, whether due to error or fraud, and performing procedures that respond to those risks. Such procedures included examining, on a test basis, evidence regarding the amounts and disclosures in the financial statements. Our audit also included evaluating the accounting principles used and significant estimates made by management, as well as evaluating the overall presentation of the financial statements. We believe that our audit provides a reasonable basis for our opinion.
Critical Audit Matters
Critical audit matters are matters arising from the current period audit of the financial statements that were communicated or required to be communicated to the audit committee and that: (1) relate to accounts or disclosures that are material to the financial statements and (2) involved our especially challenging, subjective, or complex judgments. We determined that there are no critical audit matters.
/s/ Marcum llp
Marcum llp
We have served as the Company’s auditor since 2019 through 2024.
San Francisco, California
March 22, 2024
F-4
180 LIFE SCIENCES CORP. AND SUBSIDIARIES
CONSOLIDATED BALANCE SHEETS
(Expressed in US Dollars)
| December 31, | December 31, | |||||||
| 2024 | 2023 | |||||||
| Current Assets: | ||||||||
| Cash | $ | | $ | | ||||
| Prepaid expenses and other current assets | ||||||||
| Total Current Assets | ||||||||
| Intangible assets, net | ||||||||
| Total Assets | $ | $ | ||||||
| Liabilities and Stockholders' Equity (Deficit) | ||||||||
| Current Liabilities: | ||||||||
| Accounts payable | $ | $ | ||||||
| Accounts payable - related parties | ||||||||
| Accrued expenses | ||||||||
| Loans payable - current portion | ||||||||
| Derivative liabilities | ||||||||
| Total Current Liabilities | ||||||||
| Loans payable - noncurrent portion | ||||||||
| Deferred tax liability | ||||||||
| Total Liabilities | ||||||||
| Commitments and contingencies (Note 10) | ||||||||
| Stockholders' Equity (Deficit): | ||||||||
| Preferred stock, $ | ||||||||
| Class C Preferred Stock; | ||||||||
| Class K Preferred Stock; | ||||||||
| Common stock, $ | ||||||||
| Additional paid-in capital | ||||||||
| Accumulated other comprehensive income | ( | ) | ( | ) | ||||
| Accumulated deficit | ( | ) | ( | ) | ||||
| Total Stockholders' Equity (Deficit) | ( | ) | ||||||
| Total Liabilities and Stockholders' Equity (Deficit) | $ | $ | ||||||
The accompanying notes are an integral part of these consolidated financial statements.
F-5
180 LIFE SCIENCES CORP. AND SUBSIDIARIES
CONSOLIDATED STATEMENTS OF OPERATIONS AND COMPREHENSIVE LOSS
(Expressed in US Dollars)
| For the Years Ended | ||||||||
| December 31, | ||||||||
| 2024 | 2023 | |||||||
| Operating Expenses: | ||||||||
| Research and development | $ | $ | ||||||
| Research and development - related parties | ||||||||
| General and administrative | ||||||||
| General and administrative - related parties | ||||||||
| Total Operating Expenses | ||||||||
| Loss From Operations | ( | ) | ( | ) | ||||
| Other (Expense) Income: | ||||||||
| Other income | ||||||||
| Interest expense | ( | ) | ( | ) | ||||
| Loss on asset impairment | ( | ) | ( | ) | ||||
| Change in fair value of derivative liabilities | ||||||||
| Change in fair value of preferred stock liability | ( | ) | ||||||
| Gain on settlement of liabilities | ||||||||
| Total other (expense) income, net | ( | ) | ( | ) | ||||
| Loss Before Income Taxes | ( | ) | ( | ) | ||||
| Income tax benefit | ||||||||
| Net Loss | ( | ) | ( | ) | ||||
| Deemed Dividend | ( | ) | ||||||
| Net Loss Available to Common Stockholders | ( | ) | ( | ) | ||||
| Other Comprehensive Loss: | ||||||||
| Foreign currency translation adjustments | ( | ) | ( | ) | ||||
| Total Comprehensive Loss | $ | ( | ) | $ | ( | ) | ||
| Basic and Diluted Net Loss per Common Share | ||||||||
| Basic | $ | ( | ) | $ | ( | ) | ||
| Diluted | $ | ( | ) | $ | ( | ) | ||
| Weighted Average Number of Common Shares Outstanding: | ||||||||
| Basic | ||||||||
| Diluted | ||||||||
The accompanying notes are an integral part of these consolidated financial statements.
F-6
180 LIFE SCIENCES CORP. AND SUBSIDIARIES
CONSOLIDATED STATEMENTS OF CHANGES IN STOCKHOLDERS’ EQUITY (DEFICIT)
(Expressed in US Dollars)
| For The Year Ended December 31, 2023 | ||||||||||||||||||||||||||||||||
| Series B Preferred Stock | Common Stock | Additional Paid-in | Accumulated Other Comprehensive | Accumulated | Total Equity | |||||||||||||||||||||||||||
| Shares | Amount | Shares | Amount | Capital | Income | Deficit | (Deficit) | |||||||||||||||||||||||||
| Balance - January 1, 2023 | $ | $ | $ | $ | ( | ) | $ | ( | ) | $ | | |||||||||||||||||||||
| Adjustment related to reverse stock split | ( | ) | ||||||||||||||||||||||||||||||
| Shares issued for professional services to directors | ||||||||||||||||||||||||||||||||
| Shares issued for professional services to executive chairman | ||||||||||||||||||||||||||||||||
| Issuance of April 2023 pre-funded and common warrants, net (a) | - | - | ||||||||||||||||||||||||||||||
| Shares issued from exercise of April 2023 pre-funded warrants (a) | ||||||||||||||||||||||||||||||||
| Shares issued in connection with April 2023 Offering (a) | ||||||||||||||||||||||||||||||||
| Issuance of August 2023 pre-funded and common warrants, net (b) | - | - | ||||||||||||||||||||||||||||||
| Shares issued from exercise of August 2023 pre-funded warrants (b) | ||||||||||||||||||||||||||||||||
| Shares issued in connection with August 2023 Offering (b) | ||||||||||||||||||||||||||||||||
| Issuance of pre-funded and common warrants from Amendment to August 2023 SPA, net | - | - | ||||||||||||||||||||||||||||||
| Stock-based compensation | - | - | ||||||||||||||||||||||||||||||
| Comprehensive loss: | - | - | ||||||||||||||||||||||||||||||
| Net loss | - | - | ( | ) | ( | ) | ||||||||||||||||||||||||||
| Other comprehensive loss | - | - | ( | ) | ( | ) | ||||||||||||||||||||||||||
| Balance - December 31, 2023 | $ | $ | $ | $ | ( | ) | $ | ( | ) | $ | ( | ) | ||||||||||||||||||||
| (a) |
| (b) |
F-7
| For The Year Ended December 31, 2024 | ||||||||||||||||||||||||||||||||
| Series
B Preferred Stock | Common Stock | Additional Paid-in | Accumulated Other Comprehensive | Accumulated | Total Stockholders’ | |||||||||||||||||||||||||||
| Shares | Amount | Shares | Amount | Capital | Income | Deficit | Equity | |||||||||||||||||||||||||
| Balance - January 1, 2024 | $ | $ | $ | $ | ( | ) | $ | ( | ) | $ | ( | ) | ||||||||||||||||||||
| Shares issued from exercise of pre-funded warrants related to Amendment to August 2023 Offering | ||||||||||||||||||||||||||||||||
| Adjustment related to reverse stock split in February 2024 | ( | ) | ||||||||||||||||||||||||||||||
| Shares issued in connection with December 2024 Offering | ||||||||||||||||||||||||||||||||
| Shares issued from exercise of warrants | ||||||||||||||||||||||||||||||||
| Shares issued for settlement of liabilities | ||||||||||||||||||||||||||||||||
| Stock based compensation | - | - | ||||||||||||||||||||||||||||||
| Warrants and Series B Convertible Preferred Stock issued for asset acquisition | - | - | ||||||||||||||||||||||||||||||
| Reclassification of Series B Convertible Preferred Stock liability | ||||||||||||||||||||||||||||||||
| Deemed dividend from warrant inducement | - | - | ( | ) | ||||||||||||||||||||||||||||
| Comprehensive income (loss): | ||||||||||||||||||||||||||||||||
| Net loss | - | - | ( | ) | ( | ) | ||||||||||||||||||||||||||
| Other comprehensive loss | - | - | ( | ) | ( | ) | ||||||||||||||||||||||||||
| Balance - December 31, 2024 | $ | $ | $ | $ | ( | ) | $ | ( | ) | $ | ||||||||||||||||||||||
The accompanying notes are an integral part of these consolidated financial statements.
F-8
180 LIFE SCIENCES CORP. AND SUBSIDIARIES
CONSOLIDATED STATEMENTS OF CASH FLOWS
(Expressed in US Dollars)
| For the Years Ended | ||||||||
| December 31, | ||||||||
| 2024 | 2023 | |||||||
| Cash Flows from Operating Activities | ||||||||
| Net Loss | $ | ( | ) | $ | ( | ) | ||
| Adjustments to reconcile net loss to net cash used in operating activities: | ||||||||
| Stock-based compensation | ||||||||
| Shares issued for services | ||||||||
| Amortization of stock options and restricted stock units | ||||||||
| Depreciation and amortization | ||||||||
| Loss on asset impairment | ||||||||
| Change in fair value of derivative liabilities | ( | ) | ( | ) | ||||
| Change in fair value of preferred stock liability | ||||||||
| Gain on settlement of liabilities | ( | ) | ||||||
| Deferred tax benefit | ( | ) | ( | ) | ||||
| Changes in operating assets and liabilities: | ||||||||
| Prepaid expenses and other current assets | ||||||||
| Accounts payable | ( | ) | ||||||
| Accounts payable - related parties | ||||||||
| Accrued expenses | ( | ) | ( | ) | ||||
| Accrued expenses - related parties | ( | ) | ||||||
| Total adjustments | ||||||||
| Net Cash Used In Operating Activities | ( | ) | ( | ) | ||||
| Cash Flows from Financing Activities | ||||||||
| Proceeds from exercise of pre-funded warrants from the Amendment to August 2023 Offering | ||||||||
| Proceeds from sale of December 2024 Offering stock and warrants | ||||||||
| Proceeds from the exercise of warrants | ||||||||
| Proceeds from sale of April 2023 Offering stock and warrants | ||||||||
| Proceeds from sale of August 2023 Offering stock and warrants | ||||||||
| Proceeds from exercise of April 2023 Offering pre-funded warrants | ||||||||
| Proceeds from exercise of August 2023 Offering pre-funded warrants | ||||||||
| Proceeds from the repricing of common shares and pre-funded warrants in the August 2023 Offering | ||||||||
| Proceeds from loans payable | ||||||||
| Payment of offering costs in connection with April 2023 Offering stock and warrants | ( | ) | ||||||
| Payment of offering costs in connection with August 2023 Offering stock and warrants | ( | ) | ||||||
| Payment of offering costs in connection with August 2023 sale of common stock and common stock warrants | ( | ) | ||||||
| Repayment of loans payable | ( | ) | ( | ) | ||||
| Net cash provided by financing activities | ||||||||
| Effect of Exchange Rate Changes on Cash | ||||||||
| Net Decrease In Cash | ( | ) | ||||||
| Cash - Beginning of Period | ||||||||
| Cash - End of Period | $ | $ | ||||||
| Supplemental Disclosures of Cash Flow Information: | ||||||||
| Cash paid during the period for income taxes | $ | $ | ||||||
| Cash paid during the period for interest expense | $ | $ | ||||||
| Non cash investing and financing activities | ||||||||
| Common stock issued for settlement of liabilities | $ | $ | ||||||
| Prepaid expense financed with notes payable | $ | $ | ||||||
| Warrants and Series B Convertible Preferred Stock issued for asset acquisition | $ | |||||||
| Deemed dividend from warrant inducement | $ | $ | ||||||
The accompanying notes are an integral part of these consolidated financial statements.
F-9
180 LIFE SCIENCES CORP. AND SUBSIDIARIES
NOTES TO THE CONSOLIDATED FINANCIAL STATEMENTS
(Amounts in US Dollars, except share amounts)
NOTE 1 - BUSINESS ORGANIZATION AND NATURE OF OPERATIONS
180 Life Sciences Corp., formerly
known as KBL Merger Corp. IV (“180LS”, or together with its subsidiaries, the “Company”), was a blank check company
organized under the laws of the State of Delaware on September 7, 2016. The Company was originally formed for the purpose of effecting
a merger, share exchange, asset acquisition, share purchase, reorganization, or similar business combination with
180 Life Corp. (“180”, f/k/a 180 Life Sciences Corp. and CannBioRx Life Sciences Corp.) is a wholly-owned subsidiary of the Company and was incorporated in the State of Delaware on January 28, 2019. The Company is located in the United States (“U.S.”) and is a medical pharmaceutical company focused upon unmet medical needs in the areas of inflammatory diseases, fibrosis, and chronic pain by employing innovative research and, where appropriate, combination therapies, through 180’s three wholly-owned subsidiaries, 180 Therapeutics L.P. (“180 LP”), CannBioRex Pharmaceuticals Corp. (“CBR Pharma”), and Katexco Pharmaceuticals Corp. (“Katexco”). 180 LP, CBR Pharma and Katexco are together, the “180 Subsidiaries.” Katexco was incorporated on March 7, 2018 under the provisions of the British Corporation Act of British Columbia. Additionally, 180’s wholly-owned subsidiaries Katexco Callco, ULC, Katexco Purchaseco, ULC, CannBioRex Callco, ULC, and CannBioRex Purchaseco, ULC were formed in the Canadian Province of British Columbia on May 31, 2019 to facilitate the acquisition of Katexco, CBR Pharma and 180 LP. On July 1, 2021, the assets and liabilities of the Canadian companies (Katexco and CBR Pharma) were transferred to their respective subsidiaries, which are Katexco Pharmaceuticals Corp. (“Katexco U.S.”) and CannBioRex Pharma Limited (“CBR Pharma U.K.”). In July 2019, 180 and each of 180 LP, Katexco and CBR Pharma completed a corporate restructuring, pursuant to which 180 LP, Katexco and CBR Pharma became wholly-owned subsidiaries of 180 (the “Reorganization”).
The Company has been a clinical stage biotechnology company focused on the development of therapeutics for unmet medical needs in chronic pain, inflammation, fibrosis, and other inflammatory diseases, where anti-TNF therapy will provide a clear benefit to patients, by employing innovative research, and, where appropriate, combination therapy. We had three product development platforms:
| ● | fibrosis and anti-tumor necrosis factor (“TNF”); |
| ● | drugs which are derivatives of cannabidiol (“CBD”); and |
| ● | alpha 7 nicotinic acetylcholine receptor (“α7nAChR”). |
In November 2024, the biotechnology related to α7nAChR was returned to Stanford University.
On September 29, 2024, the Company entered into an Asset Purchase Agreement (the “Purchase Agreement”) with Elray Resources, Inc. (“Elray”). Pursuant to the Purchase Agreement, Elray agreed to sell us certain source code and intellectual property relating to a “back-end technology platform” which is a key component of creating an online blockchain casino (the “Purchased Assets”) Elray agreed to provide support and assistance to the Company in connection with the building and launching of a fully operational casino operation utilizing the Purchased Assets.
Risks and Uncertainties
Management continues to evaluate the impact of the Russia-Ukraine war, the Israel-Hamas war and fluctuating interest rates and inflation on the economy and the capital markets and has concluded that, while it is reasonably possible that such events could have negative effects on the Company’s financial position, the specific impacts are not readily determinable as of the date of these consolidated financial statements. The consolidated financial statements do not include any adjustments that might result from the outcome of these uncertainties.
F-10
The current challenging economic climate may lead to adverse changes in cash flows, working capital levels and/or debt balances, which may also have a direct impact on the Company’s future operating results and financial position. The ultimate duration and magnitude of the impact and the efficacy of government interventions on the economy and the financial effect on the Company is not known at this time. The extent of such impact will depend on future developments, which are highly uncertain and not in the Company’s control.
Reverse Stock-Split during 2024
On February 26, 2024, the Company’s Board of Directors approved a one-for-19 reverse stock split of the Company’s outstanding common stock (the “Reverse Stock Split”) and the filing of a Certificate of Amendment to our Certificate of Incorporation to affect such reverse stock split, which was filed on February 26, 2024. The Reverse Stock Split became effective on February 28, 2024 at 12:01 a.m. Eastern Time (the “Effective Time”), with shares beginning trading on a split-adjusted basis at market open on February 28, 2024. In connection with the Reverse Stock Split, every 19 shares of the Company’s common stock issued and outstanding as of the Effective Time were automatically converted into one share of the Company’s common stock.
In connection with the Reverse
Stock Split, all outstanding options, warrants, and other securities entitling their holders to purchase or otherwise receive shares of
common stock were adjusted, as required by the terms of each security. The number of shares available to be awarded under the Company’s
equity incentive plans were also appropriately adjusted. Following the Reverse Stock Split, the par value of the Common Stock remained
unchanged at $
The effects of the 1-for-19 Reverse Stock Split have been retroactively reflected throughout this Report.
NOTE 2 - GOING CONCERN AND MANAGEMENT’S PLANS
The Company has not generated
any revenues and has incurred significant losses since inception. As of December 31, 2024, the Company had an accumulated deficit of $
These consolidated financial statements have been prepared under the assumption of a going concern, which assumes that the Company will be able to realize its assets and discharge its liabilities in the normal course of business. The Company’s ability to continue its operations is dependent upon obtaining new financing for its ongoing operations. Future financing options available to the Company include equity financings and loans and if the Company is unable to obtain such additional financing timely, or on favorable terms, the Company may have to curtail its development, marketing and promotional activities, which would have a material adverse effect on its business, financial condition and results of operations, and it could ultimately be forced to discontinue its operations and liquidate. These matters raise substantial doubt about the Company’s ability to continue as a going concern for a reasonable period of time, which is defined as within one year after the date that the consolidated financial statements are issued. Realization of the Company’s assets may be substantially different from the carrying amounts presented in these consolidated financial statements and the accompanying consolidated financial statements do not include any adjustments that may become necessary, should the Company be unable to continue as a going concern.
F-11
NOTE 3 - SUMMARY OF SIGNIFICANT ACCOUNTING POLICIES
Basis of Presentation
The accompanying consolidated financial statements have been prepared in accordance with accounting principles generally accepted in the Unites States of America (“U.S. GAAP”).
Principles of Consolidation
The consolidated financial statements include the accounts of the Company and its wholly-owned subsidiaries 180 LP, CBR Pharma, Katexco and 180 Life Corp. (“180LC”). All inter-company balances and transactions among the companies have been eliminated upon consolidation. The consolidated financial statements are presented in U.S. Dollars.
Use of Estimates
The preparation of financial statements in conformity with U.S. GAAP requires management to make estimates, judgments, and assumptions that affect the reported amounts of assets, liabilities, revenues and expenses, together with amounts disclosed in the related notes to the consolidated financial statements. The Company’s significant estimates and assumptions used in these financial statements include, but are not limited to, the fair value of financial instruments, warrants, options and derivative liabilities; R&D tax credits and accruals, and the estimates and assumptions related to the impairment analysis of IP R&D assets. Certain of the Company’s estimates could be affected by external conditions, including those unique to the Company and general economic conditions. It is reasonably possible that these external factors could have an effect on the Company’s estimates and may cause actual results to differ from those estimates.
Foreign Currency Translation
The Company’s reporting
currency is the United States dollar. The functional currency of certain subsidiaries is the British Pound (“GBP”). Assets
and liabilities are translated based on the exchange rates at the balance sheet date (
Comprehensive income is defined
as the change in equity of an entity from all sources other than investments by owners or distributions to owners and includes foreign
currency translation adjustments as described above. During the years ended December 31, 2024 and 2023, the Company recorded other comprehensive
loss of $
Foreign currency gains and losses resulting from transactions denominated
in foreign currencies, including intercompany transactions, are included in results of operations. The Company recognized a $
Cash and Cash Equivalents
The Company considers all
highly liquid investments with an original maturity of three months or less to be cash equivalents in the financial statements. The Company
had no cash equivalents at December 31, 2024 or 2023. As of December 31, 2024, the Company had bank accounts in the United States and
the United Kingdom; of its available cash balance, $
F-12
Goodwill
Goodwill represents the difference between the purchase price and the fair value of assets and liabilities acquired in a business combination. The Company reviews goodwill yearly, or more frequently whenever circumstances and situations change such that there is an indication that the carrying amounts may not be recovered, for impairment by initially considering qualitative factors to determine whether it is more likely than not that the fair value of a reporting unit is less than its carrying amount, including goodwill, as a basis for determining whether it is necessary to perform a quantitative analysis. If it is determined that it is more likely than not that the fair value of a reporting unit is less than its carrying amount, a quantitative analysis is performed to identify goodwill impairment. If it is determined that it is not more likely than not that the fair value of the reporting unit is less than its carrying amount, it is unnecessary to perform a quantitative analysis. The Company may elect to bypass the qualitative assessment and proceed directly to performing a quantitative analysis. See “Note 5 – Intangible Assets and Impairment of Long-lived Assets” for further information.
Intangible Assets and In-Process Research and Development (“IP R&D”)
Intangible assets consist
of licensed patents as well as technology licenses acquired in connection with the Reorganization. Licensed patents are
amortized over the remaining life of the patent. Technology licenses represent the fair value of licenses acquired for the development
and commercialization of certain licenses and knowledge. The technology licenses are amortized on a straight-line basis over the estimated
useful lives of the underlying patents. It will be necessary to monitor and possibly adjust the useful lives of the licensed patents and
technology licenses depending on the results of the Company’s research and development activities. During the year ended December 31, 2024, the Company determined that the licensed patents and technology licenses
related to its previous drug research programs were impaired, and recognized a loss on impairment of $
IP R&D assets represent
the fair value assigned to technologies that were acquired on July 16, 2019 in connection with the Reorganization, which have not reached
technological feasibility and have no alternative future use. IP R&D assets are considered to have indefinite-lives until the completion
or abandonment of the associated research and development projects. During the period that the IP R&D assets are considered indefinite-lived,
they are tested for impairment on an annual basis, or more frequently if the Company becomes aware of any events occurring or changes
in circumstances that indicate that the fair value of the IP R&D assets are less than their carrying amounts. If and when development
is complete, which generally occurs upon regulatory approval, and the Company is able to commercialize products associated with the IP
R&D assets, these assets are then deemed definite-lived and are amortized based on their estimated useful lives at that point in time.
If development is terminated or abandoned, the Company may record a full or partial impairment charge related to the IP R&D assets,
calculated as the excess of the carrying value of the IP R&D assets over their estimated fair value. During 2023, the Company recorded
a loss on impairment to IP R&D assets in the amount of $
Fair Value of Financial Instruments
The Company measures the fair value of financial assets and liabilities based on the guidance of Accounting Standards Codification (“ASC”) 820 “Fair Value Measurements” (“ASC 820”), which defines fair value, establishes a framework for measuring fair value, and expands disclosures about fair value measurements.
F-13
ASC 820 defines fair value as the exchange price that would be received for an asset or paid to transfer a liability (an exit price) in the principal or most advantageous market for the asset or liability in an orderly transaction between market participants on the measurement date. ASC 820 also establishes a fair value hierarchy, which requires an entity to maximize the use of observable inputs and minimize the use of unobservable inputs when measuring fair value. ASC 820 describes three levels of inputs that may be used to measure fair value:
| ● | Level 1 - Quoted prices in active markets for identical assets or liabilities; | |
| ● | Level 2 - Quoted prices for similar assets and liabilities in active markets or inputs that are observable; and | |
| ● | Level 3 - Inputs that are unobservable (for example, cash flow modeling inputs based on assumptions). |
The carrying amounts of certain of the Company’s financial instruments, consisting primarily of loans payable, approximate their fair values as presented in these consolidated financial statements due to the short-term nature of those instruments. The Company’s derivative liabilities were valued using level 3 inputs (see Note 7 – Derivative Liabilities for additional information).
Stock-Based Compensation
The Company measures the cost of services received in exchange for an award of equity instruments based on the fair value of the award. The fair value of the award is measured on the grant date and is estimated by management based on observations of the recent cash sales prices of common stock. The fair value amount is then recognized over the period during which services are required to be provided in exchange for the award, usually the vesting period. Upon the exercise of an option or warrant, the Company issues new shares of common stock out of its authorized but unissued shares.
Derivative Liabilities and Convertible Instruments
The Company evaluates its debt and equity issuances to determine if those contracts or embedded components of those contracts qualify as derivatives requiring separate recognition in the Company’s financial statements. Entities must consider whether to classify contracts that may be settled in its own stock, such as warrants, as equity of the entity or as an asset or liability. If an event that is not within the entity’s control could require net cash settlement, then the contract should be classified as an asset or a liability rather than as equity.
The result of this accounting treatment is that the fair value of the embedded derivative is marked-to-market at each balance sheet date and recorded as a liability and the change in fair value is recorded in other (expense) income, net in the consolidated statements of operations. In circumstances where there are multiple embedded instruments that are required to be bifurcated, the bifurcated derivative instruments are accounted for as a single, compound derivative instrument. The classification of derivative instruments, including whether such instruments should be recorded as liabilities or as equity, is reassessed at the end of each reporting period. Equity instruments that are initially classified as equity that become subject to reclassification are reclassified to liability at the fair value of the instrument on the reclassification date. Derivative instrument liabilities are classified in the balance sheet as current or non-current based on whether or not net-cash settlement of the derivative instrument is expected within twelve months of the balance sheet date.
If the embedded conversion options do not require bifurcation, the Company then evaluates for the existence of a beneficial conversion feature by comparing the fair value of the Company’s underlying stock as of the commitment date to the effective conversion price of the instrument (the intrinsic value).
Debt discounts under these arrangements are amortized over the term of the related debt to their stated date of redemption and are classified in interest expense in the consolidated statements of operations. Preferred stock discounts are only accreted to their redemption value if redemption becomes probable.
Amendments to convertible instruments are evaluated as to whether they should be accounted for as a modification of the original instrument with no change to the accounting or, if the terms are substantially changed, as an extinguishment of the original instrument and the issuance of a new instrument.
F-14
The Company has computed the fair value of warrants and options issued using the Black-Scholes option pricing model. The expected term used for warrants, convertible notes and convertible preferred stock are the contractual life and the expected term used for options issued is the estimated period of time that options granted are expected to be outstanding. The Company utilizes the “simplified” method to develop an estimate of the expected term of “plain vanilla” option grants. The Company is utilizing an expected volatility figure based on a review of the historical volatilities, over a period of time, equivalent to the expected life of the instrument being valued, of similarly positioned public companies within its industry. The risk-free interest rate was determined from the implied yields from U.S. Treasury zero-coupon bonds with a remaining term consistent with the expected term of the instrument being valued.
Net Loss Per Common Share
Basic net loss per common share is computed by dividing net loss by the weighted average number of common shares outstanding during the period. Diluted net loss per common share is computed by dividing net loss by the weighted average number of common shares outstanding, plus the number of additional common shares that would have been outstanding if the common share equivalents had been issued (computed using the treasury stock or if converted method), if dilutive.
The following common share equivalents are excluded from the calculation of weighted average common shares outstanding, because their inclusion would have been anti-dilutive:
| For the Years Ended December 31, | ||||||||
| 2024 | 2023 | |||||||
| Options | ||||||||
| Warrants | (1) | |||||||
| Total potentially dilutive shares | ||||||||
| (1) |
Research and Development
Research and development expenses
are charged to operations as incurred. During the years ended December 31, 2024 and 2023, the Company incurred $
Income Taxes
The Company accounts for income taxes under the provisions of ASC Topic 740 “Income Taxes” (“ASC 740”).
The Company recognizes deferred tax assets and liabilities for the expected future tax consequences of items that have been included or excluded in the financial statements or tax returns. Deferred tax assets and liabilities are determined on the basis of the difference between the tax basis of assets and liabilities and their respective financial reporting amounts (“temporary differences”) at enacted tax rates in effect for the years in which the temporary differences are expected to reverse.
The Company utilizes a recognition threshold and measurement process for financial statement recognition and measurement of a tax position taken or expected to be taken in a tax return. The Company’s policy is to classify assessments, if any, for tax related interest as interest expense and penalties as general and administrative expenses in the consolidated statements of operations and comprehensive loss.
Segment Reporting
ASC Topic 280, “Segment Reporting,” requires annual and interim reporting for an enterprise’s operating segments and related disclosures about its products, services, geographic areas and major customers. An operating segment is defined as a component of an enterprise that engages in business activities from which it may earn revenues and expenses, and about which separate financial information is regularly evaluated by the chief operating decision maker in deciding how to allocate resources. In November 2023, the FASB issued ASU No. 2023-07, “Segment Reporting (Topic 280): Improvements to Reportable Segment Disclosure.” The ASU updates reportable segment disclosure requirements, primarily through requiring enhanced disclosures about significant segment expenses and information used to assess segment performance. The amendments do not change how segments are determined, aggregated, or how thresholds are applied to determine reportable segments. The Company adopted ASU No. 2023-07 during the year ended December 31, 2024.
F-15
Segment information is prepared
on the same basis that our CEO, who is our Chief Operating Decision Maker (“CODM”), manages our segments, evaluates financial
results, and makes key operating decisions. We have
Recently Issued Accounting Pronouncements
In November 2023, the Financial Accounting Standards Board (FASB) issued Accounting Standards Update (ASU) No. 2023-07, “Segment Reporting (Topic 280): Improvements to Reportable Segment Disclosure.” The ASU updates reportable segment disclosure requirements, primarily through requiring enhanced disclosures about significant segment expenses and information used to assess segment performance. The amendments do not change how segments are determined, aggregated, or how thresholds are applied to determine reportable segments. We adopted ASU No. 2023-07 during the year ended December 31, 2024.
In December 2023, FASB issued ASU No. 2023-09 “Income Taxes (Topic 740): Improvements to Income Tax Disclosures.” ASU 2023-09 requires incremental annual disclosures around income tax rate reconciliations, income taxes paid and other related disclosures. This guidance requires prospective application and permits retrospective application to prior periods presented. The Company is currently evaluating the impact of the adoption of this standard to determine its impact on the Company's disclosures.
In November 2024, the FASB issued ASU 2024-03, Reporting Comprehensive Income—Expense Disaggregation Disclosures (Subtopic 220-40): Disaggregation of Income Statement Expenses, which is intended to improve disclosures related to certain income statement expenses of the Company. This ASU is effective for fiscal years beginning after December 15, 2026, and interim periods within fiscal years beginning after December 15, 2027, with early adoption permitted. The Company is currently evaluating the impact of the adoption of this standard to determine its impact on the Company's disclosures.
Management does not believe that any other recently issued, but not effective, accounting pronouncements, if currently adopted, would have a material effect on the Company’s consolidated financial statements.
NOTE 4 - PREPAID EXPENSES AND OTHER CURRENT ASSETS
Prepaid expenses and other current assets consist of the following as of December 31, 2024 and 2023:
| December 31, | ||||||||
| 2024 | 2023 | |||||||
| Insurance | $ | $ | ||||||
| Research and development expense tax credit receivable | ||||||||
| Professional fees | ||||||||
| Value-added tax receivable | ||||||||
| $ | $ | |||||||
NOTE 5 - INTANGIBLE ASSETS AND IMPAIRMENT OF LONG-LIVED ASSETS
On September 29, 2024, the
Company entered into the Purchase Agreement with Elray. Pursuant to the Purchase Agreement, Elray agreed to sell us the Purchased Assets in
consideration for
On September 30, 2024,
in contemplation of the closing of the transactions contemplated by the Purchase Agreement, and pursuant to the power provided to the
Company by the Certificate of Incorporation of the Company, as amended, the Company’s Board of Directors approved the adoption of,
and filing of, a Certificate of Designations of 180 Life Sciences Corp. Establishing the Designations, Preferences, Limitations and Relative
Rights of Its Series B Convertible Preferred Stock (the “Series B Designation”), which was filed with, and became effective
with, the Secretary of State of Delaware on the same date. The Series B Designation designated
The purchase of the Purchased Assets closed on September 30, 2024.
Based on the terms of
the Series B Preferred Stock and the Purchase Agreement, and in accordance with FASB Accounting Standards Codification (ASC) 480-10,
at issuance the Series B Convertible Preferred Stock was accounted for as a liability due to the variable number of shares issuable
under the agreement pursuant to the conversion terms, with an initial value of $
F-16
The Purchase Warrants have
an exercise price of $
The Company had capitalized
intangible cost of $
The technology acquired from
Elray will be amortized over
The Company will perform its definite-lived intangible asset impairment test on an annual basis with the initial impairment test after an acquisition completed before the expiration of the next 12 month period.
Intangible assets at December 31, 2024 and 2023 consisted of the following:
| Useful | December 31, | December 31, | ||||||||
| Life (years) | 2024 | 2023 | ||||||||
| Source code and intellectual property | $ | $ | ||||||||
| Licensed Patents | ||||||||||
| Technology license | ||||||||||
| Intangible assets | ||||||||||
| Less: accumulated amortization | ( | ) | ( | ) | ||||||
| Less: accumulated impairment loss | ( | ) | ||||||||
| Intangible assets, net | $ | $ | ||||||||
Amortization expense was
$
F-17
IP R&D Assets Impairment
During 2023, the Company assessed
the most recent delays in its commercialization timeline, general economic conditions, industry and market considerations, the Company’s
financial performance and all relevant legal, regulatory, and political factors that might indicate the possibility of impairment and
concluded that, when these factors were collectively evaluated, it was more likely than not that the asset is impaired. The Company recorded
a loss in the amount of $
Because of the write-off of
the IP R&D assets on the balance sheet and the loss on impairment to IP R&D assets on the income statement, the Company recorded
a decrease in its deferred tax liability relating to the impairment of the IP R&D assets of $
The following is a summary of IP R&D activity for the year ended December 31, 2023 for the Company, which includes the recorded losses for the IP R&D assets described above.
| CBR Pharma IP R&D Assets | 180 LP IP R&D Assets | Consolidated IP R&D Assets | ||||||||||
| Balance, December 31, 2023 | $ | $ | ||||||||||
| Impairment of IP R&D assets | ( | ) | ( | ) | ||||||||
| Balance, December 31, 2023 | $ | $ | $ | |||||||||
NOTE 6 - ACCRUED EXPENSES
Accrued expenses consist of the following as of December 31, 2024 and 2023:
| December 31, | ||||||||
| 2024 | 2023 | |||||||
| Consulting fees | $ | $ | ||||||
| Professional fees | ||||||||
| Litigation accrual (1) | ||||||||
| Employee and director compensation | ||||||||
| Research and development fees | ||||||||
| Interest | ||||||||
| Other | ||||||||
| $ | $ | |||||||
| (1) |
F-18
As of December 31, 2024 and
2023, accrued expenses - related parties were
NOTE 7 - DERIVATIVE LIABILITIES
| For the Year Ended December 31, 2024 | ||||||||||||||||||||||||
| Warrants | ||||||||||||||||||||||||
| Public | Private | Alpha | ||||||||||||||||||||||
| SPAC | SPAC | PIPE | AGP | Capital | Total | |||||||||||||||||||
| Balance as of January 1, 2024 | $ | $ | $ | $ | $ | $ | ||||||||||||||||||
| Change in fair value of derivative liabilities | ( | ) | ( | ) | ||||||||||||||||||||
| Balance as of December 31, 2024 | $ | $ | $ | $ | $ | $ | ||||||||||||||||||
| For the Year Ended December 31, 2023 | ||||||||||||||||||||
| Warrants | ||||||||||||||||||||
| Public | Private | |||||||||||||||||||
| SPAC | SPAC | PIPE | AGP | Total | ||||||||||||||||
| Balance as of January 1, 2023 | $ | $ | $ | $ | $ | |||||||||||||||
| Change in fair value of derivative liabilities | ( | ) | ( | ) | ( | ) | ( | ) | ( | ) | ||||||||||
| Balance as of December 31, 2023 | $ | $ | $ | $ | $ | |||||||||||||||
The fair value of the SPAC derivative liabilities as of December 31, 2024 and 2023 were estimated using the Black Scholes option pricing model, with the following assumptions used:
December 31, 2024 | ||||
| Risk-free interest rate | % | |||
| Expected term in years | ||||
| Expected volatility | % | |||
| Expected dividends | % | |||
December 31, 2023 | ||||
| Risk-free interest rate | % | |||
| Expected term in years | ||||
| Expected volatility | % | |||
| Expected dividends | % | |||
F-19
SPAC Warrants
Public SPAC Warrants
Participants in KBL’s
initial public offering received an aggregate of
NOTE 8 - LOANS PAYABLE
The following tables summarize the activity of loans payable during the years ended December 31, 2024 and 2023:
| Principal balance at December 31, 2023 | Principal additions | Principal repayments in cash | Cancellation | Effects of foreign exchange rates | Principal balance at December 31, 2024 | |||||||||||||||||||
| Bounce Back Loan Scheme | $ | $ | $ | ( | ) | $ | $ | ( | ) | $ | ||||||||||||||
| First Insurance - 2023 | ( | ) | ( | ) | ||||||||||||||||||||
| First Insurance - 2024 | ( | ) | ||||||||||||||||||||||
| Other loans payable | ( | ) | ( | ) | ( | ) | ||||||||||||||||||
| Total loans payable | $ | $ | $ | ( | ) | $ | ( | ) | $ | ( | ) | $ | ||||||||||||
| Less: loans payable – current portion | ||||||||||||||||||||||||
| Loans payable – noncurrent portion | $ | $ | ||||||||||||||||||||||
| Principal Balance at January 1, 2023 | Adjustments | Principal Repaid in Cash | New Issuances | Effect of Foreign Exchange Rates | Principal Balance at December 31, 2023 | |||||||||||||||||||
| Bounce Back Loan Scheme | $ | $ | $ | ( | ) | $ | $ | $ | ||||||||||||||||
| First Insurance – 2022 | ( | ) | ||||||||||||||||||||||
| First Insurance – 2023 | ( | ) | ||||||||||||||||||||||
| Other loans payable | ||||||||||||||||||||||||
| Total loans payable | $ | $ | ( | ) | $ | $ | ||||||||||||||||||
| Less: loans payable – current portion | ||||||||||||||||||||||||
| Loans payable – non-current portion | $ | $ | ||||||||||||||||||||||
F-20
Loans Payable, Current Portion
| Simple Interest Rate | December 31, 2024 | December 31, 2023 | ||||||||||
| Loan payable issued September 18, 2019 | | % | $ | $ | ||||||||
| Loan payable issued September 18, 2019 | % | |||||||||||
| Loan payable issued October 8, 2019 | % | |||||||||||
| Loan payable issued October 29, 2019 | % | |||||||||||
| Loan payable issued December 31, 2019 | % | |||||||||||
| Loan payable issued February 5, 2020 | % | |||||||||||
| Loan payable issued February 5, 2020 | % | |||||||||||
| Loan payable issued March 31, 2020 | % | |||||||||||
| Loan payable issued March 31, 2020 | % | |||||||||||
| Loan payable issued June 8, 2020 | % | |||||||||||
| Loan payable issued June 17, 2020 | % | |||||||||||
| Loan payable issued July 15, 2020 * | % | |||||||||||
| Loan payable issued July 15, 2020 | % | |||||||||||
| Loan payable issued October 8, 2020 * | % | |||||||||||
| Loan payable issued October 13, 2020 | % | |||||||||||
| Loan payable issued October 14, 2020 | % | |||||||||||
| Current portion of Bounce Back Loans (1) (2) | % | |||||||||||
| First Insurance Funding payable issued December 2024 and 2023 (2) | % | |||||||||||
| $ | $ | |||||||||||
| * |
| (1) |
| (2) |
Loans Payable, Non-Current Portion
The non-current portion of the Company’s loans payable as of December 31, 2024 and 2023 are as follows:
| Simple Interest Rate | December 31, 2024 | December 31, 2023 | Maturity Date | |||||||||||
| BBLS loan payable issued June 10, 2020 | % | |||||||||||||
| Subtotal | ||||||||||||||
| Less: Current portions of BBLS loans, respectively (see above) | ( | ) | ( | ) | ||||||||||
| Non-current portion | $ | $ | ||||||||||||
F-21
On June 10, 2020, the Company
received GBP £
On December 10, 2022,
the Company entered into a financing arrangement for a Directors and Officers Insurance Policy (the “D&O Insurance”)
with First Insurance Funding to finance $
Interest Expense (Income) on Loans Payable
For the years ended December
31, 2024 and 2023, the Company recognized interest expense — related parties associated with outstanding loans payable of $
As of December 31, 2024, the
Company had accrued interest and accrued interest — related parties associated with outstanding loans payable of $
As of December 31, 2023, the
Company had accrued interest and accrued interest — related parties associated with outstanding loans payable of $
NOTE 9 - SERIES B CONVERTIBLE PREFERRED STOCK LIABILITY
On September 30, 2024,
in contemplation of the closing of the transactions contemplated by the Purchase Agreement, and pursuant to the power provided to the
Company by the Certificate of Incorporation of the Company, as amended, the Company’s Board of Directors approved the adoption of,
and filing of, a Certificate of Designations of 180 Life Sciences Corp. Establishing the Designations, Preferences, Limitations and Relative
Rights of Its Series B Convertible Preferred Stock (the “Series B Designation”), which was filed with, and became effective
with, the Secretary of State of Delaware on the same date. The Series B Designation designated
The below is a summary of the rights and preferences of the Series B Convertible Preferred Stock:
Voting Rights. Until such time, if ever, as the shareholders of the Company have approved the issuance of shares of common stock upon the conversion of the Series B Convertible Preferred Stock (“Stockholder Approval”), the Series B Convertible Preferred Stock only has rights to vote on amendments to the Series B Designation (which are subject to the approval of a simple majority of the holders of Series B Convertible Preferred Stock), and the Protective Provisions, discussed below. Stockholder Approval was received at the Company’s annual meeting of stockholders held on December 27, 2024 (the “Stockholder Approval Date”).
F-22
The Series B Preferred Stock requires the consent of the holders of at least a majority of the issued and outstanding shares of Series B Convertible Preferred Stock to (a) increase or decrease (other than by redemption or conversion) the total number of authorized shares of Series B Convertible Preferred Stock of the Company; (b) adopt or authorize any new designation of any Preferred Stock or amend the Certificate of Incorporation of the Company in a manner which (i) provides any holder of common stock or preferred stock any rights upon a liquidation of the Company which are prior and superior to those of the holders of the Series B Convertible Preferred Stock; or (ii) adversely affect the rights, preferences and privileges of the Series B Convertible Preferred Stock; (c) effect an exchange, or create a right of exchange, cancel, or create a right to cancel, of all or any part of the shares of another class of shares into shares of Series B Convertible Preferred Stock; (d) alter or change the rights, preferences or privileges of the shares of Series B Convertible Preferred Stock so as to affect adversely the shares of such series; and (e) issue any shares of Series A Preferred Stock or Series B Convertible Preferred Stock, other than the Preferred Stock issued pursuant to the Purchase Agreement (collectively, the “Protective Provisions”).
After Stockholder Approval (which was received on December 27, 2024, as discussed above), in addition to the above voting rights, each holder of outstanding shares of Series B Convertible Preferred Stock shall be entitled to cast the number of votes in connection with the Series B Convertible Preferred Stock shares held by such holder equal to the number of whole shares of common stock into which the shares of Series B Convertible Preferred Stock held by such holder are convertible as of the record date for determining stockholders entitled to vote on such matter. Fractional votes shall not, however, be permitted and any fractional voting rights available on an as-converted to common stock basis (after aggregating all fractional shares into which shares of Series B Convertible Preferred Stock held by each holder could be converted) shall be rounded down to the nearest whole share. Except as provided by law or by the other provisions of the Certificate of Incorporation or the Series B Designation, holders of Series B Convertible Preferred Stock shall vote together with the holders of common stock as a single class and there shall be no series voting.
Dividend Rights. None, except that if the Company declares a dividend or makes a distribution of cash (or any other distribution treated as a dividend under Section 301 of the Internal Revenue Code) on its common stock, each holder of Shares of Series B Convertible Preferred Stock is entitled to participate in such dividend or distribution in an amount equal to the largest number of whole shares of common stock into which all shares of Series B Convertible Preferred Stock held of record by such holder are convertible as of the record date for such dividend or distribution or, if there is no specified record date, as of the date of such dividend or distribution. Notwithstanding the foregoing, holders shall have no right of participation in connection with dividends or distributions made to the common stock stockholders consisting solely of shares of common stock.
Liquidation Preference.
In the event of any liquidation, dissolution or winding up of the Company, either voluntary or involuntary (each a “Liquidation
Event”), the holders of Series B Convertible Preferred Stock are entitled to receive prior and in preference to any distribution
of any of the assets of the Company to the holders of the common stock or securities junior to the Series B Convertible Preferred Stock
(other than the common stock) by reason of their ownership of such stock, but after any required distribution to any holders of Series
B Convertible Preferred Stock, an amount in cash per share of Series B Convertible Preferred Stock for each share of Series B Convertible
Preferred Stock held by them equal to the greater of (x) one times the Stated Value; and (y) the total amount of consideration
that would have been payable on such share upon a Liquidation Event, had such share of Series B Convertible Preferred Stock been converted
into common stock, immediately prior to such Liquidation Event (as applicable, the “Liquidation Preference”). The “Stated
Value” is $
F-23
Conversion Rights.
None prior to Stockholder Approval. After Stockholder Approval, at the option of the holder(s) thereof, each share of Series B Convertible
Preferred Stock is convertible into a number of shares of common stock of the Company as equals the Conversion Rate. The “Conversion
Rate” initially was
Based on the terms of the
Series B Convertible Preferred Stock and the Purchase Agreement, and in accordance with FASB ASC 480-10, the Series B Convertible Preferred
Stock was accounted for as a liability at its issuance date due to the variable number of shares issuable under the agreement pursuant
to the conversion terms, with an initial fair value of $
NOTE 10 - COMMITMENTS AND CONTINGENCIES
Litigation and Other Loss Contingencies
The Company records liabilities for loss contingencies arising from claims, assessments, litigation, fines, penalties and other sources when it is probable that a liability has been incurred and the amount of the loss can be reasonably estimated. The Company has no liabilities recorded for loss contingencies as of December 31, 2024.
Legal Matters
Action Against Former Executive of KBL
On September 1, 2021, the
Company initiated legal action in the Chancery Court of Delaware against Dr. Marlene Krauss, the Company’s former Chief Executive
Officer and director (“Dr. Krauss”) and two of her affiliated companies, KBL IV Sponsor, LLC (“KBL Sponsor”)
and KBL Healthcare Management, Inc. (collectively, the “KBL Affiliates”) for, among other things, engaging in
unauthorized monetary transfers of the Company’s assets, non-disclosure of financial liabilities within the Company’s Consolidated
Financial Statements, issuing shares of stock without proper authorization; and improperly allowing stockholder redemptions to take place.
The Company’s complaint alleges multiple causes of action against Dr. Krauss and/or the KBL Affiliates, and seeks compensatory damages
in excess of $
On October 5, 2021, Dr. Krauss
and the KBL Affiliates filed an Answer, Counterclaims and Third-Party Complaint against the Company and twelve individuals who are, or
were, directors and/or officers of the Company, i.e., Marc Feldmann, Lawrence Steinman, James N. Woody, Teresa DeLuca,
Frank Knuettel II, Pamela Marrone, Lawrence Gold, Donald A. McGovern, Jr., Russell T. Ray, Richard W. Barker, Shoshana Shendelman and
Ozan Pamir (collectively, the “Third-Party Defendants”). On February 24, 2022, Dr. Krauss filed an amended
Answer, Counterclaims and Third-Party Complaint (the “Amended Counterclaims”), which, among other things, dismissed
Teresa DeLuca, Frank Knuettel II, Pamela Marrone, Russell T. Ray, Richard W. Barker and Shoshana Shendelman. In essence, the
Amended Counterclaims allege that the Company and the remaining Third-Party Defendants made alleged misstatements against Dr. Krauss in
Securities and Exchange Commission (SEC) filings, failed to register her shares in the Company so that they could be traded, and failed
to pay to Dr. Krauss the amounts alleged to be owing under a promissory note in the principal amount of $
F-24
On June 25, 2024, Dr. Krauss filed a Motion for partial summary judgment on her claim that the Company failed to register her shares.
Action Against the Company by Dr. Krauss
On August 19, 2021, Dr. Krauss initiated legal action in the Chancery Court of Delaware against the Company. The Complaint alleged that the Company is obligated to advance expenses including, attorney’s fees, to Dr. Krauss for the costs of defending against an SEC investigation and Subpoenas, and that the Company is also required to reimburse Dr. Krauss for the costs of bringing this lawsuit against the Company. On September 3, 2021, Dr. Krauss filed an Amended Complaint which further alleged that Dr. Krauss is also allegedly entitled to advancement by the Company of her expenses, including attorney’s fees, for the costs of defending against the Third-Party Complaint in the Tyche action referenced below, and the costs of defending against the Company’s own Complaint against Dr. Krauss as described above. On or about September 23, 2021, the Company filed its Answer to the Amended Complaint in which the Company denied each of Dr. Krauss’ claims and raised numerous affirmative defenses.
On November 15, 2021, Dr.
Krauss filed a Motion for Summary Adjudication as to certain of the issues in the case, which was opposed by the Company. On March 7,
2022, the Court issued a decision denying the Motion in part and granting it in part. The Court then issued an Order implementing
such decision on March 29, 2022. The parties subsequently engaged in proceedings as set forth in that Order, and the Company
was required to pay a portion of those fees while objecting to the remaining portion of disputed fees. On October 10, 2022 and January
18, 2023, Dr. Krauss filed applications to compel the Company to pay the full amount of fees requested by Dr. Krauss for May-October 2022,
and to modify the Court’s Order. On May 3, 2023, the Court issued an Order granting both of Dr. Krauss’s Applications for
payment of attorney’s fees totaling $
On February 21, 2025, the
Company entered into a Mutual Settlement and General Release Agreement with Dr. Krauss, KBL Merger Corp. IV, the Company’s
predecessor, and KBL Sponsor (the “Settlement Agreement”). Pursuant to the Settlement Agreement, the Company agreed
to (1) pay $
The Settlement Agreement also required Dr. Krauss to enter into a Voting Agreement with the Company. Pursuant to the Voting Agreement, which was entered into on February 21, 2025, by Dr. Krauss, the Company, and Blair Jordan, the Company’s Chief Executive Officer, solely for the benefit of the Company, Dr. Krauss agreed to vote the Settlement Shares as recommended by the Board of Directors of the Company, at any meeting of stockholders or via any written consent of stockholders, which may occur prior to August 21, 2025. In order to enforce the terms of the Voting Agreement, and solely for the benefit of the Company, Dr. Krauss provided Mr. Jordan (or his assigns) an irrevocable voting proxy to vote the Settlement Shares pursuant to the guidelines set forth above at any meeting of stockholders or via any written consent of stockholders. The Voting Agreement also provides a restriction on Dr. Krauss’s sale or transfer of any of the Settlement Shares until August 21, 2025, and provides for mutual general releases of the parties.
The Settlement Agreement required Dr. Krauss to file a dismissal of her court actions within ten days after receipt of the Cash Payment and that the Company, Dr. Krauss and KBL Sponsor have submitted filings with the court to dismiss all of their claims against one another with prejudice.
Action Against Tyche Capital LLC
The Company commenced and
filed an action against defendant Tyche Capital LLC (“Tyche”) in the Supreme Court of New York in the County of New
York on April 15, 2021. In its Complaint, the Company alleged claims against Tyche arising out of Tyche’s breach of its written
contractual obligations to the Company as set forth in a Guarantee and Commitment Agreement dated July 25, 2019, and a Term Sheet for
KBL Business Combination With CannBioRex dated April 10, 2019 (collectively, the “Subject Guarantee”), and claimed
that Tyche’s breach of the Subject Guarantee caused the Company damages in the amount of at least $
F-25
On or about May 17, 2021, Tyche responded to the Company’s Complaint by filing an Answer and Counterclaims against the Company alleging that it was the Company, rather than Tyche, that had breached the Subject Guarantee. Tyche also filed a Third-Party Complaint against six third-party defendants, including three members of the Company’s then management, Sir Marc Feldmann, Dr. James Woody, and Ozan Pamir (collectively, the “Individual Company Defendants”), claiming that they allegedly breached fiduciary duties to Tyche with regards to the Subject Guarantee.
On November 23, 2021, the Court granted the Company’s request to issue an Order of attachment against all of Tyche’s shares of the Company’s stock that had been held in escrow. In so doing, the Court found that the Company had demonstrated a likelihood of success on the merits of the case based on the facts alleged in the Company’s Complaint.
On February 18, 2022, Tyche filed an Amended Answer, Counterclaims and Third-Party Complaint. On August 25, 2022, the Court granted the Company’s Motion to Dismiss each of the Individual Company Defendants, and also three of the four Counterclaims brought against the Company, leaving only Tyche’s declaratory relief claim. On August 26, 2022, Tyche filed a Motion to vacate or modify the Company’s existing attachment Order against Tyche’s shares of the Company’s stock held in escrow, however, the Court summarily denied such Motion on January 3, 2023. Although Tyche subsequently filed a Notice of Appeal as to that denial, on May 4, 2023, the Appellate Court unanimously affirmed the ruling of the lower Court in the Company’s favor.
On January 30, 2023, the Company filed a Notice of Motion for Summary Judgment. In hearings held on September 11 and 19, 2023, the Court granted the Company’s Motion, but referred the question as to the amount of the Company’s damages against Tyche to a special referee. Tyche filed a Notice of Appeal as to the Court’s ruling.
On June 30, 2024, the Company
entered into a written Settlement Agreement with Tyche and Ronald Bauer & Samantha Bauer to fully resolve this action with Tyche and
the Bauer action referenced below. The Settlement Agreement has been fully signed, and the parties are currently engaged in performing
its terms, which include, among other things, transfer to the Company of Tyche’s shares of the Company’s stock in escrow,
transfer to the Company of shares of the Company’s stock held by Theseus Capital Ltd. and Astatine Capital Ltd., forgiveness of
loans to the Company by the Bauer Defendants, exoneration of the Company’s $
Action Against Ronald Bauer & Samantha Bauer
The Company and two of its wholly-owned subsidiaries, Katexco Pharmaceuticals Corp. and CannBioRex Pharmaceuticals Corp. (collectively, the “Company Plaintiffs”), initiated legal action against Ronald Bauer and Samantha Bauer, as well as two of their companies, Theseus Capital Ltd. and Astatine Capital Ltd. (collectively, the “Bauer Defendants”), in the Supreme Court of British Columbia on February 25, 2022. The Complaint claims that the Bauer Defendants misappropriated funds and stock shares, engaged in unauthorized stock sales, and obtained improper travel expenses. The Bauer Defendants filed a Response denying the Civil Claim Complaint of the Company on May 6, 2022.
On June 30, 2024, the Company Plaintiffs, Tyche and the Bauer Defendants entered into the Settlement Agreement described above, which fully resolves this action.
F-26
Declaratory Relief Action Against the Company by AmTrust International
On June 29, 2022, AmTrust International Underwriters DAC (“AmTrust”), which was the premerger directors’ and officers’ insurance policy underwriter for KBL, filed a declaratory relief action against the Company in the U.S. District Court for the Northern District of California (the “Declaratory Relief Action”) seeking a declaration that AmTrust is not obligated to reimburse the Company for fees advanced by the Company to Dr. Krauss and George Hornig, former officer and directors of the Company, under the directors’ and officers’ insurance policy.
On September 20, 2022, the
Company filed its Answer and Counterclaims against AmTrust for bad faith breach of AmTrust’s insurance coverage obligations to the
Company under the subject insurance policy, seeking at least $
On November 22, 2022, the Company filed a Motion for Summary Adjudication against both AmTrust and Freedom. The Motion was fully briefed and a hearing was held on March 9, 2023. The standard to prevail on a Motion for Summary Adjudication in the Court is high to prevail and requires a judge to find that there are no disputed issues of fact so that they can rule on the issues as a matter of law. In this instance the judge found three major issues could be decided as a matter of law in the Company’s favor and that one issue, the Change in Control exclusion, requires further discovery.
On April 21, 2023, the Court issued an Order Granting in Part and Denying in Part the Company’s Motion for Partial Summary Judgment.
Specifically, the Court granted summary adjudication in favor of the Company on the following issues: (a) that the Company is, in fact, an insured under both the AmTrust and Freedom insurance policies; (b) that certain SEC subpoena related expenses for defendants Dr. Marlene Krauss, the Company’s former Chief Executive Officer and Director, and George Hornig, the former Chairman of the Board, are within the basic scope of coverage under both the AmTrust and Freedom insurance policies; and (c) that the Insured vs. Insured exclusion relied upon by AmTrust and Freedom is not applicable to bar any such coverage.
The Court also found that there were issues of disputed facts as to the Change in Control exclusion contained within the policies, which therefore precluded the Court from granting the remainder of the Company’s requests for summary adjudication as a matter of law. Accordingly, the Court, at that time, denied the Company’s further requests for summary adjudication and deemed that for the time being, the Change in Control issue is to be determined at the time of trial, in order to find that the policies (i) provide coverage for the fees which the Company has advanced and will advance to Dr. Marlene Krauss and George Hornig; (ii) that AmTrust has breached the policy; (iii) that AmTrust must pay such expenses of the Company; and that, once the AmTrust policy has been exhausted, (iv) Freedom will be obligated to pay such expenses of the Company pursuant to its policy.
On August 4, 2023, the Court granted the Company’s request to file a second motion for partial summary judgment in the case, this one being on the issue of whether AmTrust should be required to advance to the Company the defense costs being incurred by Dr. Marlene Krauss and George Hornig during the pendency of the case. The Motion for Partial Summary Judgment was fully briefed by the parties, and a hearing for such Motion was held on January 11, 2024. After the matter was taken under submission, on February 12, 2024, the Court granted the Company’s Motion for Partial Summary Judgment against both AmTrust and Freedom, and ordered as follows: (a) AmTrust is obligated under its insurance policy with the Company to advance to the Company all defense costs in excess of the deductible that the Company has advanced, or will advance, to Dr. Krauss and Mr. Hornig in connection with certain SEC Subpoenas, and (b) upon exhaustion of the AmTrust insurance policy, Freedom is obligated to do the same pursuant to its excess liability insurance policy with the Company. This Order applies throughout the interim of the case, but does not constitute a final judgment, and both the Company and the two insurers retain their rights to contest all applicable issues at trial.
F-27
On April 16, 2024, AmTrust
paid the Company $
The Company, Freedom and Amtrust
held a mediation conference on August 21, 2024, during which, the Company agreed to the terms of a settlement with Freedom, pursuant to
which Freedom agreed to pay $
A trial on these matters was
scheduled for May 12, 2025. The Company and AmTrust conducted mediation at the end of February 2025 and early March 2025. On March 10,
2025, the court vacated all scheduled dates, and terminated AmTrust’s motion for summary judgment without prejudice. The court also
ordered that the parties file a stipulated dismissal or a status report regarding settlement within 60 days, by May 9, 2025. The Company
and AmTrust are currently negotiating settlement terms, of which no assurance can be provided that a final agreement can be reached. A
final judgment following trial could potentially confirm these obligations of the insurers or, alternatively, reverse and require the
Company to repay all or portions of such advance payments, including up to and including the $
Stanford License Agreement
During the years ended December
31, 2024 and 2023, the Company recorded patent and license fees of $
Oxford University Agreements
On September 18, 2020, CBR
Pharma entered into a 3 year research and development agreement (the “3 Year Oxford Agreement”) with Oxford to research and
investigate the mechanisms underlying fibrosis in exchange for aggregate consideration of $
F-28
On May 24, 2021, the Company
entered into a research agreement with the University of Oxford (“Oxford” and the “Fifth Oxford Agreement”), pursuant
to which the Company will sponsor work at the University of Oxford to conduct a multi-center, randomized, double blind, parallel group,
feasibility study of anti-TNF injection for the treatment of adults with frozen shoulder during the pain-predominant phase.
| Amount Due | ||||
| Milestone | (excluding VAT) | |||
| £ | ||||
| £ | ||||
| £ | ||||
| £ | ||||
The Company paid the first
milestone of $
On November 2, 2021, the Company
and Oxford University entered into a twenty-year licensed technology agreement of the HMGB1 molecule, which is related to tissue regeneration,
whereby Oxford University agreed to license the technology to the Company for research, development and use of the licensed patents. The
Company agreed to pay Oxford University for past patent costs $
Due
to the ongoing costs of the HMGB1 research program and the need for the Company to focus its resources on the Company’s primary
platform to treat fibrosis using anti-TNF (tumor necrosis factor), the Board of Directors of the Company elected to terminate the Company’s
HMGB1 license agreement with Oxford on September 22, 2023, and on September 22, 2023, the Company and Oxford entered into a termination
letter, formally terminating the License effective September 22, 2023. The termination letter also clarified amounts that we owed after
termination of the License, including approximately $
Due to recent financial constraints,
the Company has been unable to timely pay amounts due to Oxford, the licensor of the majority of the Company’s licenses and patents
and the Company’s research partner. Oxford alleges that an aggregate of approximately £
F-29
Kennedy License Agreement
On September 27, 2019, 180 LP entered into a license agreement (the “Kennedy License Agreement”) with the Kennedy Trust for Rheumatology Research (“Kennedy”) exclusively in the U.S., Japan, United Kingdom and countries of the EU, for certain licensed patents (the “Kennedy Licensed Patents”), including the right to grant sublicenses, and the right to research, develop, sell or manufacture any pharmaceutical product (i) whose research, development, manufacture, use, importation or sale would infringe the Kennedy Licensed Patents absent the license granted under the Kennedy License Agreement or (ii) containing an antibody that is a fragment of or derived from an antibody whose research, development, manufacture, use, importation or sale would infringe the Kennedy Licensed Patents absent the license granted under the Kennedy License Agreement, for all human uses, including the diagnosis, prophylaxis and treatment of diseases and conditions.
As a consideration for the
grant of the Kennedy Licensed Patents, 180 LP paid Kennedy an upfront fee of GBP £
The term of the royalties paid by the Company to Kennedy will expire on the later of (i) the last valid claim of a patent included in the Kennedy Licensed Patents which covers or claims the exploitation of a product in the applicable country; (ii) the expiration of regulatory exclusivity for the product in the country; or (iii) 10 years from the first commercial sale of the product in the country. The Kennedy License Agreement may be terminated without cause by providing a 90-day notice.
Petcanna Sub-License Agreement
On August 20, 2018, CBR Pharma entered into a sub-license agreement (the “Sub-License Agreement”) with its wholly owned subsidiary, Petcanna Pharma Corp. (“Petcanna”), of which the Company’s former Chief Financial Officer is a director. Petcanna is a private company with one common principal with the Company.
Pursuant to the terms of the
Sub-license Agreement, the Company has granted a sub-license on the Licensed Patents to pursue development and commercialization for the
treatment of all veterinary conditions. In consideration, Petcanna will (a) issue
Operating Leases
In February 2016, the FASB issued an update to the Accounting Standards Codification Topic 842, Leases (“ASC 842”). The new standard required most leases to be recognized on the balance sheet as a right-of-use (“ROU”) asset and a lease liability. The right-of-use asset is initially measured at the present value of amounts expected to be paid over the lease term. Recognition of the costs of these leases on the income statement are disaggregated and recognized as both operating expense (for the amortization of the right-of-use asset) and interest expense (for the portion of the lease payment related to interest). This standard was adopted by the Company upon issuance.
F-30
In accordance with ASC 842, the Company can elect (by asset class) not to record on the balance sheet a lease whose term is 12 months or less and does not include a purchase option that the lessee is reasonably certain to exercise. If elected, the lease would be treated like an operating lease under previous GAAP; payments would be recognized on a straight-line basis over the lease term. When determining whether the lease qualifies for this election, the Company would include renewal options only if they are considered part of the lease term, i.e., those options the Company is reasonably certain to exercise. If the lease term increases to more than 12 months, or if it is reasonably certain the Company will exercise a purchase option, the Company would no longer be able to apply this practical expedient and would apply ASC 842 guidance.
Regarding leases (of which it currently has none), the Company would use the practical expedient for short-term operating leases that are 12 months or less. This practical expedient has been elected as a package and it would be applied consistently to all leases. Additionally, if the Company’s leases are considered operating in nature and therefore not reflected on the balance sheet, the Company will recognize the short-term lease payments as rent/lease expense monthly on the income statement.
As of December 31, 2024 and 2023, the Company had no leases as well as no lease or rent expense as of those dates.
Consulting Agreements
Nanchahal Consulting Agreement
On February 22, 2021, the Company entered into a consultancy agreement (as amended, the “Consulting Agreement”) with a related party, Prof. Jagdeep Nanchahal (the “Consultant”). The Consulting Agreement was effective December 1, 2020.
Pursuant to the Consulting
Agreement, the Company agreed to pay the Consultant
| ● | the sum of £ |
| ● | the sum of £ |
| ● | the sum of £ |
| ● | the sum of £ |
The Consulting Agreement had an initial term of three years, and renews thereafter for additional three-year terms, until terminated as provided in the agreement. The Consulting Agreement can be terminated by either party with 12 months prior written notice (provided the Company’s right to terminate the agreement may only be exercised if the Consultant fails to perform his required duties under the Consulting Agreement), or by the Company immediately under certain conditions specified in the Consulting Agreement if (a) the Consultant fails or neglects efficiently and diligently to perform the services required thereunder or is guilty of any breach of its or his obligations under the agreement (including any consent granted under it); (b) the Consultant is guilty of any fraud or dishonesty or acts in a manner (whether in the performance of the services or otherwise) which, in the reasonable opinion of the Company, has brought or is likely to bring the Consultant, the Company or any of its affiliates into disrepute or is convicted of an arrestable offence (other than a road traffic offence for which a non-custodial penalty is imposed); or (c) the Consultant becomes bankrupt or makes any arrangement or composition with his creditors. If the Consulting Agreement is terminated by the Company for any reason other than cause, the Consultant is entitled to a lump sum payment of 12 months of his fee as of the date of termination.
F-31
Effective March 30, 2021,
in satisfaction of amounts owed to the Consultant for
Effective August 27, 2021,
in satisfaction of amounts owed to the Consultant for the remainder of Bonus 2, the Company issued
In December 2021, the Dupuytren’s Contracture clinical trial data was submitted for publication in a peer-reviewed journal and Bonus 1 was paid to the Consultant.
On April 27, 2022, the Company
entered into an Amendment to the Consulting Agreement, whereby upon acceptance of the data for the Phase 2b clinical trial for Dupuytren’s
Contracture for publication, the Consultant’s monthly fee will increase to £
On December 28, 2022, the
Company entered into an Amendment to the Consulting Agreement, whereby the Company agreed that the Consultant’s monthly fee increased
to £
Other Commitments
Amendments to Compensation Agreements
On January 10, 2024, and effective on January 1, 2024, the Company entered into (a) a Fourth Amendment to Amended and Restated Employment Agreement with Dr. James Woody, the then CEO and Director of the Company; (b) a Fourth Amendment to Employment Agreement with Dr. Jonathan Rothbard, the then Chief Science Officer (CSO) of the Company; (c) a Third Amendment to Consulting Agreement with Dr. Lawrence Steinman, the then Executive Chairman of the Company; and (d) a Second Amendment to Consulting Agreement with Prof. Sir Marc Feldmann, the former Executive Co-Chairman of the Company (collectively, the “Amendments”), which each amended the compensation agreements then in place with such individuals.
Pursuant to the
Amendments, each of Dr. Woody and Dr. Rothbard, effective as of January 1, 2024, agreed to a reduction of the base salaries set
forth in their respective amended employment agreements, by
F-32
Also pursuant to the Amendments,
each of Dr. Steinman and Sir Marc Feldmann, effective as of January 1, 2024, agreed to a reduction of the base salaries set forth in their
respective consulting agreements, by
Woody Separation Agreement
On May 7, 2024, Dr. James N. Woody resigned as Chief Executive Officer (Principal Executive Officer), and as a member of the Board of Directors, of the Company effective the same date, and entered into a Separation and Release Agreement with the Company (the “Woody Separation Agreement”).
Under the Woody Separation
Agreement, the Company (a) paid Dr. Woody $
Under the Woody Separation
Agreement, Dr. Woody agreed to provide a customary general release to the Company, waived any severance pay that would have been due pursuant
to the terms of his employment agreement, agreed to the termination of his employment agreement, and also agreed to certain confidentiality,
non-disclosure, non-solicitation, non-disparagement, and cooperation covenants in favor of the Company. The
On
February 5, 2025, the Company entered into a First Amendment to Separation and Release Agreement with Dr. Woody (the “First
Amendment”). Pursuant to the First Amendment, Dr. Woody agreed to amend the terms of the Woody Separation Agreement, to terminate
the prior requirement of the Company set forth therein to pay Dr. Woody the Future Contingent Payment, [A] if the Company, within
the 24 months following the date of Dr. Woody’s resignation, complete any corporate transaction, including but not limited to any
merger, reverse merger, acquisition, disposal, joint-venture and/or investment involving the Company, which results in a change of control;
or [B] if the Company raises at least $
F-33
The First Amendment also required Dr. Woody to enter into a Voting Agreement with the Company. Pursuant to the Voting Agreement, which was entered into on February 5, 2025, by Dr. Woody, the Company, and Blair Jordan, the Company’s Chief Executive Officer, solely for the benefit of the Company, Dr. Woody agreed to vote the Separation Shares as recommended by the Board of Directors of the Company, at any meeting of stockholders or via any written consent of stockholders, which may occur prior to February 5, 2026; the date after August 5, 2025, that Dr. Woody has sold all of the Separation Shares; or the date that the Company terminates the Voting Agreement. In order to enforce the terms of the Voting Agreement, and solely for the benefit of the Company, Dr. Woody provided Mr. Jordan (or his assigns) an irrevocable voting proxy to vote the Separation Shares pursuant to the guidelines set forth above at any meeting of stockholders or via any written consent of stockholders. The Voting Agreement also provides a restriction on Dr. Woody’s sale or transfer of any of the Separation Shares until August 5, 2025.
Feldmann Separation Agreement
Effective on September 5, 2024, the Company’s wholly-owned subsidiary, CBR Pharma and the Company, entered into a Separation and Release Agreement with Sir Marc Feldmann, the former Co-Executive Chairman of the Company, the former Chief Executive Officer of CBR Pharma, and the former director of CBR Pharma, which was amended to clarify such agreement was effective September 5, 2024 (as amended, the “Feldmann Separation Agreement”).
Under the Feldmann Separation
Agreement, the Company agreed to issue Sir Marc Feldmann
Also effective on September 5, 2024, the Company entered into an Indemnification Agreement with Sir Marc Feldmann to provide for indemnification to Sir Marc Feldmann under Delaware law. Among other things, consistent with the Company’s Bylaws, the Indemnification Agreement generally requires that the Company (i) indemnify Sir Marc Feldmann from and against all expenses and liabilities with respect to proceedings to which Sir Marc Feldmann may be subject by reason of Sir Marc Feldmann’s service to the Company and its subsidiaries to the fullest extent authorized or permitted by Delaware law and (ii) advance all expenses incurred by Sir Marc Feldmann in connection with the investigation, defense, settlement or appeal of any proceeding, and in connection with any proceeding to enforce Sir Marc Feldmann’s rights under the Indemnification Agreement.
Rothbard Separation Agreement and Consulting Agreement
On May 7, 2024, Dr. Jonathan Rothbard resigned as Chief Scientific Officer of the Company effective the same date and entered into a Separation and Release Agreement with the Company (the “Rothbard Separation Agreement”).
F-34
Under the Rothbard Separation
Agreement, the Company agreed to pay Dr. Rothbard $
Effective on May 7, 2024,
the Company entered into a Consulting Agreement with Dr. Rothbard pursuant to which he agreed to provide general consulting services to
the Company for a term of six months, for $
Steinman Fourth Amendment to Consulting Agreement
On May 7, 2024, the Company
entered into a Fourth Amendment to Consulting Agreement with Dr. Lawrence Steinman, the then Executive Chairman of the Board and current
director of the Company (the “Fourth Amendment”). Pursuant to the Fourth Amendment, Dr. Steinman waived and forgave
all amounts accrued and owed to him under the Consulting Agreement through such date, and agreed that compensation payable to him under
the Consulting Agreement moving forward would be $
Dr. Steinman also agreed to step down as Executive Chairman of the Board of Directors and to instead serve as a regular member of the Board of Directors, provided that on May 7, 2024, Dr. Steinman was appointed as a member of the then Strategy and Alternatives Committee of the Company.
Appointment of Interim Chief Executive Officer and Consulting Agreement
Also effective on May 7, 2024, the Board of Directors appointed Mr. Blair Jordan, a then member of the Board of Directors of the Company, as Interim Chief Executive Officer and Principal Executive Officer of the Company, to fill the vacancy left by Dr. Woody’s resignation. Upon such appointment, Mr. Jordan ceased being the Lead Independent director of the Company, and ceased being a member of the Audit Committee, Compensation Committee, and Nominating and Corporate Governance Committee of the Board of Directors.
On May 7, 2024, the Company
entered into an Executive Consulting Agreement with Mr. Jordan and Blair Jordan Strategy and Finance Consulting Inc. (an entity owned
by Mr. Jordan) (“Jordan Consulting” and the “Jordan Consulting Agreement”). Pursuant to the Jordan
Consulting Agreement, the Company agreed to engage Jordan Consulting to provide the services of Mr. Jordan to the Company as Interim Chief
Executive Officer of the Company. The Jordan Consulting Agreement has a term through April 30, 2025, unless otherwise terminated pursuant
to the terms of the agreement (discussed below) and provides for Mr. Jordan to act as Interim Chief Executive Officer of the Company,
and to be paid $
F-35
The Jordan Consulting Agreement
terminates automatically upon the completion of a Corporate Transaction (provided we pay the transaction bonus discussed above). We have
the right to terminate the Jordan Consulting Agreement at any time, provided that if we terminate the agreement after 60 days and prior
to completion of a Corporate Transaction, then we agreed to pay Jordan Consulting $
Non-Executive Director Compensation
Effective March 7, 2024, the Board of Directors of the Company appointed Omar Jimenez and Ryan L. Smith as members of the Board of Directors (“Board”), which Appointments were effective as of the same date.
Effective on May 7, 2024,
the Board of Directors set the compensation payable to non-executive members of the Board of Directors for services on the Board of Directors,
at (a) $
Effective October 24, 2024, the Board of Directors of the Company appointed Jay Goodman as a member of the Board of Directors (“Board”), which appointment was effective as of the same date.
In connection with Mr. Goodman’s
appointment to the Board, the Company entered into an offer letter with Mr. Goodman (the “Goodman Offer Letter”). The
Goodman Offer Letter provides for Mr. Goodman to be paid $
Effective on October 24, 2024, Mr. Blair Jordan’s Offer Letter entered into with Mr. Jordan in connection with his appointment to the Board in February 2024, was mutually terminated due to the fact that he is no longer serving as an independent member of the Board and because he is currently serving as Interim Chief Executive Officer of the Company, party to a Consulting Agreement with the Company providing for compensation payable to him as an officer of the Company.
On December 2, 2024, Mr. Omar Jimenez resigned effective as of December 3, 2024 from the Board. Effective as of the date of his resignation from the Board, the March 4, 2024 Offer Letter between Mr. Jimenez and the Company was terminated. Mr. Jimenez continued to serve as the Chief Financial Officer and Secretary of the Company until December 16, 2024 when he tendered his resignation.
On December 3, 2024, immediately upon Mr. Jimenez’s resignation, the Board of the Company appointed Stephen H. Shoemaker as a member of the Board. In connection with Mr. Shoemaker’s appointment to the Board, the Company entered into an offer letter with Mr. Shoemaker (the “Shoemaker Offer Letter”). The Shoemaker Offer Letter provides for Mr. Shoemaker to be paid $50,000 per year as an annual retainer fee for serving on the Board.
On February 4, 2025, the Board
of the Company appointed independent director Ryan Smith, as Lead Independent Director of the Company, and agreed to pay Mr. Smith an
additional $
F-36
Indemnification Agreements
The Company has entered into Indemnity Agreements (each an “Indemnification Agreement”) with each of its then directors and officers (each an “Indemnitee”), to provide for indemnification to the officers and directors under Delaware law. Among other things, consistent with the Company’s Bylaws, each Indemnification Agreement generally requires that the Company (i) indemnify the Indemnitee from and against all expenses and liabilities with respect to proceedings to which Indemnitee may be subject by reason of the Indemnitee’s service to the Company to the fullest extent authorized or permitted by Delaware law and (ii) advance all expenses incurred by the Indemnitee in connection with the investigation, defense, settlement or appeal of any proceeding, and in connection with any proceeding to enforce the Indemnitee’s rights under the Indemnification Agreement. The Indemnification Agreement also establishes various related procedures and processes and generally requires the Company to maintain directors’ and officers’ liability insurance coverage.
NOTE 11 - STOCKHOLDERS’ (DEFICIT) EQUITY
Reverse Stock Split during 2024
On February 16, 2024, at a
Special Meeting of the Stockholders of the Company, the stockholders of the Company approved an amendment to the Company’s Second
Amended and Restated Certificate of Incorporation, as amended, to effect a reverse stock split of our issued and outstanding shares of
our common stock, par value $
In addition, the number of shares of common stock issuable upon exercise of stock options and other equity awards (including shares reserved for issuance under the Company’s equity compensation plans) were proportionately adjusted by the applicable administrator, using the 1-for-19 ratio, and rounded down to the nearest whole share. The conversion rates of the Company’s preferred stock (of which none are outstanding) were also adjusted in a ratio of 1-for-19. The number of shares issuable upon exercise of the Company’s outstanding warrants to purchase shares of common stock outstanding were also equitably adjusted pursuant to the terms of such securities in connection with the 1-for-19 Reverse Stock Split. In addition, the exercise price for each outstanding stock option and warrant were increased in inverse proportion to the 1-for-19 split ratio such that upon an exercise, the aggregate exercise price payable by the optionee or warrant holder to the Company for the shares subject to the option or warrant will remain approximately the same as the aggregate exercise price prior to the Reverse Stock Split, subject to the terms of such securities.
Further,
pursuant to the terms of the Company’s Class K Special Voting Shares (the “Voting Stock”), following the Effective
Time, the Voting Stock was convertible into a number of shares of common stock equal to, and votes a number of voting shares equal to,
Each stockholder’s percentage ownership interest in the Company and proportional voting power remains virtually unchanged as a result of the Reverse Stock Split, except for minor changes and adjustments that will result from rounding fractional shares into whole shares. The rights and privileges of the holders of shares of common stock will be substantially unaffected by the Reverse Stock Split.
F-37
Preferred Stock
Pursuant to the Company’s
Second Amended and Restated Certificate of Incorporation filed on November 6, 2020, the Company has
The Special Voting Shares
have a par value of $
| ● | the right to vote in all circumstances in which our common stock have the right to vote, with the common stock as one class; |
| ● | the Special Voting Shares entitle the holder Odyssey Trust Company (the Trustee) to an aggregate number of votes equal to the number of shares of common stock that were issuable to the holders of the previously outstanding shares of CannBioRex Purchaseco ULC and/or Katexco Purchaseco ULC, Canadian subsidiaries of 180 (the “Exchangeable Shares”); |
| ● | the holder of the Special Voting Shares (and, indirectly, the holders of the Exchangeable Shares) has the same rights as the holders of the common stock as to notices, reports, financial statements and attendance at all stockholder meetings; |
| ● | no entitlement to dividends; |
| ● | the holder of the Special Voting Shares is not entitled to any portion of any related distribution upon windup, dissolution or liquidation of the Company; and |
| ● | the Company may cancel the Special Voting Shares when there are no Exchangeable Shares outstanding and no option or other commitment of CannBioRex Purchaseco ULC and Katexco Purchaseco ULC which could require either CannBioRex Purchaseco ULC and Katexco Purchaseco ULC to issue more Exchangeable Shares. |
As set forth above, the holders of the Exchangeable Shares, through the applicable Special Voting Share, have voting rights and other attributes corresponding to the common stock. The Exchangeable Shares provide an opportunity for certain former Canadian resident holders of CBR Pharma or Katexco securities to obtain a deferral of taxable capital gains for Canadian income tax purposes in connection with the Reorganization.
The Series B Preferred Stock is discussed above under Note 11 Stockholders’ (Deficit) Equity.
Common Stock
The Company is authorized
to issue
F-38
2024
December 2024 Offering
On December 27, 2024, the
Company entered into a Securities Purchase Agreement with certain accredited investors (the “Investors”). Pursuant to the
terms of the Purchase Agreement, the Company agreed to sell, in a registered direct offering, an aggregate of
Shares issued to former Board Members
On April 23, 2024, the Company
issued
Shares issued for Services
On July 19, 2024, the Company
issued a total of
Amendment to August 2023 Offering
As discussed below, on August
9, 2023, the Company entered into a Securities Purchase Agreement with an accredited investor (the “Purchaser”), in
addition to certain purchasers who relied on the Company’s registration statement filed with the SEC on July 25, 2023, which became
effective on August 9, 2023, pursuant to which the Company agreed to sell an aggregate of
On November 28, 2023, the
Company entered into an amendment to the August 2023 Offering (“Amendment to the August 2023 Offering”), whereby (i)
the Purchaser agreed to pay an additional $
Other than exercise price
and exercisability, the Additional Warrants have the same terms and conditions as the August 2023 Pre-funded Warrants and the August 2023
Common Warrants and, as such, were determined to be equity-classified because they met the limited exception in the case of a change-in-control.
In accordance with the Amendment to the August 2023 Offering, the Company entered into a warrant amendment agreement to amend the following
outstanding warrants held by the Purchaser: (i) warrants to purchase up to
F-39
During February and March
2024, all of the additional
Second Amendment to the 2022 Omnibus Incentive Plan
On February 16, 2024, the
Company held a special meeting of its stockholders as of December 18, 2023, and approved the adoption of a Second Amendment to the 180
Life Sciences Corp. 2022 Omnibus Incentive Plan. Such amendment increased the maximum number of shares available to be issued under the
Plan from
Third Amendment to the 2022 Omnibus Incentive Plan
On December 27, 2024, the
Company held its 2024 annual meeting of stockholders, and approved the adoption of a Third Amendment to the 180 Life Sciences Corp. 2022
Omnibus Incentive Plan. Such amendment increased the maximum number of shares available to be issued under the Plan from
2023
April 2023 Offering
On April 5, 2023, the Company
entered into a Securities Purchase Agreement with certain purchasers, pursuant to which the Company agreed to sell an aggregate of
The April 2023 Pre-Funded
Warrants had an exercise price equal to $
F-40
The April 2023 Common Warrants
have an exercise price equal to $
During 2023, the Company entered
into a series of amendments to these warrants in August and November; such amendments changed the exercise price from $
As of December 31, 2023, all
Amendment to July and December 2022 Common Warrants
On April 5, 2023, the Company
entered into an amendment to the common warrant agreements for the July 2022 and December 2022 Offerings, whereby warrants to purchase
up to
To assess for the change in
relative fair value, the Company performed a Black Scholes Option Model calculation to quantify the fair value of the common warrants
under their original terms as of the modification date using the following assumptions: a share price of $
First Amendment to the 2022 Omnibus Incentive Plan
At
the 2023 Annual Meeting of Stockholders of the Company held on July 6, 2023, the stockholders of the Company approved the First Amendment
(“First Amendment”) to the 180 Life Sciences Corp. 2022 Omnibus Incentive Plan. The First Amendment was originally approved
by the Board of Directors of the Company on May 5, 2023, subject to stockholder approval and the First Amendment became effective at the
time of stockholder approval. The First Amendment increased the maximum number of shares available to be issued under the 2022 Omnibus
Incentive Plan from
F-41
August 2023 Offering
On August 9, 2023, the Company
entered into a Securities Purchase Agreement with an accredited investor, in addition to certain purchasers who relied on the Company’s
registration statement filed with the SEC on July 25, 2023, which became effective on August 9, 2023, pursuant to which the Company agreed
to sell an aggregate of
The August 2023 Pre-Funded
Warrants have an exercise price equal to $
The August 2023 Common Warrants
have an exercise price equal to $
During 2023, the Company entered
into a series of amendments to these warrants in August and November; such amendments changed the exercise price from $
As of December 31, 2023, all
of the August 2023 Pre-Funded Warrants have been exercised for a value of $
F-42
Second Amendment to Common Warrant Agreements for the July 2022, December 2022 and April 2023 Offerings
On August 9, 2023, the Company
entered into an amendment to the common warrant agreements for the July 2022, December 2022 and April 2023 Offerings, whereby common warrants
to purchase up to
To assess for the change in
relative fair value, the Company performed a Black Scholes Option Model calculation to quantify the fair value of the common warrants
under their original terms as of the modification date using the following assumptions for the July 2022, December 2022 and April 2023
common warrants: a share price of $
Amendment to August 2023 Offering
On November 28, 2023, the
Company entered into an amendment to the August 2023 Offering (“Amendment to the August 2023 Offering”), whereby (i) the Purchaser
agreed to pay an additional $
Other than exercise price
and exercisability, the Additional Warrants have the same terms and conditions as the August 2023 Pre-funded Warrants and the August 2023
Common Warrants and, as such, were determined to be equity-classified because they met the limited exception in the case of a change-in-control.
Because the Additional Warrants are equity-classified, the placement agent fees and offering expenses will be accounted for as a reduction
of additional paid in capital. The Additional Warrants will not be exercisable until the Company obtains stockholder approval with respect
to the issuance of the
In accordance with the Amendment
to the August 2023 Offering, the Company entered into a warrant amendment agreement to amend the following outstanding warrants held by
the Purchaser: (i) warrants to purchase up to
F-43
The Company accounted for the Amendment to the August 2023 Offering as a warrant modification, whereby the effect of the modification is measured as the difference in its relative fair value immediately before the modification and after the modification; and any increase to the relative fair value is recognized as an equity issuance cost.
To assess for the change in
relative fair value, the Company performed a Black Scholes Option Model calculation to quantify the fair value of the common warrants
under their original terms as of the modification date using the following assumptions for the July 2022, December 2022, April 2023 and
August 2023 common warrants: a share price of $
As of the filing of this Report,
all of the additional
Common Stock Issued for Services during 2023
During the year ended December
31, 2023, the Company issued an aggregate of
Restricted Stock Shares Issued during 2023
The Company recognized stock-based
compensation expense related to the amortization of the Restricted Stock Shares of $
Below is a table summarizing the Restricted Stock Shares granted and outstanding as of and for the year ended December 31, 2023:
| Unvested Restricted | Weighted Average Grant Date | |||||||
| Stock | FV Price | |||||||
| Unvested as of January 1, 2023 | $ | |||||||
| Granted | ||||||||
| Vested | ( | ) | ||||||
| Forfeited | ( | ) | ||||||
| Unvested as of December 31, 2023 | ||||||||
Special Voting Shares
The Special Voting Shares were issued to the former shareholders of CBR Pharma and Katexco in connection with the reorganization of 180 prior to the Business Combination. The Special Voting Shares are exchangeable by the holder for shares of the Company’s common stock and vote together as a single class with the Company’s common stockholders. Special Voting Shares are not entitled to receive any dividend of distributions.
F-44
During the years ended December
31, 2024 and 2023, shares were issued upon the exchange of common stock equivalents associated with the Special Voting Shares, and
the balance was
Stock Options
A summary of the option activity during the years ended December 31, 2024 and 2023 is presented below:
| Number of Options | Weighted Average Exercise Price | Weighted Average Remaining Term (in Years) | Intrinsic Value | |||||||||||||
| Outstanding, January 1, 2023 | $ | |||||||||||||||
| Granted | ||||||||||||||||
| Forfeited | ( | ) | ||||||||||||||
| Outstanding, December 31, 2023 | ||||||||||||||||
| Granted | ||||||||||||||||
| Forfeited | ( | ) | ||||||||||||||
| Outstanding, December 31, 2024 | $ | $ | ||||||||||||||
| Exercisable, December 31, 2024 | $ | $ | ||||||||||||||
A summary of outstanding and exercisable stock options as of December 31, 2024 is presented below:
| Stock Options Outstanding | Stock Options Exercisable | |||||||||||||
| Weighted | ||||||||||||||
| Average | ||||||||||||||
| Exercise | Number of | Remaining | Number of | |||||||||||
| Price | Shares | Life in Years | Shares | |||||||||||
| $ | ||||||||||||||
| $ | ||||||||||||||
| $ | ||||||||||||||
| $ | ||||||||||||||
The Company recognized stock-based
compensation expense of $
F-45
The assumptions listed below for 2024 and 2023 were derived using i) the risk free interest rate published by the federal reserve on the date of grant, ii) the expected term used is the average of the contractual term plus the weighted average vesting term, iii) the volatility was derived using rates from third-party valuation reports of other financial instruments for the applicable quarter and iv) the expected dividends rate used is taken from the applicable option award agreement.
The assumptions used in the Black-Scholes valuation method for these options which were issued in 2024 were as follows:
| Risk free interest rate | % | |||
| Expected term (years) | ||||
| Expected volatility | % | |||
| Expected dividends | % |
On September 4, 2023, the
Company granted ten-year options for the purchase of an aggregate of
On September 4, 2023, the
Company also granted ten-year options for the purchase of an aggregate of
On September 4, 2023, the
Company also granted ten-year options for the purchase
The assumptions used in the Black-Scholes valuation method for these options which were issued in 2023 were as follows:
| Risk free interest rate | % | |||
| Expected term (years) | ||||
| Expected volatility | % | |||
| Expected dividends | % |
Warrants
On October 16, 2024, the Company
entered into a warrant inducement agreement (the “Inducement Agreement”) with a holder (the “Holder”)
of certain of the Company’s existing warrants to purchase shares of common stock (“Existing Warrants”). Pursuant
to the Inducement Agreement, the Holder agreed to exercise for cash Existing Warrants to purchase up to
On October 17, 2024, the Company
issued the Holder new warrants to purchase up to
F-46
The Company agreed in the Inducement Agreement to file a registration statement on Form S-1 to register the resale of the Inducement Warrant Shares upon exercise of the Inducement Warrants (the “Resale Registration Statement”) by November 15, 2024, and to use commercially reasonable efforts to have such Resale Registration Statement declared effective by the SEC within sixty (60) calendar days following the date of filing with the SEC (or ninety (90) calendar days following the date of filing with the Commission in the event of a ‘full review’ by the SEC ) and to keep the Resale Registration Statement effective at all times until no holder of the Inducement Warrants owns any Inducement Warrants or Inducement Warrant Shares. In the event that the Company fails to timely deliver to the Holder the Inducement Warrant Shares without restrictive legends, the Company has agreed to pay certain liquidated damages to the Holder. The Resale Registration Statement was timely filed and timely declared effective by the SEC.
A summary of the warrant activity (including certain warrants granted in August 2021, July 2022, December 2022, April 2023, August 2023, November 2023 and December 2024 as part of private offerings, all of which are equity-classified; see Note 11 - Stockholders’ (Deficit) Equity) during the years ended December 31, 2024 and 2023 is presented below:
| Number of Warrants | Weighted Average Exercise Price | Weighted Average Remaining Life in Years | Intrinsic Value | |||||||||||||
| Outstanding, January 1, 2023 | $ | $ | ||||||||||||||
| Issued | - | |||||||||||||||
| Exercised | ( | ) | - | - | ||||||||||||
| Outstanding, December 31, 2023 | $ | |||||||||||||||
| Issued | - | |||||||||||||||
| Exercised | ( | ) | - | - | ||||||||||||
| Expired | ( | ) | - | - | ||||||||||||
| Outstanding, December 31, 2024 | $ | $ | ||||||||||||||
| Exercisable, December 31, 2024 | $ | |||||||||||||||
A summary of outstanding and exercisable warrants as of December 31, 2024 is presented below:
| Warrants Outstanding | Warrants Exercisable | |||||||||||||
| Weighted | ||||||||||||||
| Average | ||||||||||||||
| Exercise | Number of | Remaining | Number of | |||||||||||
| Price | Shares | Life in Years | Shares | |||||||||||
| $ | ||||||||||||||
| $ | ||||||||||||||
| $ | ||||||||||||||
| $ | ||||||||||||||
| $ | ||||||||||||||
| $ | ||||||||||||||
| $ | ||||||||||||||
| $ | ||||||||||||||
F-47
NASDAQ Compliance
Nasdaq Listing Rule Non-Compliance
We have previously been out
of compliance with Nasdaq’s continued listing requirements due to our failure to maintain a minimum bid price of at least $
Also, on October 11, 2023,
the Company received written notice from Nasdaq notifying the Company that it was not in compliance with the shareholder approval requirements
set forth in Nasdaq Listing Rule 5635(d), which require prior shareholder approval for transactions, other than public offerings, involving
the issuance of
Additionally, on November
15, 2023, the Company received a letter from Nasdaq notifying the Company that it was not in compliance with the minimum stockholders’
equity requirement for continued listing on the Nasdaq Capital Market. Nasdaq Listing Rule 5550(b)(1) (the “Equity Rule”) requires
companies listed on the Nasdaq Capital Market to maintain stockholders’ equity of at least $
Nasdaq provided the Company until January 2, 2024 to submit to Nasdaq a plan to regain compliance. We submitted the plan to regain compliance in a timely manner, and on January 11, 2024, Nasdaq advised the Company that it has determined to grant the Company an extension to regain compliance with the Equity Rule.
The terms of the extension
were as follows: on or before May 13, 2024, the Company must have completed certain transactions described in greater detail in the compliance
plan, contemplated to result in the Company increasing its stockholders’ equity to more than $
Additionally, in either case the Company was required to disclose that Nasdaq will continue to monitor the Company’s ongoing compliance with the stockholders’ equity requirement and, if at the time of its next periodic report the Company does not evidence compliance, that it may be subject to delisting.
Separately, on May 14, 2024, the Staff provided us notice of our non-compliance with the audit committee requirements for continued listing on The Nasdaq Capital Market set forth in Listing Rule 5605(c)(2), which requires that listed companies maintain an audit committee of at least three independent directors. Nasdaq provided the Company a cure period in order to regain compliance as follows: until the earlier of the Company’s next annual shareholders’ meeting or May 7, 2025; or if the next annual shareholders’ meeting is held before November 4, 2024, then the Company must evidence compliance no later than November 4, 2024. In the event the Company does not regain compliance by the applicable date above, Nasdaq rules require the Staff to provide written notification to the Company that its securities will be delisted. At that time, the Company may appeal the delisting determination to a Hearings Panel. The Company is currently seeking out qualified independent directors to serve on the Company’s audit committee and plans to regain compliance with Listing Rule 5605(c)(2) in the near future.
F-48
While the Company was able to undertake some of the transactions described in the Equity Rule compliance plan, it was unable to regain compliance with the Equity Rule prior to the end of the plan period (May 13, 2024). As a result, on May 14, 2024, the Company received a delist determination letter from the Listing Qualifications department of The Nasdaq Stock Market LLC (the “Staff”) advising the Company that the Staff had determined that the Company did not meet the terms of the extension. Specifically, the Company did not complete its proposed transactions and was unable to file a Current Report Form 8-K by the May 13, 2024 deadline previously required by the Staff, evidencing compliance with the Equity Rule. On May 17, 2024, the Company requested an appeal of the Staff’s delisting determination, and on May 20, 2024, the Staff advised the Company that the delisting action referenced in the Staff’s determination letter had been stayed, pending a final written decision by the Nasdaq Hearings Panel (“Panel”). On July 2, 2024, the Company received notice that the Panel had determined to grant the Company’s request to continue its listing on The Nasdaq Stock Market, subject to the Company meeting certain conditions, including filing on or before July 31, 2024, a public disclosure describing the transactions undertaken by the Company to achieve compliance with Nasdaq’s continued listing rules and demonstrate long-term compliance with the Equity Rule and providing an indication of its equity following those transactions. On July 22, 2024, the Panel granted the Company’s request for additional time to achieve compliance with Nasdaq’s continued listing rules and demonstrate long-term compliance with the Equity Rule. Specifically, the Hearings Panel subsequently extended that deadline to September 30, 2024, and subsequently extended such deadline further to October 15, 2024, to regain compliance with the Equity Rule and to allow the continued listing of the Company’s common stock and warrants on The Nasdaq Stock Market through such date, subject to the Company’s compliance with the Equity Rule on or prior to such date.
As a result of the acquisition
of the Purchased Assets, issuance of the
If the Company’s common stock and public warrants are delisted, it could be more difficult to buy or sell the Company’s common stock and public warrants or to obtain accurate quotations, and the price of the Company’s common stock and public warrants could suffer a material decline. Delisting could also impair the Company’s ability to raise capital and/or trigger defaults and penalties under outstanding agreements or securities of the Company.
NOTE 12 - INCOME TAXES
The Company is subject to federal and state/provincial income taxes in the United States, Canada, and the United Kingdom and each legal entity files on a non- consolidated basis. The benefit of the pre-reorganization net operating losses of 180 LP were passed through to its owners.
The losses before income taxes consist of the following domestic and international components:
| For the Years Ended | ||||||||
| December 31, | ||||||||
| 2024 | 2023 | |||||||
| Domestic | $ | ( | ) | $ | ( | ) | ||
| International | ( | ) | ( | ) | ||||
| $ | ( | ) | $ | ( | ) | |||
F-49
The provision for income taxes consists of the following benefits:
| For the Years Ended | ||||||||
| December 31, | ||||||||
| 2024 | 2023 | |||||||
| Deferred tax benefits: | ||||||||
| Domestic: | ||||||||
| Federal | $ | $ | ||||||
| State | ||||||||
| International | ( | ) | ||||||
| Change in valuation allowance | ( | ) | ( | ) | ||||
| Net income tax benefit | $ | $ | ||||||
Certain deferred tax assets
are denominated in currencies other than the US dollar and are subject to foreign currency translation adjustments.
| For the Years Ended | ||||||||
| December 31, | ||||||||
| 2024 | 2023 | |||||||
| US Federal statutory rate | % | % | ||||||
| Difference between domestic and foreign federal rates | ( | )% | ( | )% | ||||
| State and provincial taxes, net of federal benefits | % | % | ||||||
| Permanent differences: | ||||||||
| Stock-based compensation | ( | )% | % | |||||
| Change in the fair value of derivatives and accrued issuable equity | ( | )% | % | |||||
| Other | % | ( | )% | |||||
| Change in valuation allowance | ( | )% | ( | )% | ||||
| Effective income tax rate | % | % | ||||||
Deferred tax assets and liabilities consist of the following:
| December 31, | ||||||||
| 2024 | 2023 | |||||||
| Deferred tax assets: | ||||||||
| Net operating loss carryforwards | $ | $ | ||||||
| Amortization | ||||||||
| Accrued compensation not currently deductible | ||||||||
| Stock compensation | ||||||||
| Accrued interest | ||||||||
| Reserve for uncollectible notes receivable | ||||||||
| Organizational costs deferred for tax purposes | ||||||||
| Other | ||||||||
| Deferred tax liabilities: | ||||||||
| Difference between book and tax basis related to: | ||||||||
| Technology license | ( | ) | ||||||
| Other | ( | ) | ||||||
| ( | ) | ( | ) | |||||
| Total of deferred tax assets and liabilities | ||||||||
| Valuation allowance | ( | ) | ( | ) | ||||
| Total of deferred tax assets and liabilities, net | $ | $ | ( | ) | ||||
F-50
The change in the valuation reserve for deferred tax assets consists of the following:
| For the Years Ended | ||||||||
| December 31, | ||||||||
| 2024 | 2023 | |||||||
| Beginning of period | $ | ( | ) | $ | ( | ) | ||
| Change in valuation pursuant to the tax provision | ( | ) | ( | ) | ||||
| True-up to a prior year’s tax return | ||||||||
| End of period | $ | ( | ) | $ | ( | ) | ||
As of December 31, 2024, the Company had net operating loss (“NOL”) carryforwards that may be available to offset future taxable income in various jurisdictions as follows:
| ● | Approximately $ |
| ● | Approximately $ |
| ● | Approximately $ |
The utilization of the domestic NOLs to offset future taxable income may be subject to annual limitations under Section 382 of the Internal Revenue Code and similar state statutes as a result of ownership changes.
The Company has assessed the
likelihood that deferred tax assets will be realized in accordance with the provisions of ASC 740. ASC 740 requires that such review considers
all available positive and negative evidence, including the scheduled reversal of deferred tax assets, projected future taxable income,
and tax planning strategies. ASC 740 requires that a valuation allowance be established when it is “more likely than not”
that all, or a portion of, deferred tax assets will not be realized. After the performance of such reviews as of December 31, 2024 and
2023, management believes that uncertainty exists with respect to future realization of its deferred tax assets and has, therefore, established
a full valuation allowance. The Company recorded increases in the valuation allowance of $
Management has evaluated and concluded that there were no material uncertain tax positions requiring recognition in the Company’s consolidated financial statements as of December 31, 2024 and 2023. The Company does not expect any significant changes in its unrecognized tax benefits within twelve months of the reporting date.
No tax audits were commenced or were in process during the years ended December 31, 2024 and 2023, nor were any tax related interest or penalties incurred during those periods. The Company’s tax returns filed in the United States, Canada, and the United Kingdom since inception remain subject to examination.
F-51
NOTE 13 - RELATED PARTIES
Accounts Payable – Related Parties
Accounts payable - related
parties were $
Research and Development Expenses - Related Parties
Research and Development Expenses
– Related Parties of $
General and Administrative Expenses - Related Parties
General and Administrative
Expenses – Related Parties during the years ended December 31, 2024 and 2023, were $
NOTE 14 - SUBSEQUENT EVENTS
On January 30, 2025, the Company
cancelled
On
February 5, 2025, the Company entered into a First Amendment to Separation and Release Agreement with James N. Woody, the Company’s
former Chief Executive Officer and director (the “First Amendment”). Pursuant to the First Amendment, Dr. Woody agreed
to amend the terms of that certain Separation and Release Agreement dated May 7, 2024, entered into between Dr. Woody and the Company,
to terminate the prior requirement of the Company set forth therein to pay Dr. Woody a bonus of $
The First Amendment also required Dr. Woody to enter into a Voting Agreement with the Company. Pursuant to the Voting Agreement, which was entered into on February 5, 2025, by Dr. Woody, the Company, and Blair Jordan, the Company’s Chief Executive Officer, solely for the benefit of the Company, Dr. Woody agreed to vote the Separation Shares as recommended by the Board of Directors of the Company, at any meeting of stockholders or via any written consent of stockholders, which may occur prior to February 5, 2026; the date after August 5, 2025, that Dr. Woody has sold all of the Separation Shares; or the date that the Company terminates the Voting Agreement. In order to enforce the terms of the Voting Agreement, and solely for the benefit of the Company, Dr. Woody provided Mr. Jordan (or his assigns) an irrevocable voting proxy to vote the Separation Shares pursuant to the guidelines set forth above at any meeting of stockholders or via any written consent of stockholders. The Voting Agreement also provides a restriction on Dr. Woody’s sale or transfer of any of the Separation Shares until August 5, 2025.
F-52
Effective on February
15, 2025, the Company appointed Eric R. Van Lent as the Chief Accounting Officer (Principal Accounting/Financial Officer) of
the Company (the “Appointment”), which Appointment was effective as of the same date. As a result of the
Appointment, Blair Jordan, the Chief Executive Officer (Principal Executive Officer) of the Company, who had served as the
Principal Accounting/Financial Officer of the Company since October 16, 2024, stepped down from such role, also effective on
February 15, 2025. Effective on February 15, 2025, the Company entered into an Executive Consulting Agreement dated January 30, 2025
with Mr. Van Lent and EVL Consulting, LLC (an entity owned by Mr. Van Lent)(“EVL Consulting” and the
“EVL Consulting Agreement”). Pursuant to the EVL Consulting Agreement, the Company agreed to engage EVL
Consulting to provide the services of Mr. Van Lent to the Company as Chief Accounting Officer of the Company. The EVL Consulting
Agreement has a term through July 30, 2025, unless otherwise terminated pursuant to the terms of the agreement (discussed
below) and provides for Mr. Van Lent to act as Chief Accounting Officer of the Company, and to be paid $
The Company has the right
to terminate the EVL Consulting Agreement at any time, provided that it pays EVL Consulting $
The Company is also able to terminate the EVL Consulting Agreement at any time, without notice upon: (a) the death or physical or mental incapacity of Mr. Van Lent if as a result of which Mr. Van Lent is unable to perform services for a period in excess of 30 days; (b) in the event Mr. Van Lent or a related party to Mr. Van Lent ceases to own or control 100% of EVL Consulting; or (c) “just cause”, which means any of the following events: (i) any material or persistent breach by EVL Consulting or Mr. Van Lent of the terms of the agreement; (ii) the conviction of EVL Consulting or Mr. Van Lent of a felony offence, or the equivalent in a non-American jurisdiction, or of any crime involving moral turpitude, fraud or misrepresentation, or misappropriation of money or property of the Company or any affiliate of the Company; (iii) a willful failure or refusal by EVL Consulting or Mr. Van Lent to satisfy its respective obligations to the Company under the agreement including without limitation, specific lawful directives, reasonably consistent with the agreement, or requests of the Board; (iv) any negligent or willful conduct or omissions of EVL Consulting or Mr. Van Lent that directly results in substantial loss or injury to the Company; (v) fraud or embezzlement of funds or property, or misappropriation involving the Company’s assets, business, customers, suppliers, or employees; (vi) any failure to comply with any of the Company’s written policies and procedures, including, but not limited to, the Company’s Corporate Code of Ethics and Insider Trading Policy, provided that subject to certain limited exceptions, we must first give written notice to EVL Consulting and Mr. Van Lent, as applicable, advising them of the acts or omissions that constitute failure or refusal to perform their obligations and that failure or refusal continues after EVL Consulting and Mr. Van Lent, as applicable, has had thirty (30) days to correct the acts or omissions as set out in the notice.
If the Company terminates the EVL Consulting Agreement for just cause, we are required to pay EVL Consulting any unpaid fees and/or unpaid and unreimbursed expenses accrued but unpaid prior to the effective termination date.
On February 20, 2025, the Company entered into an Executive Consulting Agreement with Mr. Blair Jordan dated February 21, 2025, the Company’s Chief Executive Officer and director, and Blair Jordan Strategy and Finance Consulting Inc. (an entity owned by Mr. Jordan)(the “Jordan Agreement”). The Jordan Agreement replaced and superseded a prior Executive Consulting Agreement with Mr. Jordan and Jordan Consulting dated May 7, 2024 (the “Prior Agreement”).
Pursuant to the Jordan Agreement, the Company agreed to continue to engage Jordan Consulting to provide the services of Mr. Jordan to the Company as Chief Executive Officer of the Company. The Jordan Agreement has a term beginning effective January 1, 2025, and continuing through December 31, 2026, unless otherwise terminated pursuant to the terms of the agreement (discussed below), provided that in the event that the parties have not agreed to an extension or termination of the Jordan Agreement with at least 30 days written notice at the end of the term, the agreement automatically renews for successive terms of one year upon the expiration of the primary term or any renewal.
F-53
The
Jordan Agreement provides for Mr. Jordan to act as Chief Executive Officer of the Company, and to be paid $
The
agreement also allows the Company to pay Mr. Jordan or Jordan Consulting an incentive bonus of up to
The
Company also agreed to grant
The Jordan Agreement includes customary confidentiality, non-disclosure and proprietary right requirements of Jordan Consulting and Mr. Jordan, and a prohibition on Jordan Consulting and Mr. Jordan competing against us during the term of the agreement. Jordan Consulting may terminate the Jordan Agreement at any time for “good reason” as described in the Jordan Agreement, subject to certain cure rights; at any time without “good reason”; and upon expiration of the term or any renewal.
The Company may terminate the Jordan Agreement at any time for “just cause” (as described below) and for any reason other than “just cause”. “Just cause” means the occurrence of any of the following events: (i) any material or persistent breach by Jordan Consulting or Mr. Jordan of the terms of the agreement; (ii) the conviction of Jordan Consulting or Mr. Jordan of a felony offence, or the equivalent in a non-American jurisdiction, or of any crime involving moral turpitude, fraud or misrepresentation, or misappropriation of money or property of the Company or any affiliate of the Company; (iii) a willful failure or refusal by Jordan Consulting or Mr. Jordan to satisfy its respective obligations to the Company under the agreement including without limitation, specific lawful directives, reasonably consistent with the agreement, or requests of the Board; (iv) any negligent or willful conduct or omissions of Jordan Consulting or Mr. Jordan that directly results in substantial loss or injury to the Company; (v) fraud or embezzlement of funds or property, or misappropriation involving the Company’s assets, business, customers, suppliers, or employees; (vi) any failure to comply with any of the Company’s written policies and procedures, including, but not limited to, the Company’s Corporate Code of Ethics and Insider Trading Policy, provided that subject to certain limited exceptions, we must first give written notice to Jordan Consulting and Mr. Jordan, as applicable, advising them of the acts or omissions that constitute failure or refusal to perform their obligations and that failure or refusal continues after Jordan Consulting and Mr. Jordan, as applicable, has had thirty (30) days to correct the acts or omissions as set out in the notice, if such acts are correctable.
The
Company is also able to terminate the Jordan Agreement at any time, without notice upon: (a) the death or physical or mental incapacity
of Mr. Jordan if as a result of which Mr. Jordan is unable to perform services for a period in excess of 60 days; or (b) in the event
Mr. Jordan or a related party to Mr. Jordan ceases to own or control
If the agreement is terminated by Jordan Consulting for “good reason”, or by the Company without “just cause” (other than due to death or disability), Jordan Consultant is required to be paid, in a lump sum on the thirtieth day following such termination, a severance payment equal to (i) half of the then current annualized Fee, in the event such termination occurs during the first twelve months of the agreement and 100% of the then current annualized Fee, in the event such termination occurs after the first twelve months of the Agreement, together with all outstanding expenses and pro-rated Fee (through the date of termination); (ii) any unpaid annual cash bonus in respect of any completed fiscal year that has ended prior to the date of such termination with such amount determined based on actual performance during such fiscal year as determined by the compensation committee; and (iii) immediate vesting of any and all equity or equity-related awards previously awarded. Any equity awards that vest based on various performance metrics will be vested only if such performance metrics have been met at the time of termination of service and will be determined solely by the Compensation Committee.
F-54
If the agreement is terminated without “good reason” by Jordan Consulting or for “just cause” by the Company, Jordan Consulting is entitled to the Accrued Liabilities (as defined below), and any equity awards or equity-related awards that are not vested as of the date of termination will be cancelled and forfeited and any vested awards will be exercisable pursuant to their terms.
If the agreement is terminated due to Mr. Jordan’s death or disability, Jordan Consulting or Mr. Jordan’s estate or his beneficiaries, as the case may be, will be entitled to receive (i) any accrued but unpaid Fee through the date of termination, any unpaid or unreimbursed expenses incurred in accordance with the terms of the agreement, (collectively, the “Accrued Liabilities”); (ii) any unpaid annual cash bonus in respect of any completed fiscal year that has ended prior to the date of such termination, with such amount determined based on actual performance during such fiscal year as determined by the Company’s Compensation Committee on the sixtieth day following termination; (iii) a lump sum payment of any non-discretionary annual cash bonus that would have been payable based on actual performance with respect to the year of termination in the absence of Mr. Jordan’s death or disability, pro-rated for the period that Mr. Jordan worked prior to his death or disability, and payable at the same time as the bonus would have been paid in the absence of Mr. Jordan’s death or disability; and (iv) immediate vesting of any and all equity or equity-related awards previously awarded to Jordan Consulting, irrespective of the type of award.
As a condition precedent to payment of any amount or provision of any benefit to Mr. Jordan upon termination (the “Severance Benefits”), Jordan Consulting and Mr. Jordan or Mr. Jordan’s estate, as applicable, shall execute and shall not rescind, a release in favor of the Company and all related companies, individuals, and entities, in a form satisfactory to the Company.
Upon termination of the agreement or for any reason other than “good reason” by Jordan Consulting or the Company without “just cause”, Jordan Consulting and Mr. Jordan agreed that, for a period ending six months from the date of termination, Jordan Consulting and Mr. Jordan shall not (except on behalf of the Company or with the prior written consent of the Company), directly or indirectly, compete with the Company for a period of one year, neither Mr. Jordan, nor Jordan Consulting shall solicit employees or consultants of the Company, each as discussed in greater detail in the Jordan Agreement.
On
February 21, 2025, the Company entered into a Mutual Settlement and General Release Agreement with Dr. Marlene Krauss, the former Chief
Executive Officer and director of KBL and KBL IV Sponsor, LLC. Pursuant to the Settlement Agreement, the Company agreed to (1) pay $
The Settlement Agreement also required Dr. Krauss to enter into a Voting Agreement with the Company. Pursuant to the Voting Agreement, which was entered into on February 21, 2025, by Dr. Krauss, the Company, and Blair Jordan, the Company’s Chief Executive Officer, solely for the benefit of the Company, Dr. Krauss agreed to vote the Settlement Shares as recommended by the Board of Directors of the Company, at any meeting of stockholders or via any written consent of stockholders, which may occur prior to August 21, 2025. In order to enforce the terms of the Voting Agreement, and solely for the benefit of the Company, Dr. Krauss provided Mr. Jordan (or his assigns) an irrevocable voting proxy to vote the Settlement Shares pursuant to the guidelines set forth above at any meeting of stockholders or via any written consent of stockholders. The Voting Agreement also provides a restriction on Dr. Krauss’s sale or transfer of any of the Settlement Shares until August 21, 2025 and provides for mutual general releases of the parties.
On
March 7, 2025, the Compensation Committee and the Board approved the grant of
On March 27, 2025, Elray Resources, Inc., the sole holder of the Series
B Convertible Preferred Stock of the Company converted all
F-55